GW Pharmaceuticals
The UK biopharmaceutical company GW Pharmaceuticals, a global developer of medical cannabis products, has released 2.1 million shares on the US stock exchange,
raising US$345m.
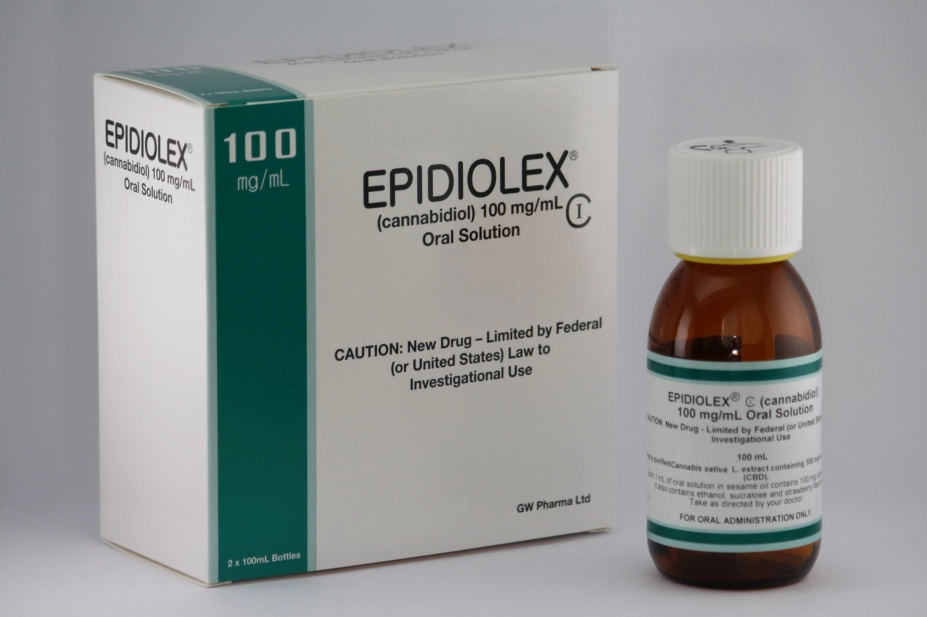
The move comes four months after Epidiolex, an oral solution of purified cannabidiol (CBD) manufactured by GW Pharmaceuticals’s subsidiary Greenwich Biosciences, became the first cannabis-derived medicine approved for use in the United States for the treatment of seizures caused by Lennox-Gastaut and Dravet syndromes – both severe forms of epilepsy.
The release of shares follows the company’s work to receive marketing authorisation from the European Medicines Agency for another CBD product, which it hopes to be made available across Europe in 2019.
The US Food and Drug Administration’s decision to approve Epidiolex came just before Sajid Javid, the UK’s home secretary, announced in July 2018 that cannabis-derived medicinal products should be moved from Schedule I to Schedule II of the 2001 Misuse of Drugs Regulations Act — the first step towards clinicians being able to prescribe these kinds of products.
The decision to reschedule cannabis-derived products was based on conclusions drawn by the UK government’s official drug advisers and Dame Sally Davies, the chief medical officer for England, who believed that there was sufficient evidence that such medicines offer therapeutic benefit for some conditions.
A spokesperson for GW Pharmaceuticals said: “The decision to raise finance was about maintaining our financial strength as we execute the launch of Epidiolex and as we maintain investment in new product development.”
The spokesperson also confirmed that the fundraising was not linked to the UK government’s review of cannabis scheduling.