
shutterstock.com
A career as a Qualified Person (QP) is both varied and interesting. The role comes with a high level of responsibility but also provides flexibility, excellent remuneration and a good work–life balance.
What is a Qualified Person?
Most QPs in the UK are employed by, or contracted to work in, the pharmaceutical industry or work in licenced NHS manufacturing units. The lead QP is usually head of (or holds a very senior role in) the quality assurance team.
The QP is the person who accepts and signs off batches of medication to be released to the market or used in a clinical trial (for humans or animals). The role has a legal responsibility to ensure that every batch of a medicinal product released onto the market complies with its specification — i.e. complies with its marketing authorisation and has been made according to good manufacturing practice. The QP will then release the batch onto the market. Each manufacturer of a human or veterinary medicinal product requires at least one QP to be named on their manufacturer’s authorisation. The same processes apply in clinical trials.
What do QPs do?
The QP will usually spend their day working as part of the quality assurance team. Some of this time may be spent reviewing batch records and any deviations occurred during manufacture or testing, with a view to certifying that the batch may be released for sale. Sometimes this may not be straightforward and the QP will request additional actions, such as further quality control analysis or even rework. On rare occasions the QP will have to state that the batch must be destroyed. Other activities include meeting regulatory colleagues, clients and marketing teams in project meetings, which could include discussions such as improving packaging or updating a formulation for the manufacture and testing of a product. Another day could be spent auditing the manufacturer of an active substance and checking that it is purchased from the site named on the relevant marketing authorisation. The QP usually leads staff training in good manufacturing practice.
One myth about working in the pharmaceutical industry is that a PhD is a prerequisite — this is not so
QPs do not usually work on weekends or bank holidays. Many QPs will be part-time employees of a pharmaceutical company or manufacturing unit, although more often they will be contractors who assist the in-house QP, attending manufacturing sites on a set number of days per week or month, and/or work from home.
How do pharmacists become QPs?
One myth about working in the pharmaceutical industry is that a PhD is a prerequisite — this is not so. Current legislation only requires membership of the Royal Pharmaceutical Society (RPS), the Royal Society of Chemistry (RSC) or Royal Society of Biology (RSB) with an acceptable, relevant qualification[1],[2]
.
QP assessment in the UK is managed jointly by these three professional bodies and there is useful information on their websites. The General Pharmaceutical Council is not involved.
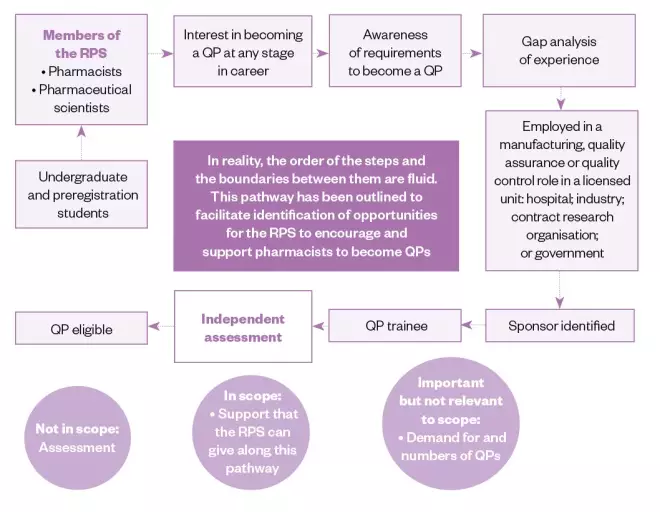
Figure: Royal Pharmaceutical Society Qualified Person career pathway
Source: Royal Pharmaceutical Society
The career pathway for a pharmacist or pharmaceutical scientist to become a Qualified Person
QP: Qualified Person; RPS: Royal Pharmaceutical Society
Getting started
To qualify as a QP in the UK, members of the RPS need to complete at least one years’ practical experience working day-to-day in a licenced unit under the supervision of their QP sponsor, and complete a prescribed syllabus detailed on the RPS website. Members of the RSC or RSB require a minimum of two years’ practical experience. Members should apply to their own professional society.
Unless already employed in a licensed unit, individuals may need to look for a new job. This may necessitate a pay cut for those coming from community or hospital pharmacy. However, in the longer term the salary of a QP is probably much higher than in either of these roles (except for the very most senior managers or owners).
The QP sponsor will certify that trainees have adequate knowledge and certify that the experience requirement has been met. It may be simpler to think of the QP training year as being like the preregistration year.
Once the training year is complete, trainees will need to submit an ‘Application form for certification of eligibility’, in order to sit the final oral assessment (which is by a viva). The application must demonstrate how applicants have met each area of the QP Study Guide and requires sign-off by the QP sponsor.
After a successful viva, trainees will be certified as ‘eligible to be a QP’. This means that individuals are able to be named as a QP on a manufacturing authorisation.
Those wishing to become a QP also often choose to study a relevant MSc or take several modules from a suitable provider.
Recruitment
Job advertisements for trainee QPs are not commonplace. Instead, it is usual for a suitable candidate to be selected internally from within quality assurance, quality control or good manufacturing practice teams as a person with the right attitude and attributes to be a QP. Any trainee QP or any other suitable ‘stepping stone’ jobs within the pharmaceutical industry may not be advertised in the same way as other pharmacy jobs. Recruitment agencies are often used to find suitable people and many of these will ‘headhunt’ using social media platforms such as LinkedIn.
There is a shortage of QPs in the UK and, as such, the salaries and contract day rates are rising rapidly to keep pace with demand
Salaries for QPs in the UK generally begin at about £50,000 per year and move upwards of £70,000–£100,000 per year for an employee (depending upon experience), and from approximately £90–£100 per hour for a self-employed contractor QP. At the time of writing, there is a shortage of QPs in the UK and, as such, the salaries and contract day rates are rising rapidly to keep pace with demand. However, the effect Brexit will have on QPs’ salaries is unknown.
Necessary knowledge and skills
Pharmacist QPs are in a special position to be able to provide insight into the needs of the patient when they are working to sort out problems in the manufacturing and analysis of a batch of medicines and in drug development or clinical trials. The most important skill required for the role is attention to detail, but a QP must also be able to handle pressure — especially when things go wrong — and be able to make decisions under pressure, even if theie boss does not like it.
CV and interview tips
The big advantage that a pharmacist has over other scientists is the breadth of the pharmacy degree, which includes study of microbiology, formulation and law. However, for a pharmacist wanting to become a trainee QP, one of the best attributes is a knowledge of clinical pharmacy, dealing with the public and subjects such as acceptability to a patient of particular dosage form and packaging. During an interview, applicants should be ready to expand on all of these themes and demonstrate knowledge of any products manufactured by the particular company or NHS establishment.
More information on pharmacist QPs can be found on the RPS Qualified Persons site or via RPS Support on 0845 2572570 .
Royal Pharmaceutical Society 16th Joint QP Symposium
For a face-to-face update on the latest changes in the QP sector, to meet industry experts and network with QPs, book onto the Royal Pharmaceutical Society’s
16th Joint QP Symposium taking place on 16 May 2019.
About the authors:

Carol Candlish
Carol Candlish is the Qualified Persons (QP) lead for the Industrial Pharmacists Forum — one of the professional groups of the Royal Pharmaceutical Society (RPS). She works as a consultant in the pharmaceutical industry and as a contract QP and Responsible Person.

Kina Vyas
Kina Vyas is a professional development pharmacist at the RPS.
References
[1] The European Parliament & The Council of the European Union. Directive 2001/83/EC of the European Parliament and of the Council of the European Union on the community code relating to medicinal products for human use. 2001. Available at: https://ec.europa.eu/health/sites/health/files/files/eudralex/vol-1/dir_2001_83_consol_2012/dir_2001_83_cons_2012_en.pdf (accessed February 2019)
[2] The European Parliament & The Council of the European Union. Directive 2001/82/EC of the European Parliament and of the Council of the European Union on the community code relating to veterinary medicinal products. 2001. Available at: https://ec.europa.eu/health/sites/health/files/files/eudralex/vol-5/dir_2001_82_cons2009/dir_2001_82_cons2009_en.pdf (accessed February 2019)

