As a researcher, imagine being able to launch your own smartphone app and recruit thousands of participants to a clinical trial, quickly, from all corners of the globe and at a fraction of the cost of traditional methods. Then being able to collect data daily, and in real-life situations, rather than in the confined environment of a doctor’s office.
This is the potential of ResearchKit, Apple’s new software designed to get iPhone users involved in medical research. Turning iPhones into diagnostic devices that collect scientific data could be an exciting new frontier for medical studies. But there are inherent weaknesses in this way of gathering data, which will need to be overcome if the iPhone is to become a fully-fledged medical research tool.
“I think we’ll learn a lot over this coming year that’ll tell us more about what we have on our hands,” says John Wilbanks, chief commons officer at Sage Bionetworks, a Seattle, Washington-based non-profit biotech company that worked closely with Apple and medical researchers to build the software platform. “We’ll see doctors and app developers coming together and kicking the tyres, so to speak — really testing the capabilities to see how far they can take this.”
The software is in its early stages, but for now, researchers looking to launch a study app (which, as for any study, should be approved by medical ethicists and an institutional review board) can work with app developers to tailor three customisable modules. In ‘participant consent’, they can upload videos to explain their research and what volunteers will be asked to do, and they can create quizzes to test understanding before getting signatures of consent. In ‘surveys’, they can use a pre-built user interface to ask qualitative questions. And in ‘active tasks’, they can tap into the iPhone’s sensors — for example, the accelerometer, microphone, gyroscope and GPS sensors — to gather quantitative measurements of a user’s gait, motor impairment, fitness, speech and memory.
The potential
The software is open-sourced, and Apple is encouraging developers to create and share new modules. In the meantime, to showcase ResearchKit’s capabilities the company launched the platform with five pre-built apps. The apps are only available from the US App Store, but the company plans to extend access to other countries soon.

Source: John Wilbanks
John Wilbanks worked closely with Apple to build the software platform
“I’m probably a bit biased but I think the mPower study is the coolest in terms of showing the out-of-the-box potential of these things,” says Wilbanks, referring to a Parkinson’s disease app developed by Sage Bionetworks and researchers at the University of Rochester in New York. The other apps are designed to study breast cancer, asthma, heart disease and diabetes (see ‘ResearchKit apps: the first five’).
In mPower, once participants have consented to their involvement, they are asked to perform daily tasks within the app. They tap two circles as fast as possible as a test of dexterity. They speak into the microphone to check for signs of early disease. They perform walking tests with their phone in their pocket to test gait and balance.
“These are all assessments that the doctor would do in the clinic,” says Todd Sherer, chief executive officer of The Michael J Fox Foundation, a charity based in New York dedicated to finding a cure for Parkinson’s disease. The foundation is working with the mPower investigators to help them figure out how best they can use their dataset, combining it with data from Fox Insight — an online Parkinson’s study — to validate the power of the two approaches.
With this technology, we can now collect data daily and in real-life situations to get a better handle on Parkinson’s disease and its subtypes
“In a traditional observational study, the doctor would see the patient only every, say, three months, which really limits how much we can learn,” Sherer says. “With this technology, we can now collect data daily and in real-life situations to get a better handle on [Parkinson’s] disease and its subtypes.” The hope is that detailed, real-world information will help scientists figure out new treatments.
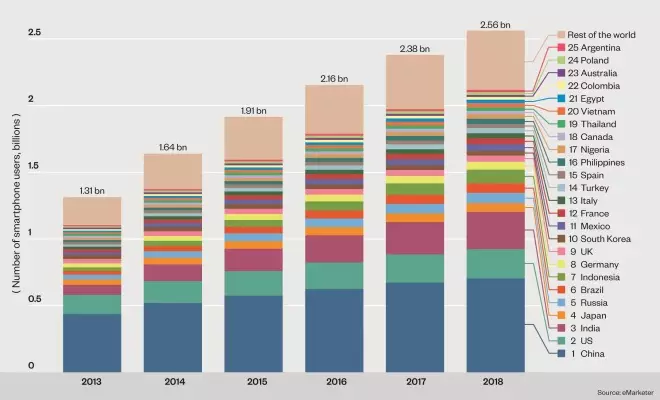
Global Reach
Almost 2 billion people worldwide – more than a quarter of the global population – currently use smartphones, a figure estimated to rise to 2.56 billion by 2018. Although Apple’s ResearchKit is currently only available on iOS, a similar product on the Android platform is likely to be developed at some point in the future. The chart below, which shows the top 25 countries ranked by smartphone users in 2014, gives an idea, therefore, of the potential reach of studies conducted using smartphone technology
The benefits
Frequent data collection is one of the two main benefits of doing studies this way. The other is potential reach. “The initial take-up was a lot better than we’d expected,” says Wilbanks of mPower. In fact, within six hours of the app launching, nearly 7,500 potential participants had signed up. Researchers at Stanford University awoke the day after launching their app, a heart disease study, to find that 11,000 people had signed up. A number of participants, they said in subsequent media reports, which would usually take a year and 50 medical centres to accrue.
“I think a lot of that spike we got is because when Apple launches something a lot of people pay attention,” Wilbanks says. “Enrolment numbers will look different when you don’t have the world watching. But it definitely shows there is interest at the volunteer’s end.”
There is interest from the health technology sector, too. Probably not by chance, Apple is entering the field at a very busy time: in 2014, investors pumped more than US$4bn into health technology start-ups, more than the amount invested in the previous three years combined. Devices such as Bluetooth asthma inhalers, wearable health monitors and glucose-monitoring contact lenses — a Google project in the early stages of development — could all one day connect with ResearchKit apps.
Software developers are also set to benefit. “It’s the improvements in the back-end, geeky stuff that excites us,” says Doug Bain, chief executive officer of eClinicalHealth in Stirling, UK, a consulting and software development company that built Clinpal, an online platform for clinical-trial engagement. The benefits of ResearchKit, he says, are that it standardises the way that data can be obtained from and exchanged with participants. “That method of interfacing — the handshake, the dialogue — that will always be the same with ResearchKit apps, meaning we can offer a less-expensive and faster service in the future. Not having to build that interface from scratch every time could shave tens of thousands of pounds off a project.
That method of interfacing — the handshake, the dialogue — that will always be the same with ResearchKit apps, meaning we can offer a less-expensive and faster service in the future
Another advantage of having the development process streamlined, Bain says, is that medical software need no longer be the province of only the biggest institutions or companies. Groups of patients with rare diseases and links to research may be able to launch their own apps to gather data for all-important hypothesis-generating work — work that drug companies or large institutions may not have the financial incentive to do themselves.
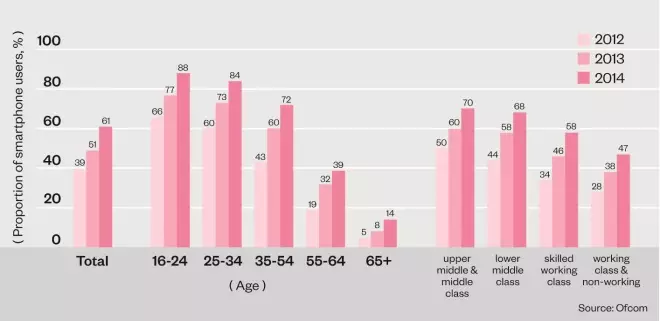
Demographics of UK smartphone users
Smartphone users in the UK tend to be younger and more affluent, so clinical trials conducted using smartphone technology may struggle to recruit participants from a diverse demographic.
The drawbacks
Not everyone is so enthusiastic about the potential of ResearchKit. One of the main criticisms levied at the platform is that trial participants are limited to those who own an iPhone, a population that polls have shown is more likely to be richer and better educated than Android users[1]
. This selection bias may affect the ability of researchers to gather good quality data on, for instance, diet-related heart disease, which disproportionately affects poorer segments of society.
Lisa Schwartz, a professor of medicine at the Dartmouth Institute for Health Policy and Clinical Practice in Lebanon, New Hampshire, sees more fundamental problems with the software. “There have been so many hyperbolic quotes around the launch, saying that it’s going to lead to better health,” she says, “but, on its own, it’s very unclear to me how it’s going to do that.”
There have been so many hyperbolic quotes around the launch, saying that it’s going to lead to better health but, on its own, it’s very unclear to me how it’s going to do that
Schwartz worries that data from volunteers self-reporting about their symptoms “is very susceptible to bias”. “Unless the study has a standardised way of vetting fundamental information — do the people actually have the disease? — it is a big black box.” The system relies on trusting that people are who they say they are, she says. “Trust without vetting is a very weak research design.”
She points out another area of uncertainty about the quality of data collected: people get tired of answering lots of questions. “So it might be that these questions are valid when you ask them every month, but what about when you ask them every day? At some point, people fatigue or lose interest, they may just click through screens rather than really try to answer.”
Wilbanks concedes that these problems are major kinks that need to be ironed out, but stresses that new challenges should stimulate researchers to think differently about how they can test research questions.
“If you try and plug a traditional observational study into an app, it might not work,” he says. “Or at least the output might not be that interesting.” He predicts that initially researchers will use the apps for low-risk observational studies, but looks forward to “some more interesting, experimental stuff”.
More ethical and regulatory work is needed before ResearchKit apps can be considered for use in interventional or randomised trials, Wilbanks believes. But he says that, if linked with other technologies, observational studies can be powerful. He sees the greatest potential for development in linking these apps with electronic health records, clinical and laboratory test values, and genetic screens.
Tying in genetic data might seem farfetched but is probably not that far off. In 2014, Google launched its genomes project, Google Genomics, a cloud storage platform for genetic sequences that it hopes will help lead to new medical discoveries. And a few weeks after Apple launched ResearchKit, Facebook unveiled Genes for Good, an app developed by researchers at the University of Michigan in Ann Arbor — users fill in surveys and are then sent a spit kit to have their genomes sequenced. The idea is that scientists can use the data to find correlations between participants’ survey answers and their underlying genetic variation.
Data security
But the more data that are collected, the more some people worry. “As these technologies get more pervasive,” says Stephen Fairclough, a professor of psychophysiology at Liverpool John Moores University, UK, “the potential from a science perspective gets more interesting in some ways but it also gets potentially more dodgy in other ways.”

Source: Apple
The ResearchKit app has been developed for iPhones
Apple has been clear that it believes the information collected within the first five ResearchKit apps is safe. It has made much of the fact that it does not see or store the data and that information is all anonymised before leaving the phone. “Apple saying they’re not going to see the data is one thing but it’s going to be a lot down to the integrity of those institutions making the apps that do collect the data,” Fairclough says.
Apple saying they’re not going to see the data is one thing but it’s going to be a lot down to the integrity of those institutions making the apps that do collect the data
Participants do have some control over their data, choosing to share it with only the investigators of the study they sign up for or with a wider group of researchers, an option that around 70% of participants have thus far opted for. Sage Bionetworks is storing all the data for the first five apps and, in April 2015, IBM announced a partnership with Apple to offer its secure cloud storage services for future ResearchKit apps. IBM says that the data — which, again, should not be identifiable — might also be used for research by insurance companies.
However, protecting self-reported data, Fairclough says, is only one aspect of concern. “The other is when you start donating your movement data and heart rate data and so on.” He worries that collating large amounts of information could reveal things that the people submitting the data are not even aware of, such as “information about their lifestyle choices or whether they have a disease such as Parkinson’s”. And, he continues, “I find it hard to believe that this whole technology push into health is not eventually going to tie up with the health insurance industry in America at some point.”
At present, the profit motive is being kept at arms’ length, with the terms and conditions for developers signing up to ResearchKit banning the commercial resale of any data collected. However, there does not seem to be a restriction on, for example, drug companies making an app themselves — provided they get ethics approval — and using the data gathered to profit directly.
Fairclough is not entirely negative. “For the time being, this is a very interesting development,” he says of technology giants such as Apple testing the waters for their ability to help in science and health. But he thinks we need to start asking more questions. For example, Fairclough says, the health data collected by tools like ResearchKit could be seen in another light. “I’m not saying that there’s anything subversive going on, but… consumer behaviour data is valuable to big business,” he says. “Health is a very unthreatening face for these technologies, and one that we will probably welcome into our homes and lives. The discussion about what else these things could be used for should start now.”
ResearchKit apps: the first five
Share the Journey
Aim: To figure out why some survivors of breast cancer recover faster than others, why the severity of symptoms during recovery varies with time and what can be done to help speed up rehabilitation.
How: Participants fill out a diary in the app describing their recovery. Using this diary and the handset’s sensors, the app tracks five common symptoms that can linger after cancer treatment: fatigue, mood and cognitive changes, trouble sleeping and a reduction in activity level.
Developed by: The Dana-Farber Cancer Institute; Perelman School of Medicine, University of Pennsylvania; University of California, Los Angeles Jonsson Comprehensive Cancer Center; and Sage Bionetworks.
Asthma Health
Aim: To help people with asthma to adhere to treatment and to better understand and avoid triggers that can affect their health. The data gathered will also enable researchers to learn more about this complex disease at a population level.
How: Participants record their asthma symptoms and triggers (for example, colds, physical activity, strong smells, or house dust). They also record peak flow measurements, medicine use, hospital and emergency department visits and the number of steps they take a day through the Apple Health app pedometer function. The app then sends reminders to participants about when to take their medications and, using GPS sensors, informs them about local air quality conditions.
Developed by: The Icahn School of Medicine at Mount Sinai; Weill Cornell Medical College; and digital therapeutics company LifeMap Solutions.
MyHeart Counts
Aim: To improve understanding of which lifestyle factors lead to good heart health — specifically the relationship between physical activity and heart disease. Researchers also want to learn more about the potential of mobile health technology to encourage people to look after their heart and take regular activity.
How: Either through the new Apple Watch (released at the same Apple media event as ResearchKit), the iPhone’s sensors, or an external fitness-tracking device, users measure their daily activity and heart rate. They can also input their cholesterol and blood pressure information to get an estimate of their future risk of heart disease or stroke.
Developed by: The Stanford University School of Medicine in collaboration with the American Heart Association; and the University of Oxford.
mPower
Aim: To track Parkinson’s disease symptoms and learn more about disease subtypes, why and when patients have good and bad days, and more about the natural history of the condition. mPower stands for ‘mobile Parkinson observatory for worldwide, evidence-based research’.
How: The app asks users to perform tests for walking, speech, memory and dexterity (finger tapping), and to complete symptom diaries.
Developed by: The University of Rochester; Beijing Institute of Geriatrics; and Sage Bionetworks.
GlucoSuccess
Aim: To build a database of health behaviours and glucose values for people with type 2 diabetes. Also to enable participants with the disease to monitor their progress and learn more about behaviours that affect their health.
How: Participants track activity, medicine use, blood glucose values, body weight and waist measurements. They can also log finger-stick blood glucose measurements and diet information using the nutrition-tracking app LoseIt.
Developed by: The Center for Assessment Technology and Continuous Health (CATCH) at Massachusetts General Hospital.