MAG / The Pharmaceutical Journal
In April 2014, a 13-year-old girl from Newcastle, England, died from an asthma attack. Her GP had not referred her to a specialist or increased her medication despite her worsening condition in the months leading up to her death. The coroner’s report says she was seen by doctors in primary and secondary care 47 times in the last four years of her life, but offered only treatment for acute attacks and no long-term care plan[1]
. Her death preyed on anxieties among respiratory specialists that asthma care is failing patients.
This tragedy was no isolated case. A national review of asthma deaths that same year found that two-thirds of the 1,200 asthma deaths that happen in the UK each year could be avoided with simple routine care[2]
.
“Our current model for asthma care is no longer fit for purpose,” says Joseph Clift, senior policy officer at charity Asthma UK. The vast majority (85%) of people with asthma are treated in the UK’s overstretched primary care service. “One of the core parts of asthma care is an annual asthma review with GPs, but lots of people don’t value it and most end up not going,” he adds. GPs, too, struggle to eke out value in the review, which usually takes mere minutes: a patient might not accurately recall how their asthma was controlled over the preceding year; clunky, paper-based reports are difficult to analyse or share.
Our current model for asthma care is no longer fit for purpose
The upshot is an apathy among asthma patients, with many not adhering to their prescribed medication — estimates of non-adherence range between 30% and 70% of patients.
“Control your asthma better and you’ll have a better quality of life and be less likely to have an attack,” says Clift. This is where smart inhalers could come in. Currently only available as part of a clinical trial, smart inhalers are Bluetooth-enabled devices that detect inhaler use, remind patients when to take their medication, and gather data that can guide care.

Source: Courtesy of Joseph Clift
Joseph Clift, senior policy officer at charity Asthma UK, says it’s important that smart inhalers are designed with health systems and patients in mind so that they can give maximum benefit.
“Smart inhalers certainly have promise in helping patients better adhere to their medication and keep on top of their asthma,” says Clift. “It’s early days but it’s important that they are designed with health systems and patients in mind so that they can give maximum benefit,” he adds.
Smart inhalers certainly have promise in helping patients better adhere to their medication and keep on top of their asthma
In a report published in January 2017, ‘Smart asthma’, Asthma UK outlined its concerns about the need for health systems to plan ahead so that the full potential of smart inhaler technology can be realised[3]
. Future inhalers might alert users to potential environmental triggers and, if used widely enough, big data analysis could help researchers answer important questions about asthma and other respiratory disorders that require inhaled drugs, such as chronic obstructive pulmonary disease (COPD). Managing competition between device manufacturers so that the devices and the data they gather are interoperable will be important.
Getting the basics right
Patients with asthma need two types of inhaled medication: to prevent attacks they take inhaled corticosteroids that dampen the inflammation that drives the disease, and when symptoms such as coughing or wheezing begin they use bronchodilators, known as reliever or rescue medication. For now, smart inhalers record only actuation — a time and date stamp of when a patient uses their inhaler. Several companies are developing products that clip onto existing inhalers; leading the pack are United States-based company Propeller Health and New Zealand-based Adherium. Both have data from clinical trials that suggest their products might help patients.
A randomised controlled trial of 495 patients showed that people using Propeller’s add-on smart inhaler and smart phone apps over 12 months used less reliever medication, had more reliever-free days and had improved asthma control[4]
. Meanwhile, a randomised control trial of 220 school-aged children showed that Adherium’s smart inhaler improved adherence to preventer medication over a 6-month period[5]
.
These data, and the potential of smart inhalers, have piqued the interest of pharmaceutical companies. GlaxoSmithKline, Boehringer Ingelheim and Novartis have signed development deals with Propeller Health, while AstraZeneca has signed a master supply agreement with Adherium. Novartis is also working with US mobile technology company Qualcomm to develop the first integrated smart inhaler, which they hope to launch in 2019.
“Activity in this area [developing and testing smart inhalers] is intense,” says Jon-Paul Sherlock, director of AstraZeneca’s Intelligent Pharmaceutical Respiratory division. “Selecting which company to work with was based on a number of factors, including clinical evidence, strategic fit and the quality and range of the devices. Propeller’s [add-on product] would not work with many of our devices, including the Turbuhaler, which a lot of people use. We chose Adherium because we were in a position to evolve with them starting from them having a device that was ready for us.”
AstraZeneca, says Sherlock, is testing Adherium’s add-ons in countries including the Netherlands, and plans to roll out to other markets. He expects development to be rapid, with more advanced inhalers available by 2020.
Source: Adherium/Smartinhaler
New Zealand-based Adherium is one of several companies developing smart inhaler devices and apps.
“It’s a very exciting area and has the potential to be truly disruptive, which is why AstraZeneca is exploring it,” says Sherlock. “There’s a misnomer that drug companies just make medicines and push them out, but the idea here is that we can improve clinical outcomes and clinical benefits.”
It’s a very exciting area and has the potential to be truly disruptive
He adds: “We anticipate that the expectations of patients, healthcare professionals and ultimately payers will change as smart inhalers become more established. This new technology introduces the potential to support patients, ensuring they get the best from the medicine they have been prescribed.” Helping patients get the most from their asthma medicine, he says, is something that drug companies have struggled with.
The need for routine care
Although of undoubted potential, there’s a feeling that if smart inhalers are adopted too quickly, without addressing the shortfalls in the asthma care model, they might only paper over the cracks in the healthcare system that patients are falling though. “In the right situation, smart inhalers can be an immense boon,” says Dermot Ryan, a doctor and respiratory disease researcher at the University of Edinburgh. “But people think machines of one sort or another are going to obviate the need for a patient attending a clinician. That’s not the case.”
People think machines of one sort or another are going to obviate the need for a patient attending a clinician; that’s not the case
Smart inhalers can give a signal that a patient is overusing their preventer medicine, suggesting poorly controlled asthma. An informed and engaged clinician, though, can get the same signal without smart inhalers by looking at how much preventer or reliever medication a patient has used. “Anyone taking more than two blue [rescue] inhalers a year has poorly controlled asthma,” says Ryan. “You don’t need a gizmo to tell you that. You just need to recognise that a patient is overusing and investigate the reasons why.”

Source: Courtesy of Dermot Ryan
Dermot Ryan, a doctor and respiratory disease researcher at the University of Edinburgh, highlights the importance of patient contact with their clinician.
A patient may not be using their preventer medication or not using it properly. Maybe they have a new job with different environmental exposures, a new pet, or have started smoking again. Whatever the circumstance, Ryan says that doctors need to help patients understand the nature of their disease and how important it is that they take their medication. They also need to make sure that a patient uses the correct inhaler technique.
The importance of technique
Future smart inhalers might help by monitoring and correcting a patient’s inhalation technique. Researchers led by Richard Costello at the Royal College of Surgeons in Ireland, based in Dublin, have developed a device that attaches to GlaxoSmithKline’s popular Diskus inhaler. The device uses acoustic sensors to monitor technique and can detect errors in both time and technique of inhalation[6]
.
An EU-funded multi-institutional research project, MyAirCoach, is assessing how mobile health technologies including smart inhalers with new sensors can help patients bring their asthma under control.
MyAirCoach began testing at the end of 2016 in patients at two sites in the UK — the University of Manchester and Imperial College London — and at Leiden University in the Netherlands. The study researchers are developing data analysis and modelling software to help patients and doctors make sense of the data collected.
Omar Usmani, a respiratory medic at Imperial College London, is clinical lead for MyAirCoach. “More often than not you see that a patient is using the wrong inhalation technique with their inhaler, which means in most instances they may as well not be taking it,” he says.
There are two main types of inhalers. Dry-powder devices require a short, sharp inhalation. “The medicine in these devices is attached to a lactose molecule so inspiratory force needs to be sharp and fast to pull it off and get it into the airstream and into the lung,” says Usmani.
By contrast, patients using aerosols, such as pressurised metered-dose inhalers and soft mist inhalers, which release a measured, aerosolised dose of medication, must breathe slowly and deeply over about four seconds. “But many patients inhale too quickly — they see it on TV and in movies — and most of the drug gets stuck on the back of the throat and doesn’t reach the lungs.”
Usmani also stresses the importance of improving primary care and doctor training to allow these smart inhalers to be used properly. “Patients are not properly taught by their healthcare professionals — and most [healthcare professionals] really don’t know anything about how to use a device properly,” he says. He refers to a 2012 study of over 1,500 physicians in Spain who frequently prescribe inhaled medicine, which showed that more than 85% had inadequate knowledge of inhaled therapy[7]
.
Most healthcare professionals really don’t know anything about how to use a device properly
Technique training for both patients and healthcare professionals, he says, would offer huge potential benefits to patient care. It can offer savings to health systems, too. A 2016 study of poor inhaler technique with medicine for asthma and COPD in Spain, Sweden and the UK concluded that the economic impact is around €782m each year[8]
. Similarly, a Goldman Sachs report in 2015 said that the cost of poor inhaler technique to the United States was US$19.3bn[9]
. “There’s certainly a health economic argument for an inhaler that gets the most out of medicine,” says Usmani.
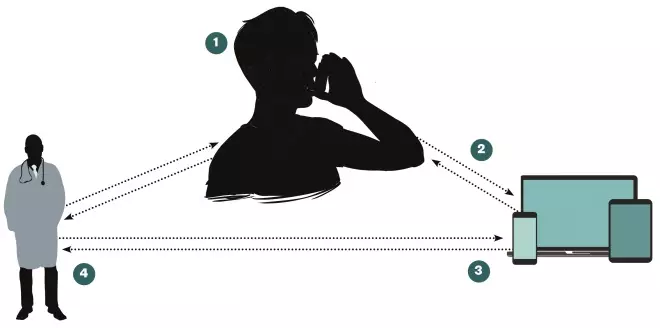
Figure: How smart inhalers work
Smart inhalers use sensors and Bluetooth technology to detect inhaler use, remind patients when to take their medication and gather data to help guide care.
1. Smart inhalers have sensors that record every time a patient uses their inhaler
2. Data are communicated to a mobile app or desktop computer, and are stored in the cloud
3. Patients receive audio and visual reminders about inhaler use via their mobile devices
4. Healthcare professionals can track inhaler use and provide patients with personalised feedback and education
Big data
Much of the big-picture thinking about smart inhalers concerns their potential to provide big data that researchers could analyse to learn more about the disease and look for new interventions. The project at MyAirCoach, for instance, is trying to understand how data can be fed back to patients to allow them to better understand and avoid their triggers.
“We are assessing how a biomonitor device that includes an environmental sensor — so nitric oxide, particulate matter, some environmental sensing parameters — can be useful for patients and give them personalised information about if they might be leading up to an attack,” says Konstantinos Votis, a computer engineer and informatics researcher at the Centre for Research and Technology Hellas in Thessaloniki, Greece, who co-ordinates MyAirCoach.
“It can also be helpful for doctors to see what is happening and we are looking at developing a modelling tool to help researchers understand more about the disease.”
Such big data analysis could open up interesting research questions, says Ryan. “For COPD, a question would be: ‘Can we find a way to predict an exacerbation through drug use and environmental factors and then find a way to intervene to prevent or lessen the exacerbation?’.
“If we could, we would also be able to reduce the severity and duration of each exacerbation and the permanent damage to lung tissue that follows,” he adds.
The data could also be used to personalise treatment. “If a clinician is talking to a patient who is, say, 40 years old, has smoked ten cigarettes a day for ten years, is married with three kids, at an income level of whatever,” says Ryan, “if you can go to the cloud and see maybe 10 to 15 patients with a similar story and see what treatment worked for them, you can explain that to the patient and say ‘Let’s try this’.” He adds: “We’re miles away from that, of course, but being able to do that would really help to get buy-in from the patient around drug adherence.”
The need for co-operation
For this future potential to come to fruition, the data need to be freely available and shared. Free sharing of valuable data, however, is a concept not always associated with commercial organisations such as drug companies.
“Pharma companies won’t do something unless there’s money in it for them,” says Ryan. “If you take a look at the bronchodilators on the market [and] look at the literature in terms of benefits, the difference between them is tiny. So there’s two ways they can compete: on cost and on added value. Added value is where smart inhalers come in.”
With fierce competition in a large market, tiny margins make big bottom-line differences: the data collected from the inhalers might also give a competitive edge.
Sherlock is tight-lipped on ways data can inform drug development, saying that there’s much to learn about how algorithms can guide product development. Possible uses include long-term monitoring of drug effectiveness and improvements in understanding about diseases to better develop interventions that work.
“Companies aren’t going to talk to each other because they’ve got commercial interests at heart,” says Usmani, “but is there a mechanism at a pre-competition stage that they can come together and talk so we can make sure that we get full benefits to patients?”
At the end of 2017, when the one-year data are in from MyAirCoach’s pilot tests, Usmani is planning to hold an open day to discuss the project and to encourage dialogue between industry, engineers, manufacturers, healthcare professionals and patients.
Another concern, says Clift, is how easily patients can switch between different smart inhalers. “We know that people with asthma need to change medicines and inhalers at times, so there’s an interoperability angle that needs to be addressed so smart inhalers don’t place barriers on patients easily switching.”
There’s an interoperability angle that needs to be addressed
Clift says he would like to see technical standards laid out, too, so that each data collection and patient management system will fit in with existing software systems in the NHS. “We also know that these devices have encouraging results in terms of increasing adherence but we haven’t seen conclusive evidence at scale that that has knock-on improvements in clinical outcomes and quality of care.
“Of course we need to decide who owns the data — is it the NHS or the manufacturers?” he adds.
Anna Murphy, a consultant respiratory pharmacist at University Hospitals of Leicester NHS Trust, is similarly enthused, yet cautious about the potential of smart inhalers. Her trust is carrying out both NHS and industry-funded assessments of smart add-ons for both dry-powdered and metered-dose inhalers in patients with severe asthma.

Source: Courtesy of Anna Murphy
Anna Murphy, a consultant respiratory pharmacist at University Hospitals of Leicester NHS Trust, is cautious about the potential of smart inhalers, saying that behaviour change is not easy.
“Yes, the technology can in theory help patients to adhere to their medicines,” she says, “but I’m worried that it’s a bit of a false security, thinking that we’ve solved the problem of adherence with technology and ticking that box but I don’t think we have. The whole of public health history tells us that behaviour change isn’t that easy.”
There’s a long way to go, says Clift, but the potential improvements that smart inhalers could bring means it’s worth the effort. But health systems should not be blinded by the lights and rush into adopting the new technology before all potential problems are ironed out. “Patients, doctors, drug companies, health systems — everyone stands to benefit,” he says, “but to work for one part it needs to work for all of them — the patients first.”
References
[1] Senior coroner, Gateshead & South Tyneside. Regulation 28: report to prevent future deaths. Available at https://www.judiciary.gov.uk/wp-content/uploads/2016/01/Mills-2015-0416.pdf (accessed 30 March 2017).
[2] Royal College of Physicians. National review of asthma deaths. Available at https://www.rcplondon.ac.uk/projects/national-review-asthma-deaths (accessed 10 March 2017).
[3] Asthma UK. Smart inhalers. Available at https://www.asthma.org.uk/get-involved/campaigns/publications/smartasthma/ (accessed 10 March 2017).
[4] Merchant RK, Inamdar R & Quade RC. Effectiveness of population health management using the Propeller Health asthma platform: a randomized clinical trial. J Allergy Clin Immunol 2016;4:455–463. doi: 10.1016/j.jaip.2015.11.022
[5] Chan AH, Stewart AW, Harrison J et al. The effect of an electronic monitoring device with audiovisual reminder function on adherence to inhaled corticosteroids and school attendance in children with asthma: a randomised controlled trial. Lancet Respir Med 2015;3:210–219. doi: 10.1016/S2213-2600(15)00008-9
[6] D’arcy S, MacHale E, Seheult J et al. A method to assess adherence in inhaler use through analysis of acoustic recordings of inhaler events. PLoS ONE 2014;9:e98701. doi: 10.1371/journal.pone.0098701
[7] Plaza V, Sanchis J, Roura P et al. Physicians’ knowledge of inhaler devices and inhalation techniques remains poor in Spain. J Aerosol Med Pulm Drug Deliv 2012;25:16–22. doi: 10.1089/jamp.2011.0895
[8] Lewis A, Torvinen S, Dekhuijzen PN et al. The economic burden of asthma and chronic obstructive pulmonary disease and the impact of poor inhalation technique with commonly prescribed dry powder inhalers in three European countries. BMC Health Serv Res 2016;16:251. doi: 10.1186/s12913-016-1482-7
[9] Roman DH and Conlee KD. The digital revolution comes to US healthcare. Goldman Sachs, 29 June, 2015. Available at: http://www.scbio.org/resources/Documents/Internet%20of%20Things%20-%20Volume%205%20-%20The%20Digital%20Revolution%20comes%20to%20US%20HC%20-%20Jun%2029,%202015%5B1%5D.pdf (accessed 10 March 2017)