This content was published in 2010. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
Summary
The management of chronic myeloid leukaemia has been revolutionised by the discovery of imatinib and other tyrosine kinase inhibitors. However, there is still significant progress to be made in the pharmacological treatment of acute myeloid leukaemia (AML) and the myelodysplastic syndromes, since the prognosis of particular subgroups (eg, elderly patients with AML and patients with MDS who are deemed to be at higher risk) remains poor.
With research ongoing into several novel, targeted therapies, the hope is that the incremental improvements in patient outcomes, which have been demonstrated over the past few decades, will continue to be seen in the future. Concerns remain about whether the significant acquisition costs associated with these novel therapies will prevent their adoption into routine clinical practice.
Treatment options for acute myeloid leukaemia (AML), chronic myeloid leukaemia (CML) and the myelodysplastic syndromes (MDS) have grown over the past decade with several important new therapies having been licensed or currently under development, alongside advances in stem cell transplantation.
Acute myeloid leukaemia
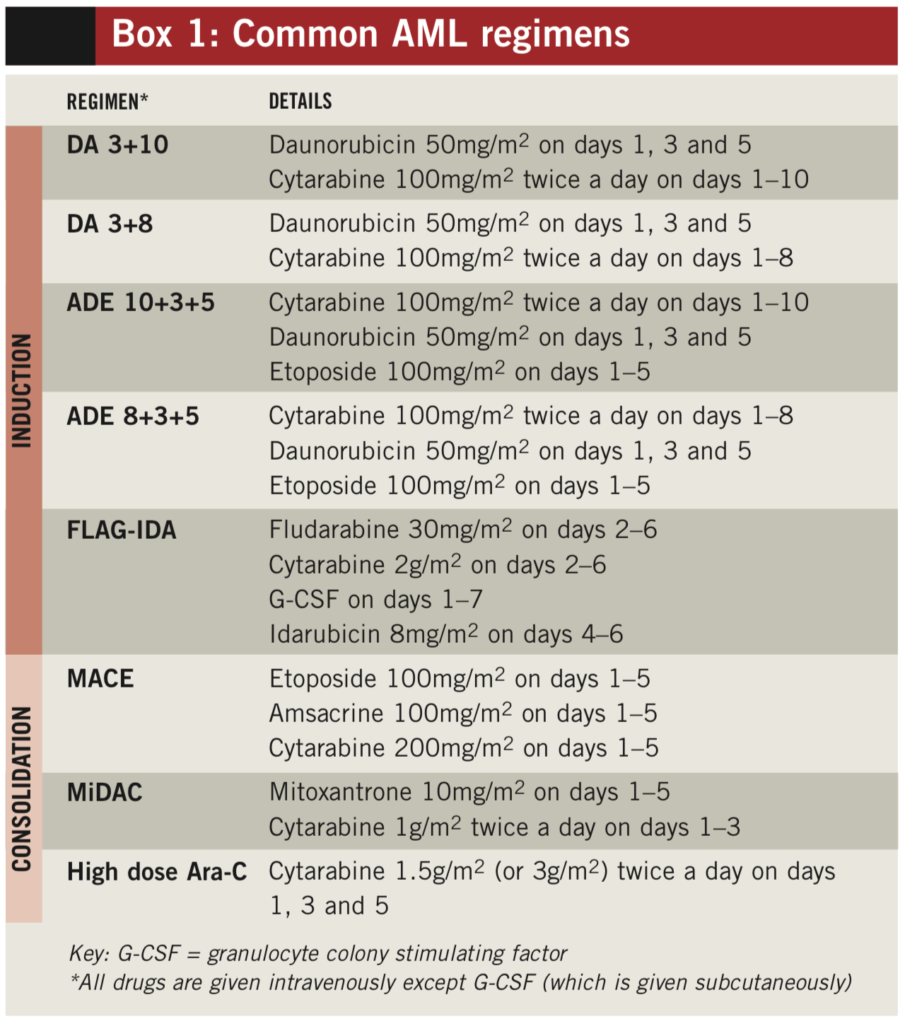
Chemotherapy for AML is divided into two phases: induction and consolidation. The initial aim of induction treatment is to produce a “morphological complete remission” — ie, normal neutrophil and platelet counts and a reduction in the number of leukaemic blast cells to <5% of the total white blood cell count in the bone marrow. Induction regimens include cytarabine and an anthracycline (see Box 1). Between 70% and 90% of patients achieve complete remission after one or two courses of induction treatment.
Recent research suggests that adding the immunoconjugate gemtuzumab ozogamicin (a combination of an anti-CD33 monoclonal antibody and an anthracycline antibiotic known more commonly by its brand name Mylotarg) to regimens containing cytarabine and daunorubicin might increase response rate and lengthen disease-free survival.1 However, this treatment is not licensed in Europe at present (see “Future therapies” below).
Patients designated as “good risk” according to cytogenetic profiling are normally treated with two courses of induction chemotherapy followed by one or two courses of consolidation chemotherapy. However, uncertainty remains as to the most effective consolidation schedule and the optimal number of courses.
Patients designated as “intermediate risk” or “poor risk” according to their cytogenetic profile and who respond to induction chemotherapy can be considered for an allogeneic stem cell transplant (SCT).2 Cemotherapy alone is associated with a significant risk of disease relapse in these patient groups. The treatment of AML differs for certain patient populations.
Elderly
Over the past 30 years of Medical Research Council (now National Cancer Research Institute [NRCI]) trials, the use of intensive chemotherapy has produced significant improvements in outcome for younger patients. However, approximately 70% of patients presenting with AML in the UK are over 60 years of age.6 Improvements in survival have been lacking among these older patients, presumably because:
- The leukaemic cells in this group are intrinsically more resistant to chemotherapy
- Comorbidities and reduced marrow reserve make it difficult to deliver intensive chemotherapy
Currently, although the complete remission rate is about 60% for patients over 60 years of age, their median survival is only 5–10 months.6 There is concern that high intensity chemotherapy might actually reduce the duration of survival for some older patients. Consequently, individualising therapy by using low-dose cytarabine (eg, 20mg twice a day by subcutaneous injection for 10 consecutive days every four weeks) or other low-intensity treatments is regarded as the most appropriate approach to treating less fit patients.
Acute promyelocytic leukaemia
Acute promyelocytic leukaemia (APML) is a subtype of AML with a better prognosis than other subtypes. Treating the condition with a combination of an anthracycline and all-trans retinoic acid (ATRA) (eg, the AIDA schedule) produces cure rates in excess of 80%.7 ATRA’s mechanism of action is novel since it promotes the differentiation (maturation) of malignant cells.
ATRA is given orally at a dose of 45mg/m2 once a day during induction and consolidation. Although generally well tolerated, it is associated with a serious adverse event called differentiation syndrome or ATRA syndrome, characterised by fever, fluid retention, low blood pressure and dyspnoea. Treatment requires ATRA to be withheld and high-dose dexamethasone (10mg intravenously every 12 hours for at least three days) to be commenced promptly.
For the 15–20% of patients who are not cured by first- line treatment, arsenic trioxide is now widely used second line and is associated with high complete remission rates.8 Building on the proven efficacy of arsenic as a salvage therapy, the current AML 17 trial (see below) is investigating whether a chemotherapy-free combination of arsenic trioxide and ATRA is better than the AIDA schedule as first-line therapy for APML.
Future therapies
The AML 16 and AML 17 trials (both currently being conducted by the NCRI), are assessing several novel therapies for AML. The AML 16 trial is investigating the role of clofarabine (a second-generation purine nucleoside analogue), gemtuzumab ozogamicin, arsenic trioxide and azacitidine (see below) in older patients with AML or high-risk MDS. (AML 17 is focusing on younger patients with these conditions.)
In AML 17, patients found to have a FLT3 mutation are randomised to receive the orally administered FLT3 inhibitor lestaurtinib (CEP-710), sequentially with conventional chemotherapy, or placebo. Another arm of the same trial is investigating everolimus, an oral inhibitor of the mammalian target of rapamycin (mTOR) protein kinase, based on evidence that survival of AML stem cells may be regulated by mTOR.
A further arm of the AML 17 trial is investigating whether a higher dose of gemtuzumab ozogamicin confers greater benefit and whether adding etoposide to induction chemotherapy improves patients outcomes. Gemtuzumab ozogamicin is currently approved in the US for the treatment of elderly patients with relapsed AML. However, in 2007, its application for licensing in Europe was rejected by the European Medicines Agency due to a paucity of trial data and toxicity concerns.9 Side effects of this treatment include bone marrow suppression, infusion reactions and veno-occlusive disease of the liver. Because of the risk of hepatic toxicity, patients receiving gemtuzumab ozogamicin should not be prescribed concurrent azole antifungals.
Robert Becker | Custom Medical Stock Photo | SPL
Myelodysplastic syndromes
Supportive care is the cornerstone of treating the myelodysplastic syndromes; this encompasses antibiotic therapy, transfusions of red blood cells and platelets, and stimulation of haematopoiesis using epoetin and granulocyte colony stimulating factor (G-CSF).10 Iron chelation therapy (usually with desferrioxamine) is recommended for patients with long-term transfusion requirements who have already been administered 5g iron (approximately 25 units of red blood cells) or more. Immunosuppression with rabbit antithymocyte immunoglobulin or ciclosporin can be considered in patients with a hypocellular marrow.10
Allogeneic SCT is the only potential cure for MDS; this more intensive approach is considered for patients with intermediate- or high-risk scores according to the international prognostic scoring system. Although SCT is associated with long-term disease-free survival in up to 40% of patients,11 less than 10% of patients are likely to be young enough and have a suitable donor to be considered for a transplant.12
Azacitadine
For patients with MDS who are not eligible for an SCT, one treatment option is azacitidine — a hypomethylating agent. A recent study involving 358 patients compared azacitidine with conventional care (low-dose cytarabine, intensive chemotherapy or best supportive care).13 After a median follow-up of 21.1 months, median survival (the primary endpoint) was 24.5 months for the azacitidine group compared with 15.0 months for the control group. (P=0.0001). The median number of azacitidine cycles received by study patients was nine with 14% of patients requiring dose adjustments due to toxicities. Side effects were primarily haematological but gastrointestinal disturbances and injection site reactions were also common.
Current challenges associated with the use of azacitidine include funding (the cost per cycle is over £4,500) and a lack of extended stability data. Recommended shelf life after reconstitution is only 45 minutes at room temperature and eight hours under refrigerated conditions. Therefore it is difficult to prepare doses in advance for seven consecutive days (as recommended by the manufacturer) unless aseptic manufacturing facilities are operating seven days a week. For this reason, some oncology centres have switched to a “5+2” schedule where treatment is given on Monday to Friday of week 1 and on Monday and Tuesday of week 2.
Currently, azacitidine is being appraised by the National Institute for Health and Clinical Excellence with final guidance due to be published shortly. However, NICE’s preliminary recommendation was negative because the incremental cost-effectiveness ratio for the drug is likely to be in excess of £60,000 per quality-adjusted life year gained.14
Lenalidomide
Another novel therapy that has generated considerable attention for the treatment of MDS is lenalidomide. This thalidomide analogue is only licensed in the UK for multiple myeloma but is approved in the US as a treatment for patients with lower risk MDS and who display the specific chromosomal abnormality del 5q. A large phase II trial demonstrated that lenalidomide produced a significant reduction in transfusion requirements in a population of 148 patients with 5q deletion MDS, as well as benefits at a cytogenetic level (ie, the mutation could no longer be detected in some patients).15 Nevertheless, in 2008, the EMA rejected the manufacturer’s submission for licensing, based on concerns over the trial design and the way it was conducted.16
Other novel treatments under investigation for MDS include farnesyl transferase inhibitors, arsenic trioxide and tyrosine kinase inhibitors.17
Chronic myeloid leukaemia
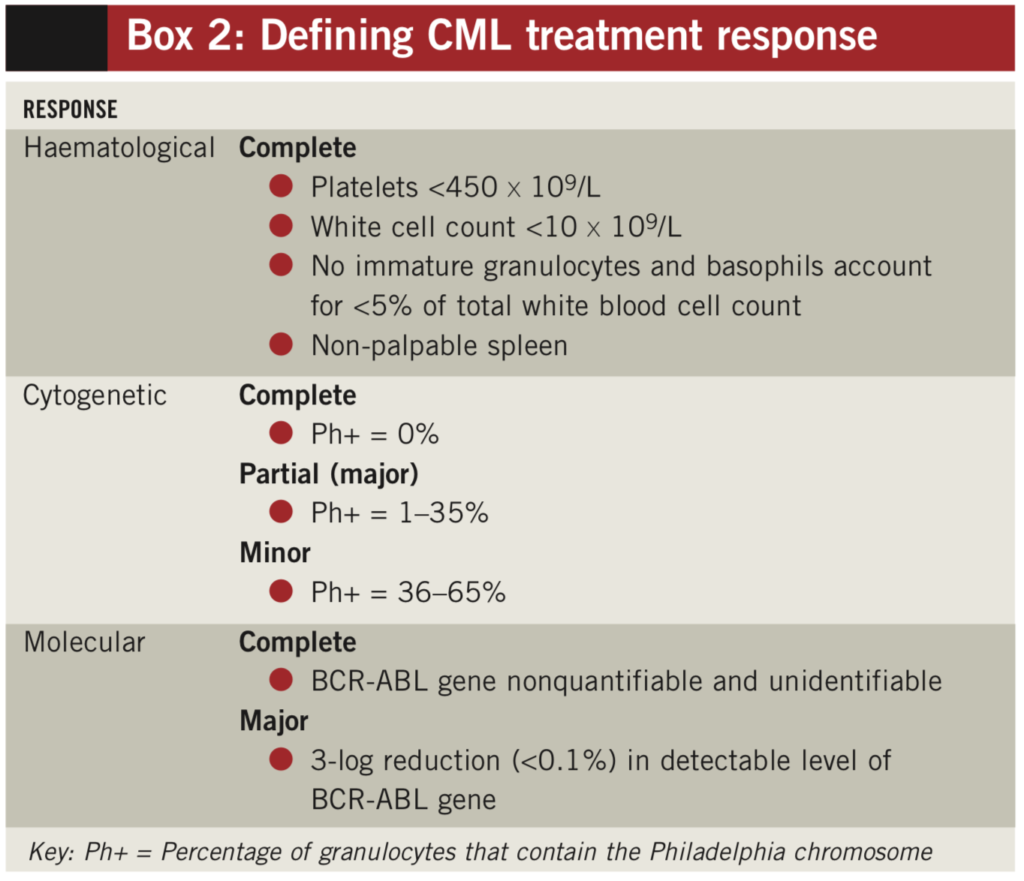
The initial aim of treatment for patients newly diagnosed with CML is to achieve a complete haematological, cytogenetic and molecular response — these are defined in Box 2.18 To achieve these responses, several treatment options exist.
Tyrosine kinase inhibitors
The introduction of imatinib, an oral BCR-ABL tyrosine kinase inhibitor (TKI), has revolutionised the management of CML and the drug is now regarded as first-line treatment.18,19 This is based on the results of the IRIS study, which compared imatinib with the previous gold standard treatment of interferon alfa and cytarabine.20 This study demonstrated marked superiority in favour of imatinib in terms of haematologic, cytogenetic and molecular response rates, tolerability and quality of life. Long-term follow-up data21 have shown an overall survival rate for patients receiving imatinib of 89% after five years — results better than those for any other treatment for the condition. Response to imatinib is monitored closely using cytogenetic and molecular analysis at three- to six-month intervals and specific guidelines exist for defining failure or suboptimal response.18
When a patient’s response to imatinib is suboptimal or deteriorates, dose escalation can be considered. However, resistance to imatinib has been identified as an important clinical problem.21 Consequently, there has been considerable interest in finding novel treatments for this population. This has led to the development of new tyrosine kinase inhibitors — dasatinib and nilotinib — that have greater potency and the ability to retain activity in most imatinib-resistant patients.22 It is possible that dasatinib or nilotinib could be future first-line treatments for CML.
Dasatinib: Dasatinib can generate complete cytogenetic response in about 40% in patients who are resistant to or intolerant of imatinib.23 It has also demonstrated encouraging efficacy in accelerated phase and blast crisis disease.22 However, the drug can cause severe neutropenia and thrombocytopenia in up to 50% of patients. Pleural effusions have also been reported.
Nilotinib: Nilotinib is generally used third line after imatinib and dasatinib. It appears to be similarly effective to dasatinib in chronic phase CML24 but is not licensed to treat blast crisis. Although it appears to be less myelosuppressive than dasatinib, it is more likely to produce biochemical abnormalities such as elevated bilirubin, transaminases and lipases.
A recent phase III trial involving 846 newly diagnosed CML patients demonstrated superior response rates for nilotinib compared with imatinib.25 After one year, 44% of patients receiving nilotinib had achieved a major molecular response compared with 22% of patients receiving imatinib (P<0.0001).
Side effects
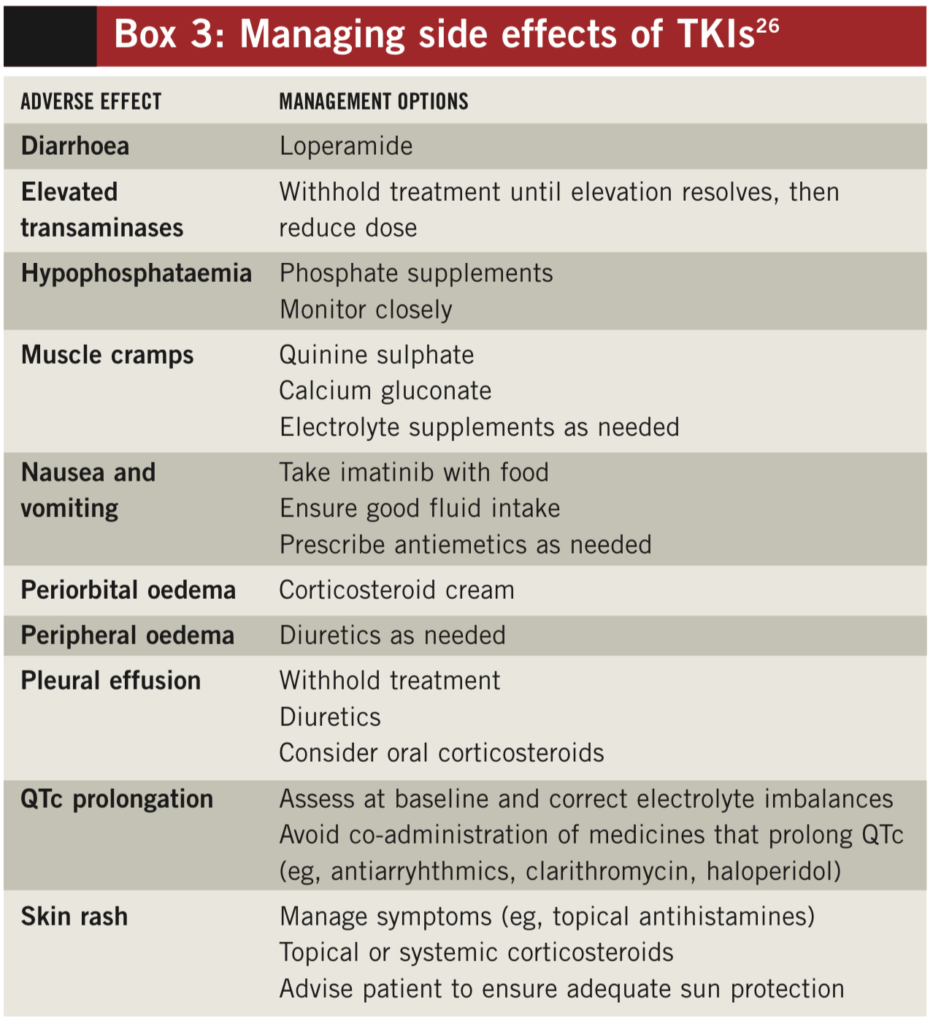
Pharmacists should be aware of the toxicity profiles of TKIs and to be in a position to advise on appropriate management of side effects — see Box 3. For example, nausea with imatinib may be reduced by taking the dose after the main meal of the day or by splitting the daily dose into morning and evening doses. Conversely, toxicities with nilotinib are generally greater if patients take their capsules after food because this increases the drug’s absorption.
All three medicines are metabolised by cytochrome P450 enzymes in the liver, primarily CYP3A4, so a large number of clinically significant drug interactions are possible. Enzyme inducers (eg, rifampicin and phenytoin) should not be given concurrently with imatinib, dasatinib or nilotinib. Potent CYP3A4 inhibitors (eg, itraconazole, clarithromycin, grapefruit juice) should also be avoided due to the risk of increased toxicity.
Other treatments
Although it was previously considered to be the gold standard drug therapy for CML, interferon alfa is no longer recommended as a first-line treatment for the disease. However, it can be considered for patients who are unable to tolerate TKI therapy.18,19
Less common treatment options include hydroxy- carbamide or busulfan. Although these treatments are effective at reducing white cell count, they have no effect on the long-term outcome of CML.
Stem cell transplantation
Allogeneic SCT is an important treatment option for AML, CML and MDS. Historically, only patients who were under 45 years of age were considered for such treatment because of the significant toxicity of the full- intensity conditioning regimens used to prepare patients for transplantation. However, there has recently been a marked increase in the use of reduced intensity conditioning (RIC) regimens. This less intensive approach is associated with lower toxicity and has meant that patients aged 45–65 years can now be considered for a potentially curative transplant.
However, the applicability of SCT is still limited by the morbidity and mortality associated with the procedure and a lack of suitable donors. Ideally, the donor should be a sibling of the recipient with the same human leucocyte antigen type. However, a matched unrelated donor may also be considered.
AML
When treating AML, allogeneic SCT from a sibling donor has shown to be better than chemotherapy alone in patients with intermediate- or poor-risk cytogenetics who have achieved first complete remission.3
Patient age is crucial for determining whether an RIC schedule should be used, with full transplantation generally being reserved for those under 40 years of age. Phase II data for RIC allografts have demonstrated a 50% survival rate in high-risk patients, illustrating the ability of the graft to act against any residual leukaemic cells in this population (this is known as the graft versus leukaemia effect).4
CML
Imatinib is generally preferred over allogeneic SCT as first-line treatment for patients with newly diagnosed, chronic phase CML.18,19 However, SCT is currently the only treatment that has been shown to cure CML. In those patients eligible for this procedure, the rates of long-term molecular remission or cure are about 60%.18
The development of RIC schedules for CML has extended the upper age limit of transplant eligibility; results to date are promising with a three-year overall survival of 70% in “good risk” patients.27
Autologous transplants
Although widely used in the past, autologous SCT — where the stem cells are harvested from the recipient’s own blood or bone marrow — is now rarely used to treat AML. However, it may be considered for patients with APML who have not been cured by chemotherapy alone.5
In a recent meta-analysis, autologous SCT was shown to offer no survival advantage over interferon-based therapy for patients with CML so is not recommended as an initial treatment for this condition either.28
References
- Burnett A, Kell W, Goldstone A, et al. The addition of gemtuzumab ozogamicin to induction chemotherapy for AML improves disease free survival without extra toxicity: preliminary analysis of 1115 patients in the MRC AML15 trial (abstract). Blood 2006;108:8a.
- Fey M, Dreyling M. Acute myeloblastic leukemia in adult patients: ESMO clinical recommendations for diagnosis, treatment and follow-up. Annals of Oncology 2009;20 (s4):100–1.
- Milligan D, Grimwade D, Cullis J, et al. Guidelines on the management of acute myeloid leukaemia in adults. British Journal of Haematology 2006;135:450–74.
- Tallman M, Nabhan C, Feusner J, et al. Acute promyelocytic leukemia: evolving therapeutic strategies. Blood 2002;99:759–67.
- Douer D, Tallman M. Arsenic trioxide: new clinical experience with an old medication in haematologic malignancies. Journal of Clinical Oncology 2005;23:2396–410.
- European Medicines Agency. Questions and answers on recommendation for the refusal of the marketing authorisation for Mylotarg. September 2007. www.ema.europa.eu (accessed 19 March 2010).
- Bowen D, Culligan D, Jowitt S, et al. Guidelines for the diagnosis and treatment of adult myelodysplastic syndromes. British Journal of Haematology 2003;120:187–200.
- Anderson J, Appelbaum F, Fisher L. Allogeneic bone marrow transplantation for 93 patients with myelodysplastic syndrome. Blood 1993;82:677–81.
- Cazzola M, Malcovati L. Myelodysplastic syndromes — coping with ineffective haematopoiesis. New England Journal of Medicine 2005;352:536–7.
- Fenaux P, Mufti G, Hellstrom-Lindberg E, et al. Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: a randomised, open-label, phase III study. Lancet Oncology 2009;10:223–32.
- National Institute for Health and Clinical Excellence. Myelodysplastic syndromes — azacitidine: appraisal consultation document (final appraisal determination published March 2010). www.nice.org.uk (accessed 19 March 2010).
- List A, Dewald G, Bennett J, et al. Lenalidomide in the myelodysplastic syndrome with chromosome 5q deletion. New England Journal of Medicine 2006;355:1456–65.
- European Medicines Agency. Questions and answers on recommendation for the refusal of the marketing authorisation for Lenalidomide Celgene Europe. May 2008. www.ema.europa.eu (accessed 19 March 2010).
- Faderl S, Kantarjian HM. Novel therapies for myelodysplastic syndromes. Cancer 2004;101:226–41.
- Baccarani M, Saglio G, Goldman J, et al. Evolving concepts in the management of chronic myeloid leukemia: recommendations from an expert panel on behalf of the European Leukemia Net. Blood 2006;108:1809–20.
- British Committee for Standards in Haematology. Recommendations for the Management of BCR-ABL-positive chronic myeloid leukaemia. 2007. www.bcshguidelines.com (accessed 19 March 2010).
- O’Brien SG, Guihot F, Larson R, et al. Imatinib compared with interferon and low dose cytarabine for newly diagnosed chronic phase chronic myeloid leukemia. New England Journal of Medicine 2003;348:994–1004.
- Druker BJ, Guilhot F, O’Briem SG, et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukaemia. New England Journal of Medicine 2006;355:2408–17.
- Melo J, Chuah C. Novel agents in CML therapy: tyrosine kinase inhibitors and beyond. Haematology ASH Educational Program 2008;427–35.
- Hochhaus A, Kantarjian HM, Baccarani M, et al. Dasatinib induces notable hematologic and cytogenetic responses in chronic-phase chronic myeloid leukemia after failure of imatinib therapy. Blood 2007;109:2303–9.
- Kantarjian H, Giles F, Hochaus A, et al. Nilotinib in patients with imatinib-resistant or –intolerant chronic myelogenous leukemia in chronic phase (CML-CP): updated phase II results (abstract). Journal of Clinical Oncology 2008;26:374.
- Saglio G, Kim D-W, Issaragrisil S, et al. Nilotinib demonstrates superior efficacy compared with imatinib in patients with newly diagnosed chronic myeloid leukemia in chronic phase: results from the international randomized phase III ENESTnd Trial. Blood (ASH Annual Meeting Abstracts) 2009;114:LBA-1.
- Quintás-Cardama A, Kantarjian HM, Cortes JE. Management of toxicity associated with chronic myeloid leukaemia therapy. October 2009. www.clinicaloptions.com (accessed 19 March 2010).
- Cornelissen J, van Putten W, Verdonck L, et al. Results of a HOVON/SAKK donor versus no-donor analysis of myeloablative HLA- identical sibling stem cell transplantation in first remission acute myeloid leukemia in young and middle-aged adults: benefits for whom? Blood 2007;109:3658–66.
- Tauro S, Craddock C, Peggs K, et al. Allogeneic stem cell transplantation using a reduced-intensity conditioning regimen has the capacity to produce durable remissions and long-term disease-free survival in patients with high-risk acute myeloid leukemia and myelodysplasia. Journal of Clinical Oncology 2005;23:9387–93.
- Crawley C, Szydio R, Lalancette M, et al. Outcomes of reduced-intensity transplantation for chronic myeloid leukemia: an analysis of prognostic factors from the Chronic Leukemia Working Party of the EBMT. Blood 2005;106:2969–76.
- Meloni G, Diverio D, Vignetti M et al. Autologous bone marrow transplantation for acute promyelocytic leukemia in second remission: prognostic relevance of pretransplant minimal residual disease assessment by reverse-transcription polymerase chain reaction of the PML/RAR alpha fusion gene. Blood 1997; 90: 1321-5
- CML Autograft Trials Collaboration. Autologous stem cell transplantation in chronic myeloid leukaemia: a meta-analysis of six randomized trials. Cancer Treatment Reviews 2007;33:39–47.