This content was published in 2003. We do not recommend that you make any clinical decisions based on this information without first ensuring you have checked the latest guidance.
We have an American dentist to thank for anaesthesia. In 1844, Horace Wells (a dentist from Vermont) noticed during a demonstration of a new recreational gas, nitrous oxide, that a member of the audience who had been exposed to the gas felt no pain when he knocked his shin. He suggested it be used for dental extraction, demonstrating its suitability by having his tooth painlessly extracted the next day. This humble and serendipitous beginning was the basis for what would become modern
anaesthesia.
To understand drugs for general anaesthesia, it is first helpful to understand how these drugs are used in theatre. General anaesthesia consists of three stages: induction, maintenance and emergence from anaesthesia (ie,“we put patients to sleep, keep them asleep and wake them up”). The anaesthetic drugs used can be divided into intravenous and inhalational (or volatile) agents. A typical anaesthetic would involve an intravenous induction followed by an inhalational maintenance.
Why an intravenous induction? Intravenous induction is preferred because with older inhalational agents it was faster and smoother with less risk of excitement as the patient passes through the phases of anaesthesia, ie, laryngospasm, coughing, bucking. Even with newer anaesthetics, this reason is still valid. Inhalational induction is children, patients with airway obstruction (where an inhalational anaesthetic can maintain muscular tone in the airway), or people with needle phobia.
Why are inhalational agents used for maintenance? They are used because of tradition (they have been used for over 150 years), cheapness, easy titration of depth of anaesthesia and because expired levels of agent can be monitored in real time. This not only confirms that the patient is receiving the anaesthetic but improves agent dose titration.
Total intravenous anaesthesia (TIVA) Over the past 10-15 years, improved syringe pump technology has been developed (target con- trolled infusion [TCI]). A new intravenous agent, propofol, has also been developed, which can be used for both induction and maintenance. These advances mean that total intravenous anaesthesia (TIVA) is increasing in use. TIVA is commonly used in cardiac and neurosurgical cases and increasingly in other fields of surgery, eg, day case surgery. It has the advantages of avoiding environmental pollution and the unwanted effects of inhalational agents but has a disadvantage in that it is difficult to monitor levels of the agent in the patient, and there is a danger of pump failure and disconnection/displacement of intravenous cannula which could lead to potential awareness.
INHALATIONAL ANAESTHESIA
The quest for the ideal inhalational agent has led to 18 compounds being introduced into clinical practice over the past 150 years[1]. The majority have fallen by the wayside as a consequence of their various side effects. Subsequently, there are only five agents used regularly in the UK: halothane, enflurane, isoflurane, sevoflurane and desflurane. Despite over a century of anaesthetic evolution, the perfect inhalational agent has still to be developed. Properties that would be of benefit are set out in Panel 1.
Halothane Halothane was the first halogenated anaesthetic and was introduced in the 1950s. Its role in anaesthetic practice is in inhalational induction, but its use is declining as newer drugs with better safety profiles have been developed[2]. The primary concerns with halothane are its arrhythmogenic potential and hepatotoxicity.
Enflurane Enflurane can excite the CNS and so carries a risk of post-operative convulsions in predisposed patients. It is generally considered more depressant to respiration and cardiovascular function than its isomer isoflurane.
Isoflurane Isoflurane is a good, general-purpose anaesthetic and is probably the most widely used currently. Metabolism to other potentially toxic substances is minimal. It produces less depression of the cardiovascular system than halothane or enflurane and is fairly potent. However, as a sole agent, it produces tachycardia vasodilation, particularly in younger patients. Its blood solubility is greater than that of halothane or enflurane and so recovery from prolonged anaesthesia can be slower.
Desflurane Desflurane has the lowest blood solubility of the modern agents. This in theory means that it should be the best agent for induction and rapid recovery. Unfortunately, at concentrations of 6 per cent, it is a severe airway irritant resulting in coughing, bucking, apnoea and laryngospasm making it unsuitable for gaseous induction. Its main role is perhaps in long surgical procedures where recovery to consciousness may be more prolonged with other agents or in day case surgery. Desflurane has a low boiling point (23.5C), which means that there are some difficulties in producing a controlled vapour concentration. Some early vapourisers also had accuracy problems. The vapourisers used for desflurane require more frequent servicing than those used for other anaesthetics.
Sevoflurane Sevoflurane is closest to being the ideal agent, as it can be used for induction and for maintenance of anaesthesia. Its pleasant odour (pear drops), low blood solubility and low arrhythmogenic risk mean that it is becoming the agent of choice for gas induction. Recovery times have been shown to be faster than with the other agents and similar to that of desflurane, which means that it is a good agent for day case anaesthesia[3].
Concerns have been raised about sevoflurane and fluoride production, and interaction with carbon dioxide absorbers (present in the anaesthesia apparatus). Sevoflurane under- goes 5 per cent metabolism to produce serum fluoride concentration levels of greater than 50nM/L in 7 per cent of patients. Despite this being thought to be the threshold for nephrotoxicity, clinically significant nephrotoxicity has not been reported even though sevoflurane has been used in more than 45 million patients.
In the presence of soda lime and baralyme (in the carbon dioxide absorber), sevoflurane is degraded into a potentially toxic compound called compound A. Compound A produces dose-dependent tubular damage in rats. However in humans, there have been a number of studies which have shown that sevoflurane has produced no renal renal damage even at low flows.

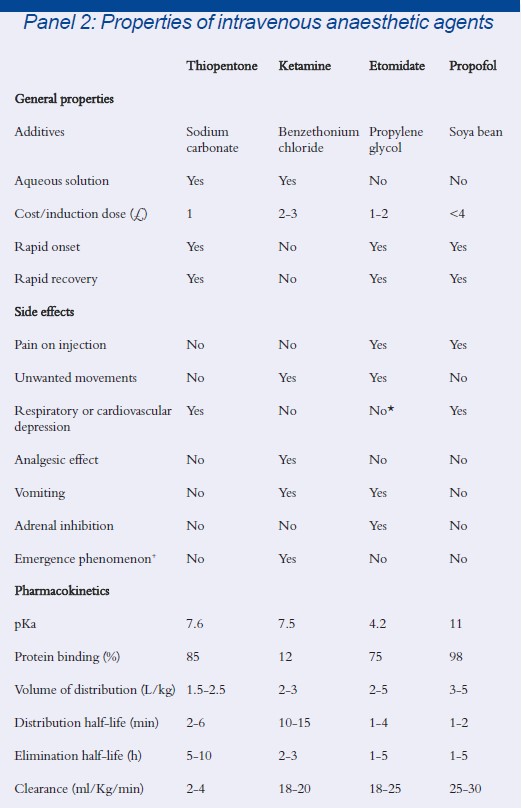
* Etomidate can cause hypotension in hypovolaemic patients
INTRAVENOUS ANAESTHETICS
Intravenous anaesthetic agents were developed later than inhalational anaesthetics. Although one of the first intravenous anaesthetics using choral hydrate was introduced in 1872, it was not until the 1930s that using the intravenous route was popularised. Numerous agents have been introduced and subsequently discarded from clinical practice over the past 70 years, but those that are commonly in use today include etomidate, thiopentone, ketamine and propofol. The properties of these agents are listed in Panel 2.
Of these agents, propofol represents the major advance in intravenous anaesthesia in the past 20 years. It is the closest agent to the ideal that we have, but still falls short in certain areas (see Panel 3 for the properties of an ideal intravenous anaesthetic agent).
Propofol Propofol (2,6-diisopropylphenol) is a hindered phenol, presented as a white oil-in-water emulsion containing 1 per cent or 2 per cent (w/v) of propofol in soybean oil, egg phosphatid and glycerol.‘ The induction dose is 1.5-2.5mg/kg. It is 98 per cent protein bound and undergoes hepatic glucuronide metabolites of 2,6-diisopropylphenol and 2,6-diisopropylquinol that are subsequently excreted in urine.
Propofol is the most widely used intra- venous anaesthetic because it produces a smooth, rapid loss of consciousness with a rapid clear-headed recovery. This rapid recovery is secondary to its short distribution half-life and high clearance. Propofol produces a depression of laryngeal reflexes allowing easier insertion of airway devices. It has a low incidence of post-operative nausea and vomiting as well as a low incidence of hypersensitivity reactions (1 in 80,000 to 1 in 100,000). Propofol does not significantly accumulate after repeat boluses or infusion compared with other agents and so it is suitable not only for induction but also for maintenance of anaesthesia, ie, TIVA.
Deviations of propofol from the profile of the ideal anaesthetic include:
- Pain on injection (up to 28 per cent of patients in one study). This can be reduced by the (unlicensed) addition of lignocaine and by using the large proximal veins
- Respiratory depression on induction
- Dose-related hypotension caused by a reduction in systemic vascular resistance
- The fact that its formulation has been shown to support microbial growth at room temperature. Up to 175 cases of systemic infection can be traced back to injection of contaminated propofol[5]
- The fact that it is not licensed for use in children under three years old
- Occasional excitatory side — effects including myoclonus and opisthonus (hyperextension of the body)
- Unexpected deaths in children who had undergone long-term sedation with propofol on intensive care units, the presenting symptoms being increasing metabolic acidosis, bradycardia myocardial failure
Remifentanil Remifentanil is an ultra-short acting synthetic opioid analgesic drug whose use is increasing in anaesthesia not only as an analgesic but also as an adjunct to induction and maintenance of anaesthesia. It is interesting for not only is it extremely potent, even to the extent that it removes the need for neuro-muscular relaxants to paralyse patients, but no matter how long the drug is infused, the half life will remain the same (about 5 minutes). Therefore, the effects of remifentanil will have worn off in 5 minutes whether the operation took 10 minutes or 10 hours. This is due to its unique metabolism by tissue esterases including cholinesterases in erthrocytes[6].
THE FUTURE
Xenon Xenon may be the inhalational anaesthetic of the future[7]. It is an inert gas which is present in the atmosphere in a concentration of no more than 0.086 parts per million. It has many attractive properties such as the lowest blood/gas solubility of all anaesthetics (0.115) and this results in extremely rapid uptake and excretion. Studies in patients have shown that it does not depress myocardial contractility making it useful in situations where cardiac stability needs to be maintained. It is not metabolised, is not a trigger of malignant hyperthermia and does not cause environmental pollution. But the potency of xenon is low (minimum alveolar concentration of 71 per cent) so when it is used as an anaesthetic, the highest oxygen concentration that can be used with it is 30 per cent.
Xenon is one of the rarest gases in the atmosphere. It cannot be synthesized but is produced by air liquification, so it is extremely expensive which has implications for its widespread use. Solutions to this problem may be recycling the gas, or selectively using it in patients with limited cardiac reserve.
New syringe driver technology Future advances in intravenous anaesthesia are likely to be in the technological rather than the pharmacological area. New syringe driver technology, in the form of target controlled infusions (eg, a microprocessor chip which calculates the dose required to achieve a desired blood concentration and adjusts the syringe pump rate accordingly), combined with depth of consciousness or awareness monitors, have led to preliminary studies looking at closed loop systems. In these systems, the awareness monitor assesses how asleep or awake the patient is and communicates with the syringe driver to give more or less intravenous anaesthetic[8]

CONCLUSION
General anaesthetics have been used for over 150 years. Through incremental improvements they now show some of the properties needed of an ideal agent. The chance of further drug development in general anaesthetia is poor. The available general anaesthetics, even though they do not fit the ideal profile, are safe. For this reason, and because the costs of pharmaceutical research are high, there is little chance of radically better drugs being developed in the future.
The use of regional anaesthesia has increased at the expense of general anaesthesia over the past 20 to 30 years, either alone or as an adjunct to general anaesthesia. However, there will always be a place for general anaesthesia due to the nature of the surgery required (eg, trunk, cardiac or neurological) or due to patient preference.
References
- Hutton P, Cooper GM, James III FM, Butterworth IV JE editors. Fundamental principles and practice of anaesthesia. London:Martin Dunitz; 2002. pp596-619.
- Eger I EI. New inhaled anaesthetics. Anesthesiology 1994; 80:906-22.
- Ghatge S, Lee J, Smith I. Sevoflurane: an ideal agent for adult day-case anaesthesia. Acta Anaesthesiol Scand 2003; 47:917-31.
- Hutton P Cooper GM, James II FM, Butterworth IV JE editors. Fundamental principles and practice of anaesthesia. London:Martin Dunitz; 2002 pp583-95.
- Trepanier CA, Lessard MR. Propofol and the risk of transmission of infection. Can J Anaesth 2003; 50:533-7.
- Duthie DJR.Remifentanil and tramadol. Br J Anaesth 1998; 81:51-7.
- Lynch III C, Baum J, Tenbrink R. Xenon anaesthesia. Anesthesiology 2000; 92:865-70.
- Mortier EP Struys MMRE Monitoring the depth of anaesthesia using bispectral analysis and closed loop controlled administration of propofol. Best Pract Res Clin Anaesthesiol 2001;15:83-96.


