This content was published in 2005. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
Arhythmia is a term which refers to any change in the normal rate or rhythm of the heart. Arrhythmias occur secondary to alterations in the generation or conduction of electrical activity within the myocardium. In order to understand them fully it is necessary first to know a little about how the heart’s normal rate and rhythm are maintained.
Normal activity of the heart
Four structures are key to the conduction of electrical impulses through the heart muscle:
- The sinoatrial (SA) node located in the posterior wall of the right atrium near the superior vena cava
- The atrioventricular (AV) node located between the right atria and ventricles
- The bundle of His (or AV bundle) and bundle branches, which carry current from the AV node to the base of the ventricles
- The Purkinje fibres, which conduct the current throughout the ventricular tissue.
In the healthy heart the SA node acts as the cardiac pacemaker, generating electrical impulses which are then conducted via the atria to the ventricles, hence the term “sinus rhythm”.
Electrical activity
Myocardial contraction is governed by the movement of electrolytes, particularly sodium, potassium and calcium, across cell membranes. At rest, there exists an uneven distribution of ions with a higher concentration of potassium ions inside the myocardial cells and a higher concentration of sodium and calcium ions outside the cells. Within the specialised pacemaker cells of the SA node, this creates a negative electrical charge of about -60mV across the cell membrane (known as the membrane potential). Changes in membrane permeability allow a slow movement of sodium ions into the cell until, at about -40mV, the “threshold potential” is reached. At this point, voltage-sensitive calcium channels open to allow an influx of positively charged calcium ions leading to full depolarisation. A wave of electrical activity sweeps across the myocardium as the current is conducted from cell to cell.
Impulses arising in the pacemaker cells of the SA node are conducted across both atria initially, then into the AV node situated between the right atria and the ventricles. After a short conduction delay across the AV node, the current passes down the bundle of His and bundle branches, and then across the ventricles via the Purkinje fibres. Myocardial contraction occurs at the point of depolarisation with the atria contracting slightly before the ventricles (due to AV nodal conduction delay) to ensure optimal ventricular filling.
Control of heart rate
The normal resting heart rate is approximately 70 beats per minute and is maintained by electrical impulses arising from within the SA node.
The rate of firing of the SA node can be altered by changes in the autonomic nervous system. Increased sympathetic activity (ie, during exercise) allows an influx of calcium ions leading to rapid depolarisation as the threshold potential is reached more quickly and hence the heart rate rises. Parasympathetic stimulation via the vagal nerve increases the outflow of potassium ions, decreasing the membrane potential. This increases the time to reach threshold potential and therefore the heart rate falls.
If the SA node should fail, additional sec- ondary pacemaker mechanisms are present to maintain cardiac output. Firstly, the AV can generate electrical impulses to keep the heart rate at approximately 40-60 beats per minute. This is known as a junctional escape rhythm. Even if this fails, a ventricular escape rhythm will maintain a heart rate of approximately 30 beats per minute. These escape rhythms are usually overridden by the SA node which fires at a much higher rate and suppresses spontaneous impulse generation in these other sites.
Electrocardiogram
In addition to under- standing the conduction pathways of the heart, it is also important to know a little about the electrocardiogram (ECG). The ECG maps the electrical activity occurring within the heart and is the most useful investigation in determining the presence and nature of arrhythmias (see Figure 1 and Figure 2). A standard ECG has 12 leads which are attached to the patient and labelled I-III,V1-V6, aVR, aVL and aVF.
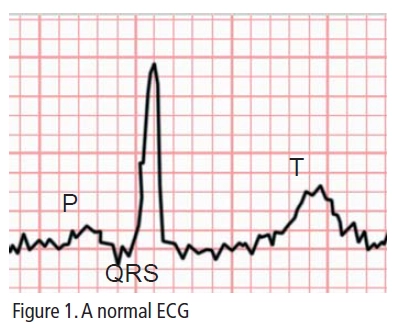
The P wave equates to the passage of electrical activity across the atria, the QRS complex to ventricular depolarisation and the T wave to repolarisation of the ventricles during diastole. The distance between the beginning of the P wave and the beginning of the QRS (the PR interval) corresponds to the slowing of conduction as the electrical impulse passes through the AV node. The QT interval gives an indication of the over- all duration of the action potential.
Pathogenesis of arrhythmias
Arrhythmias develop by one of two mechanisms:
- Altered impulse generation, for example, changes in the automaticity (ability to generate electrical impulses spontaneously leading to depolarisation) of the pacemaker cells in the SA node, or through the occurrence of action potentials from sites other than the SA node
- Altered impulse conduction, for example, complete or partial block of conduction pathways within the myocardium.
The implications of these changes depend on their haemodynamic consequences and the associated risk of degener- ation into potentially life-threatening arrhythmias.
Bradycardias
A heart rate of less than 60 beats per minute is considered a bradycardia. If the heart rate slows but the rhythm remains unchanged (ie, is still controlled by impulses generated in the SA node) this is known as sinus bradycardia. It can be entirely normal, for example in athletes or during sleep, but it may also occur secondary to acute myocardial infarction, sick sinus syndrome or drug therapy, particularly beta-adrenoceptor blockers. Sick sinus syndrome generally presents as syncope (ie, fainting), dizziness and palpitations in the elderly, although it can occur at any age. The underlying cause is usually fibrosis of the SA node. This can result in:
- Sinus bradycardia due to a reduction in the frequency of impulse generation within the nodal pacemaker cells (reduced automaticity)
- Sinus arrest ifthe node completely fails to generate an action potential, resulting in a complete absence of P waves on ECG with an AV escape rhythm
- Sinoatrial block where there is intermittent failure to generate an action potential resulting in intermittently dropped P waves on ECG.
Bradycardias can also be of neurocardiogenic origin, for example, they can be due to excessive vagal tone or can arise during carotid sinus massage. Carotid sinus massage involves the rubbing of the large part of the arterial wall at the point where the common carotid artery, located in the neck, divides into its two main branches. This stimulates an area in the artery wall that contains nerve endings which respond to changes in blood pressure and are capable of slowing the heart rate.
Heart block
Bradycardias can be caused by heart block, where there is a failure to conduct the electrical signal. AV nodal block is the most commonly identified in clinical practice. In this situation, the electrical impulse generated by the SA node travels across the atria but is blocked at the level of the AV node before it can be conducted to the ventricles. AV nodal block can be described as:
- First degree AV block, where AV nodal conduction is slow, resulting in a prolonged PR interval on ECG
- Second degree AV block, where only some of the atrial beats are conducted to the ventricles, resulting in the intermittent appearance of P waves on the ECG which are not followed by a QRS complex
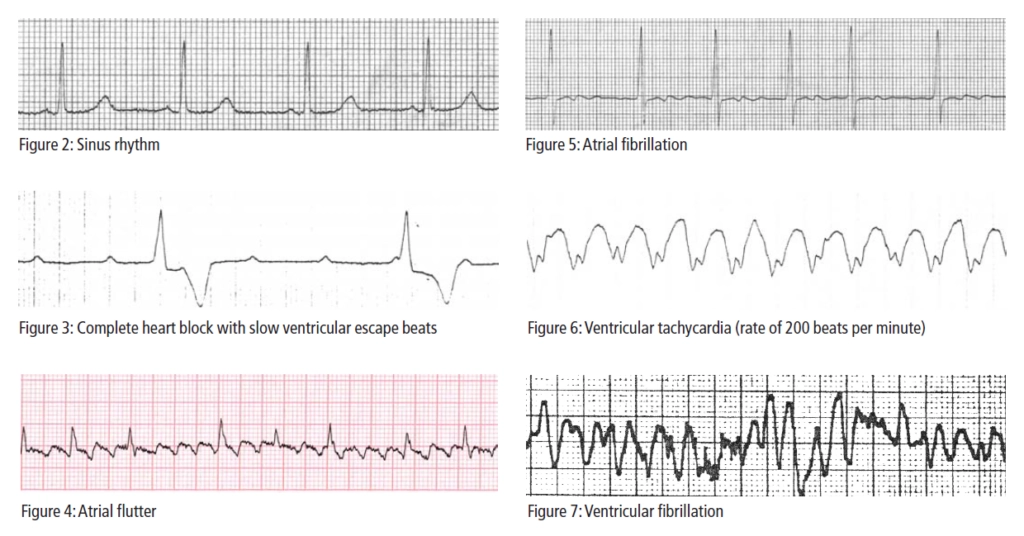
- Third degree or complete heart block, where there is complete failure of AV nodal conduction, and hence dissociation of atrial and ventricular contraction. In this case, the ECG shows no association between P wave activity and ventricular QRS complexes (see Figure 3).
Tachycardias
A heart rate of more than 100 beats per minute is considered a tachycardia. They can be divided into:
- Supra-ventricular tachycardias, which are arrhythmias that arise above the level of the ventricles either within the atria (eg, sinus tachycardia, atrial fibrillation [AF], atrial flutter and atrial tachycardia), or in the AV junction (eg, AV junctional tachycardia or Wolff-Parkinson White syndrome)
- Ventricular tachycardias, which arise within the ventricles themselves.
Sinus tachycardia
Sinus tachycardia occurs if the heart rate increases but the rhythm remains unchanged. Sinus tachycardia is common during exercise or excitation but may also occur during infection, hypovolaemia, anaemia, thyrotoxicosis, pulmonary embolus and shock. It can also occur as a side effect of many drugs, such as nicotine, beta 2 agonists, thyroxine and aminophylline.
Supra-ventricular re-entry tachycardias
A number of re-entry tachycardias (see Panel 1) can occur in, above or within the AV junction.
Panel 1: What is a re-entry circuit?
During normal depolarisation, electrical impulses are conducted from cell to cell in an organised fashion across the myocardium. After excitation, each cell has a short refractory period during which it can cannot be re-excited to conduct any further electrical impulses. This ensures conduction of electricity in the right direction — from atria to ventricles.
If a section of tissues fails to be excited during the initial wave of excitation (because of an anatomical or functional defect), it is possible for this to be activated retrogradely, allowing conduction of an impulse to areas of the heart which have already recovered from the refractory period and are thus ready for re-excitation. In this way, a re-entry circuit is formed, allowing electricity to cycle between or around different areas of the myocardial tissues. Examples of arrhythmias occurring due to re-entry circuits are SA nodal re-entry tachycardia, atrial flutter, AV nodal re-entry tachycardia, Wolff-Parkinson White Syndrome and ventricular re-entry tachycardia.
Sinus node re-entry tachycardia
Sinus node re-entry tachycardia accounts for between 5 and 10 per cent of all atrial tachycardias. In comparison to other atrial tachycardias, it usually results in a relatively slow rate of approximately 120–140 beats per minute, although on occasion can reach 200 beats per minute. The underlying re-entry circuit may exist entirely within the SA node or may involve some atrial tissue.
Atrial flutter
Atrial flutter occurs less frequently than AF, but the two often have similar underlying causes (see AF section). Re-entry is the most common mechanism of atrial flutter. In most cases, a re-entry circuit occurs within the confines of the right atrium, with electrical impulses circulating anti-clockwise from the SA node, down the outer edge of the right atrium and back up via the inter-atrial septum, although clockwise conduction does occur more rarely. Conduction along the re-entry circuit results in a rapid but regular atrial rate of between 250 and 350 beats per minute (often around 300 beats per minute). The resulting ventricular rate is usually regular, but slower than the atrial rate as the AV nodal delay prevents these rapid atrial impulses from being conducted to the ventricles in a 1:1 ratio. Conduction to the ventricles may be 2:1 (one ventricular depolarisation for every two atrial impulses), 3:1 or 4:1, resulting in ventricular rates of approximately 150, 100 and 75 beats per minute, respectively.
The typical atrial flutter ECG shows a regular saw-tooth flutter wave pattern, particularly in leads II and V1, P waves are absent (see Figure 4). Carotid sinus massage may be required to slow the ventricular rate to reveal the underlying flutter waves.
The rapid atrial rate and disturbance of conduction pathways in atrial flutter increases the risk of localised thrombus formation and secondary embolic events (ie, thrombotic stroke) in this group of patients.
AV nodal re-entry tachycardias (AVNRT) and AV re-entry tachycardias (AVRT)
AVNRT and AVRTs are common and frequently present as palpitations which patients find distressing. Many patients suffer recurrent episodes precipitated by exercise or other normal daily activities. Episodes vary from patient to patient in both frequency (from multiple episodes in one day to infrequent occurrence over many months) and duration (minutes to several hours).
In AVNRT and AVRT, a re-entry pathway exists between the atria and the ventricles. This allows electrical impulse to pass between atria and ventricles independently of normal AV nodal conduction. The accessory pathway itself may exist entirely separately from the AV nodal tissue (in AVRT) or as part of the AV nodal tissue itself (as in AVNRT). In most cases, electricity is conducted normally to the ventricle via the AV node and retrogradely, from the ventricle via the accessory pathway back to the atria. In this way a continuous cycling of electricity between atria and ventricles results in fast atrial and ventricular rates. In everyday life the implications of these accessory pathways may not be apparent, as the refractory period inherent in the myocardial conduction tissues prevents unexpected depolarisation and retrograde conduction. However, tachycardias (for example, following exercise or caffeine) or premature beats (ectopics) may be sufficient to initiate and sustain these tachycardias.
In most cases, these tachycardias are regular and characterised by a narrow QRS complex on ECG with rates of up to 250 beats per minute. In many cases the P wave (indicating electrical activity within the atria) may be concealed within the QRS complex or may occur slightly after the QRS complex. Often it is inverted as conduction to the atria is retrograde via the accessory pathway.
Wolff-Parkinson White syndrome (WPW) is an example of a pre-excitation syndrome which is mediated by an accessory pathway. In this case, the accessory pathway conducts the electrical impulse more rapidly than the AV node activating the ventricles (hence the term pre-excitation). The ECG shows a characteristic short PR and a delta wave (slurred up-stroke of the QRS) due to early ventricular activation. WPW patients are at high-risk of developing life-threatening AF as atrial activity can be conducted directly to the ventricles via the accessory pathway resulting in rates of up to 600 beats per minute. AVRTs are also common in this group of patients.
Atrial fibrillation
As one of the most common arrhythmias seen in both general practice and in acute care, AF is a major cause of morbidity and mortality. Overall prevalence in those over 65 years of age is approximately 4 per cent, and this group account for the vast majority of cases of AF. AF incidence increases with age and is more common in patients with hypertension, coronary artery disease and heart failure. Other causative factors include sick sinus syndrome, diabetes, hyperthyroidism, cardiothoracic surgery, high alcohol consumption and chronic obstructive pulmonary disease.
The severity of symptoms related to AF depends on a number of factors including the ventricular rate, presence or absence of concurrent heart disease, ventricular systolic function and in some cases the precipitating cause. In some cases AF can be asymptomatic, although many patients will present with breathlessness, lightheadedness or fatigue. Patients may complain of palpitations, describing their heart as racing, pounding or thumping in their chest. In more severe cases, patients may present with heart failure, angina and syncope or the first presentation may be with a related thromboembolic event. Symptoms are usually exacerbated in those with co-morbidities. This is particularly true for those with concurrent heart failure, where cardiac output is already compromised or significant ischaemic disease, where exacerbation of angina is common.
AF may be acute or chronic. Some patients “flip” into and out of AF at intervals of a few minutes to many days or even weeks.
At an electrical level, AF is caused by disordered conduction within the atria. It is often triggered by an atrial extra-systole (early atrial contraction) but may result from local re-entry circuits within the atria (micro re-entry) or focal trigger sites remote from the SA node generating inappropriate action potentials.
The inappropriate electrical signals lead to repetitive and chaotic depolarisation of the atrial myocardium (see Figure 5). The atria fail to contract in a co-ordinated fashion (ie, fibrillate) and atrial rates of between 350 and 600 beats per minute result. The disordered depolarisation pattern is conducted to the AV node where the intrinsic AV nodal delay prevents this rapid atrial rate from being transmitted directly to the ventricles. However, as soon as the refractory period in the AV node has passed, another atrial impulse can be conducted. As a result the ventricular rate is usually rapid (around 100–180 beats per minute) and irregular, depending on when atrial impulses are conducted. The ECG characteristics of AF are the absence of P waves (as atrial depolarisation is entirely chaotic), presence of f (fibrillation) waves (usually visible in lead II or V1, but may be present in any lead) and irregular ventricular activity.
Failure of co-ordinated atrial contraction results in stasis of blood within the atria. This can lead to the formation of local thrombi. As a result, one of the most important complications of AF is the increased risk of thromboembolic stroke by up to five-fold. In addition to treating the arrhythmia, assessing risk of thromboembolic complications and ensuring adequate thromboprophylaxis is therefore essential. In terms of reducing morbidity this may be more important than controlling heart rate or rhythm.
Ventricular tachycardias (VT)
Most people experience occasional palpitations caused by extra ventricular beats, known as ectopics. Frequent ectopic beats or runs of ventricular ectopics can be a cause for concern as they are associated with increased mortality in those with pre-existing cardiac disease, such as ischaemic heart disease or cardiomyopathy. VT is defined as five or more consecutive ventricular ectopics. This is considered sustained VT if the duration is greater than 30 seconds or non-sustained if the VT spontaneously reverts to sinus rhythm within 30 seconds. Ventricular rates during episodes of VT vary from 120–250 beats per minute.
Patients may present with a wide spectrum of symptoms from palpitations, generally associated with slower ventricular rates, to cardiac arrest, syncope or severe haemodynamic compromise in patients with more rapid ventricular rates. These more serious symptoms are associated with a poor prognosis, particularly in patients with pre-existing ischaemic heart disease or cardiomyopathy.
VTs can be monomorphic or polymorphic. In monomorphic VT the ECG shows a succession of rapid ventricular ectopic beats with the same configuration (hence the term monomorphic VT). The shape of the VT complex will be abnormal and prolonged compared with normal sinus rhythm (see Figure 6). There may be evidence of atrial activity, becasue the SA node will continue to fire. P waves will be dissociated from the ventricular rhythm and will appear at regular intervals throughout the VT, but may be concealed within the VT complex. Evidence of atrial activity on the ECG can be useful in distinguishing a VT from a sustained VT, because these arrhythmias can otherwise look similar. The most common underlying causative factors in monomophic VT are acute myocardial infarction, ischaemic heart disease, cardiomyopathies, myocarditis and valvular disease.
In contrast to the uniformity of ventricular complexes in monomorphic VT, polymorphic VT is characterised by repeated and progressive changes in the complexes, resulting in the classic twisting around the baseline. Polymorphic VT may occur secondary to acute ischaemic damage. An example of polymorphic VT is torsades de pointes (twisting of the points) which can occur secondary to drug therapy, particularly anti-arrhythmics, as a result of QT prolongation (see Panel 2). Care should be taken to exclude QT prolongation during sinus rhythm before prescribing anti-arrhythmic therapies. Torsades de pointes can rapidly degenerate into ventricular fibrillation and must therefore be treated as a medical emergency.
Panel 2: Causes of torsades de pointes
- Bradycardia secondary to sick sinus syndrome
- Congenital conditions leading to prolongation of the QT interval
- Electrolyte disturbances such as hypokalemia or hypomagnesaemia
- Anti-arrhythmic drug therapies
- Other drug therapy such as erythromycin, clarithromycin, anti-depressants (particularly tricyclics), domperidone, droperidol, thioridazine and cisapride
There are two underlying mechanisms to VT, the most common being the presence of a re-entry circuit. This requires the presence of two areas of ventricular tissue which conduct electrical activity at different rates. This allows electrical impulses to pass down one pathway and retrogradely via the other pathway, creating a circuit. Cycling of electricity around this circuit allows rapid activation of the surrounding myocardial tissue and resultant VT. In this case, VT is generally precipitated by a premature ventricular beat. Less frequently, an area or areas of the ventricular tissues may, following damage, acquire enhanced automaticity resulting in the spontaneous generation of electrical impulses at a rate faster than that of the SA node, thereby taking over control of heart rate and rhythm.
Ventricular fibrillation
Ventricular fibrillation (VF) is a rapid and unco-ordinated contraction of the ventricular tissue. It severely compromises cardiac output to the extent that patients usually lose consciousness within 10–20 seconds of onset. It is responsible for most deaths caused by myocardial infarction and there is high risk of VF in patients with severe ischaemic heart disease, cardiomyopathy or myocarditis. VF is a medical emergency, because without prompt treatment irreversible cerebral and myocardial damage will occur. In VF, the ECG shows irregular, chaotic electrical activity (see Figure 7).
Conclusion
This article has outlined the arrhythmias that pharmacists are likely to encounter in clinical practice. Drug therapies form some of the treatment options available and these in addition to other non-drug interventions are reviewed in the second article.

