
This content was published in 2006. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
Our skeletons provide us with a strong yet light protective framework, due to a unique structure comparable to reinforced concrete. Bone is not merely an inert material, however, but a highly dynamic metabolic reservoir of calcium ions. The skeleton stores 99 per cent of all calcium ions contained in the body.1 There are two main types of bone in the skeleton. These are distinguished by their microscopic architecture:
- Cortical bone is hard and dense, and comprises the shafts of the long bones which bear most of the body’s weight.
- Cancellous or trabecular bone is found in the ends of long bones, vertebrae and pelvis. This has a honeycomb like appearance, with plates known as trabeculae arranged in such a way as to provide resistance to forces. There is also a greater internal surface area and blood supply than in cortical bone, which means it is involved in calcium homeostasis.
Bone consists of an organic phase (matrix) and an inorganic mineral phase, together with a highly varied population of cells responsible for its development and maintenance. The matrix is composed of structural proteins, collagen and mucopolysaccharides, which provide resistance and flexibility. The main mineral present is hydroxyapatite (crystalline calcium phosphate) which is responsible for the rigidity and compressibility of bone.
Bone growth (modelling) begins in fetal development and continues to between 25–40 years of age when peak bone mass is said to be attained.2 During puberty there is an accelerated period of growth where most of this mass is laid down. The timing and nature of this process is initially dictated by genetic factors, followed by environmental factors in adulthood, which are described below.
After modelling there follows a process of consolidation, known as remodelling, which is necessary to allow the skeleton to respond to external forces and maintain body mineral levels. Up to 10 per cent of existing bone per year3 is restructured, at sites known as bone multicellular units.
Bone multicellular units consist of teams of osteoblasts and osteoclasts acting in a coordinated fashion under the influence of numerous cellular factors. The resorption process follows a clear series of stages lasting approximately 100 days:2
- Osteoclasts initiate remodelling through specific activation of their membrane receptors. They attach to internal bone surfaces and secrete protons and enzymes which degrade and release minerals from the tissue.
- Osteoblasts then replace the resorbed material with immature matrix protein (osteoid), which later becomes mineralised.
Examples of factors necessary for co-ordinating osteoblasts and osteoclasts include:
- Calcium availability
- Mechanical forces experienced by the skeleton
- Endocrine, autocrine and paracrine factors
The last category is of particular significance once peak bone mass is attained.
Endocrine factors
Endocrine factors are released into the systemic circulation and have a generalised effect on the skeleton.
Parathyroid hormone is a peptide hormone secreted in response to low concentrations of serum calcium and directly enhances osteoclast activation via interaction with a membrane receptor. This stimulates resorption and release of free calcium ions to restore the imbalance.
Vitamin D is obtained from the diet or synthesised in the skin by ultraviolet light. It acts by increasing intestinal calcium absorption and promoting osteoclast resorption.
Calcitonin is a peptide hormone that inhibits the action of osteoclasts in response to elevated serum calcium levels.
Growth hormone promotes the synthesis of collagen and other bone proteins by osteoblasts.
Oestrogens, such as oestradiol, are thought to be important in maintaining bone mass in women, and are thought to act by inhibiting paracrine factors that promote bone resorption (see below). Androgens such as testosterone have a similar role to oestrogens in men, and also contribute to the sudden growth spurt at puberty.1
Autocrine and paracrine factors
Autocrine and paracrine factors are secreted by cells present within bone tissue and have more localised targets of action.
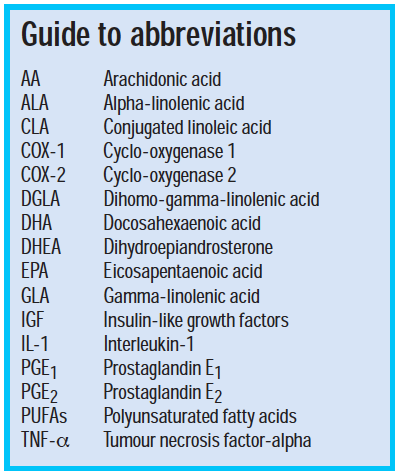
Prostaglandins are derived from membrane lipid precursors and are thought to be the main local mediators of bone metabolism. Prostaglandins E1 and E2 (PGE1 and PGE2) both act via binding to various membrane receptors, and their effects are also mediated through cytokines known as insulin-like growth factors (IGF). PGE1 is made through the action of the enzyme cyclo-oxygenase 1 (COX-1) in many tissues and is thought to increase bone formation, inhibit osteoclasts and promote calcium uptake.
PGE2 is synthesised by the COX-2 isoenzyme in response to factors released during inflammation, and can inhibit bone formation and promote osteoclast activity. This may be through promoting expression of IGF.
Cytokines and growth factors are peptides secreted by osteoblasts and other cells, and have both inhibitory and stimulatory effects on bone remodelling. Examples of cytokines include interleukin-1 (IL-1) and tumour necrosis factor-alpha (TNF-α).4
The effects of both local and systemic molecules are ultimately mediated by transcription factors. Osteoblasts are thought to secrete an osteoclast-differentiation factor initially following stimulation by vitamin D or parathyroid hormone. This factor (known as “receptor activator of nuclear factor-kappa” or the RANK ligand) is a transmembrane protein which specifically binds to a RANK receptor on the immature osteoclast membrane and promotes its maturation. Opposing this action is a second protein known as osteoprotegerin, also secreted by the osteoblast, which inhibits the differentiation of osteoclasts through binding and blocking of the RANK ligand.
In addition to the RANK and osteoprotegerin system, another potential transcription factor mechanism has been identified, known as core binding factor α-1, which promotes expression of genes in osteoblasts necessary for their development from marrow precursor cells. This factor is thought to be more active during fetal and childhood development.5
Bone multicellular units are thus tightly regulated to prevent overall changes in bone mass, but this can be lost with ageing or disease. Until peak mass formation, 100 per cent of the bone multicellular units are active. Following this is a gradual decline in bone strength and increasing porosity due to reduced activity at each bone multicellular unit — a reduction, with age, by 30 per cent in men and 50 per cent in women.3 This can lead to the most prevalent disease of bone known as osteoporosis.
Osteoporosis
Osteoporosis literally means “porous bones” and is defined as a progressive, systemic skeletal disease that leads to loss of bone and its microscopic structure.1 The consequences of this are reduced strength and an increased risk of low-impact fracture. This can lead to associated complications of mortality, morbidity and reduced quality of life. The three most common sites of fracture are due to their having the greatest proportion of cancellous bone, namely the hip (femoral neck), lumbar vertebrae and the forearm or wrist. Hip fractures are clinically the most serious and require long stays in hospital.3 Osteoporosis is a clinically silent condition. Its prevalence increases with age (seen in up to 70 per cent of people over 80 years of age3) and it is often not diagnosed until a fracture occurs.
With increasing life expectancy in the West, osteoporosis looks set to become increasingly prevalent. The World Health Organization has estimated that over 75 million osteoporotic fractures will occur each year by 2025 in Europe, Japan and the US.3 The disease therefore represents a great financial burden on health care systems in terms of secondary care for fractures and long-term primary and social care, with current estimated costs to the NHS being £5m per day.6
Aetiology
There are a number of risk factors which include:
- Advancing age — a decline in osteoblast activity and reduced calcium absorption and retention.
- Sex — women have a lower peak bone mass than males and live longer, and so have a longer period of bone loss. There are also effects of the menopause described below. This means that primary osteoporosis is three times more common in women3, but as life expectancy increases the condition is becoming more common in elderly males.
- The menopause — this occurs in women aged between 45 and 55 years and there is a period of accelerated bone loss (2–3 per cent per year) immediately after the menopause before returning to the same rate as males (0.25–1 per cent per year).2 This is due to a decline in oestrogen levels favouring bone resorption over formation. There can be a difference in bone mass of up to 5 per cent between men and women over the age of 60.1
- Diet — those low in calcium, vitamin D or high in animal protein can increase the risk of osteoporosis. Although protein is necessary in the diet, a large intake of animal protein containing larger quantities of acidic or sulphurous amino acids interferes with renal calcium reabsorption.
- Smoking — this is a significant risk factor as large numbers of children and teenagers continue to smoke. Toxic free radicals in cigarette smoke interfere with blood supply to bone and directly damage osteoblasts. In women, smoking can reduce active oestrogen levels and promote an earlier onset of menopause.
- Excessive alcohol — this is becoming a greater problem as binge drinking increases, particularly in young women. Alcohol has both direct and indirect toxic effects on bone formation such as directly inhibiting osteoblast activity and interfering with the balance of cytokine activity.
- Drug-related — corticosteroids are strongly associated with osteoporosis and increased fracture risk, particularly at vertebrae.6 They mediate their effects via osteoblast receptors and can reduce osteoblast activity and bone mineral content within months of their initiation.3 They can also cause adrenal suppression which reduces oestrogen and androgen synthesis, and consequently exacerbates the situation.

Prevention and treatment
Due to the slow onset of osteoporosis over many years, much of the focus of therapy is concerned with prevention from a young age rather than active treatment. Prevention aims to promote a larger peak bone mass in youth and slow the rate of bone loss with age, unlike treatment to prevent fractures or further fracture after diagnosis. These measures ideally need to be adopted from childhood and include:
- A balanced diet, which includes adequate levels of calcium, vitamin D and protein. A diet generally lacking in any essential requirements can lead to malnutrition, oestrogen deficiencies in women and direct loss of bone mass in both sexes.
- Adequate exercise, including regular weight bearing exercise at least three times a week from a young age. Mechanical forces on the bones stimulate bone formation and help build up a greater peak bone mass. In older people exercise can help reduce the rate of bone loss, although the level of activity should be realistically tailored to ability with increasing age.
Nutraceuticals for bone — melatonin
Melatonin is an endogenous hormone secreted by the pineal gland of the brain. It occurs naturally in various fruits and vegetables, particularly in tomatoes, cucumbers and bananas, although only in small quantities compared with those in the body (5–25μg secreted daily7). As a consequence it is suggested that unfeasibly large quantities of melatonin-rich foods would have to be eaten in order to obtain significant physiological concentrations.
Melatonin is synthesised by the pineal gland in a cyclical fashion over 24 hours, with most released during the hours of darkness. One of the main roles of this hormone is regulating the sleep-wake cycle in response to changes in daylight detected by the retina.
There is also variation in the total amounts of melatonin found in the body over the lifespan, the greatest quantities being found around 1–3 years of age followed by a rapid decline by old age.7 It is for this reason that melatonin has been implicated as a protective nutraceutical against degenerative conditions associated with old age, including osteoporosis. Bone formation and resorption cycles are also thought to follow a circadian pattern which might be in part be modulated by the cyclical secretion of melatonin.
In vitro research has suggested that one of its primary modes of action is as an antioxidant that neutralises the action of toxic free radicals, which cause cellular damage.8 Since it is a highly lipophilic molecule it is able to penetrate lipid membranes of cells readily and exert its effects on intracellular enzymes, such as through inhibition of oxidases. The relevance of this to bone health is that osteoclasts utilise oxygen radicals including superoxide, hydroxyl and peroxyl to initiate the resorption of bone matrix, and so melatonin may halt this activity.1
Another mechanism of melatonin may be through its binding to various intracellular receptors relevant to bone cell regulation and physiology. A number of in vitro and animal studies have been carried out which support this function.
Melatonin may itself have a local hormone action on bone, as there is evidence for its synthesis by both rat and human bone marrow cells.9,10 High concentrations of both melatonin and enzymes involved in its synthesis have been found in cultures of rat osteoblast precursors.11 Application of exogenous melatonin to these types of cell lines led to their differentiation into fully mature osteoblasts which secreted measurable amounts of bone formation markers such as osteocalcin. A similar action was observed in vitro in human osteoblast cultures, in a dose related manner, together with increased secretion of bone collagen precursors. Melatonin, therefore, may act in a paracrine manner through modulation of the RANK- and osteoprotegerin receptorligand signalling pathways that link osteoblast and osteoclast activities described above.
There have also been studies investigating the effects of melatonin in live animals. Providing four-week-old mice with exogenous melatonin led to significant increases in bone mineral density (36 per cent increase) and thickness of trabeculae (19 per cent increase), together with reduction in serum markers of bone resorption compared with a control group.13 Therefore melatonin may act by inhibiting resorption rather than promoting formation.
The effects of melatonin on bone may also be mediated through the presence or absence of oestrogens, particularly as there is a decline in melatonin levels following the menopause. A number of studies have investigated this effect further by measuring melatonin effects in ovariectomised rodents, which serve as a model of the human postmenopausal state. A detailed study examined how melatonin influenced bone metabolism markers and bone mineral density in ovariectomised mice in the presence or absence of oestrogen supplementation.15 In some cases, melatonin augmented the effects of oestrogen in ovariectomised mice with regards to lowering of urine deoxypyrolidine, serum phosphate and serum alkaline phosphatase. Oestradiol and melatonin together also reduced the loss of bone mineral density seen after ovariectomy to a greater extent than either given alone. It therefore appears from this study that melatonin may require a certain quantity of oestrogens to exert its most useful bone protecting action.15
As mentioned in the introduction, growth hormone is implicated in bone formation, particularly in longitudinal growth during childhood, and through promotion of cartilage synthesis and bone cell precursor differentiation in adolescence. One study found a link between melatonin and growth hormone. Eight young male volunteers were administered 0.05, 0.5 and 5mg doses of melatonin, and serum growth hormone levels were measured at regular intervals for up to 150 minutes afterwards. Compared with a placebo, growth hormone levels were found to increase for doses of 0.5 mg and greater.16 Suggested mechanisms are diverse and unclear. It may be through:
- A direct paracrine effect on bone cells as a free radical scavenger or via receptors
- Indirect effects through modulating the action of systemic hormones with known roles in bone metabolism, including oestrogens and growth hormone
Melatonin’s potential as a nutraceutical therefore warrants further study, particularly in clinical trials, and it appears, at present, to have minimal toxicity at standard doses of 0.3–15mg daily.7
L-Carnitine
L-Carnitine acts as a cofactor in oxidative metabolism of fatty acids and is synthesised in the kidney and liver. It is found in the greatest quantities in skeletal and cardiac muscle. It can also be obtained from the diet from meat and dairy produce, with typical intakes being 100–200μmol daily.7
With ageing there is gradual decline in the ability to generate efficiently the energy necessary for anabolic processes, including the protein synthesising activity of osteoblasts. L-Carnitine is necessary as a carrier molecule for the transport of acyl and acetyl groups from fatty acids across the mitochondrial membrane. There is evidence of a decline in both the total levels of this molecule in muscle tissue and also its carrier activity with advancing age, which suggests its link with senile reduction in bone synthesis.17
Endogenous dihydroepiandrosterone (DHEA) is thought to affect levels of L-carnitine through promoting the expression of carnitine-synthesising enzymes.18
An in vitro study on cultured pig osteoblast-like cells examined the action of L-carnitine and DHEA on fatty acid oxidation and osteoblast activation.18 Cells treated with high concentrations of L-carnitine (with or without DHEA) were found to have evidence of increased oxidative respiration and markers of bone formation, namely, collagen and alkaline phosphatase. However, the concentrations of L-carnitine necessary to induce these changes were considerably greater than those known to occur naturally in tissues, which suggests that there may be additional factors in vivo that are needed for its efficient use.
In vitro studies of human osteoblast cell cultures have investigated a second mechanism for L-carnitine involvement in bone metabolism, based on the expression of insulin-like growth factor-I (IGF-I).17 IGF is one of the paracrine factors which mediates osteoblast differentiation and proliferation. Its effects are further fine-tuned via its binding to IGF binding proteins. So far up to six of these proteins have been identified as being expressed in bone tissue, each having different effects on IGF and suggesting a highly sophisticated mechanism of regulation. For example, binding to type 2 inactivates IGFs and leads to decreased osteoblast function, whereas type 3 binding enhances IGF actions.
Administration of L-carnitine or an isovalyl fumarate derivative to the osteoblasts may have influenced function by altering IGF binding protein expression, measured as an increase in mRNA levels. The main changes were found to be an increase in type 3 binding protein mRNA (an activator of IGF action) and a decrease in type 5 binding protein expression (an inhibitor of IGF action). L-Carnitine and its derivative were therefore both able to stimulate osteoblast proliferation (measured as an increase in markers of bone formation, including collagen) although the isovalyl form was found to be more efficient, showing a maximal effect at concentrations 10-fold less than carnitine.
The studies described have only investigated in vitro models and so their findings are of limited value in terms of the use of L-carnitine and DHEA as supplements for bone health. L-Carnitine is, however, used medically at doses of up to 600mg/kg daily in cases of carnitine deficiency, with few serious side effects.7 They serve as useful starting points for future in vivo studies including clinical trials.

Polyunsaturated fatty acids
Fatty acids are derived from triglycerides (which comprise animal fats and plant oils) in the diet, and many can also be synthesised de novo. There is, however, a group which can only be obtained from the diet because the body lacks the ability to synthesise them, and these are known as essential fatty acids.
Many of those of interest derive from fish and plant oils and include:
- Linoleic acid — mainly found in evening primrose, corn and safflower oils
- Eicosapentanoic acid (EPA) — mainly found in oily fish
- Docosahexaenoic acid (DHA)
- Alpha-linolenic acid (ALA) — mainly found in soy and flaxseed
- Gamma-linolenic acid (GLA)
All these fatty acids possess multiple unsaturated double bonds and hence are termed polyunsaturated fatty acids (PUFAs). The first of these double bonds in ALA, EPA and DHA is located at the third carbon atom from its terminal, hence they are described as omega-3 or n-3 PUFAs. Linoleic acid and GLA have their first double bonds at the sixth carbon, and are termed omega-6 or n-6 PUFAs.
Metabolic pathways have been identified which outline how the n-3 and n-6 PUFAs act as precursors for the formation of prostaglandins:
- The n-6 PUFA linoleic acid is converted to GLA, then dihomo-gamma-linolenic acid and finally to arachidonic acid (AA) through the action of a series of enzymes. COX-2 then ultimately converts AA to PGE2. DGLA also forms the precursor of PGE1 through the action of COX-1 enzymes.
- The n-3 PUFA alpha-linolenic acid is converted to EPA and then DHA, which can compete with and inhibit the action of COX-2 and reduce PGE2 formation.
These prostaglandins are released into the immediate bone tissue environment and have conflicting actions.
Dietary fat intake, predominantly of saturated fatty acids (those that comprise animal fat), is thought to be detrimental to bone mineral density, leading to a greater risk of fracture. A recent epidemiological study of almost 15,000 people (the NHANES III trial19) found an inverse relationship between bone mineral density and fat intake in postmenopausal women. A further study found a similar relationship with fracture risk.20
There is evidence from intervention studies that providing supplements of unsaturated fatty acids can prevent a loss in bone mineral density. Elderly women with established osteoporosis were provided with a GLA, EPA and DHA supplement. When compared with a control group receiving coconut oil (containing saturated fatty acids) there was reduced bone loss at the spine and hip.
Animal models have also suggested that n-3 and n-6 PUFAs may have a beneficial effect on bone strength, although if given in excess they were found to lead to bone marrow damage through oxidation (DHA supplements)21 and loss of bone strength in ageing rats (GLA supplements).19 This suggests that optimal concentrations of PUFAs may differ with stages of the life cycle.
There are a number of postulated mechanisms as to how PUFA supplements may promote bone health. PUFAs constitute part of the structures of lipid bilayers of cell membranes and can be enzymatically converted into important mediators of inflammation and bone metabolism, notably the prostanoids.
As components of lipid bilayers, certain PUFAs may influence the structure and function of intestinal brush borders and membranes of kidney tubules. As a consequence this may have an impact on calcium absorption from the gut and renal reabsorption from the urine. One study found that providing fish oil supplements altered intestinal membrane structure such that calcium absorption was enhanced.22
An increase in the n-6:n-3 ratio for PUFA intake may have a negative effect on bone formation, due to an increase in PGE2 over PGE1 formation, resulting in increased bone resorption through the mechanisms described above. In recent times there has been a gradual substitution of saturated animal fats for polyunsaturated plant oils in foods, although many of these substitutes are of the n-6 rather than n-3 type. This means that many diets at present have an n-6:n-3 imbalance by up to 30 times in favour of n-6 which may also be detrimental to health. As a result there is a suggestion that eating n-3 PUFA rich oils (found in large quantities in fish oils and flaxseed) may help redress this imbalance and promote bone formation.
PUFAs of the n-3 type, in particular DHA and EPA, can block the formation of PGE2 through competition with AA for the synthetic enzyme COX-2. Studies of the direct effects of n-3 PUFAs on in vitro cultures of bone cells showed evidence of reduced osteoclast differentiation and bone resorption. In addition, EPA treated osteoblasts were found to promote alkaline phosphatase enzyme expression.5 Studies in intact animal models have further supported this role, with evidence of increased bone formation markers seen in rats26 and piglets25 fed n-3 PUFAs.
Studies have been carried out in vitro to examine the effects of PUFAs and their derivatives on core binding factor α-1 expression, involved in fetal bone development.23 Cultured osteoblast-like cells from rat fetuses, when treated with AA and EPA, demonstrated an increase in expression probably mediated through prostaglandins.
As in the case of melatonin there have also been studies on ovariectomised rats, where it was found that an EPA supplement administered for 35 days arrested the decline in bone strength due to lack of oestrogens, even at minimal calcium levels.27 These results also suggest there may be a synergistic interaction of n-3 PUFAs with oestrogen in preventing bone loss, possibly via the transcription factor mechanism described above. This suggests that n-3 PUFAs may be of value as postmenopausal supplements.
It is also thought that n-6 GLA supplementation may be of benefit in the elderly by favouring formation of PGE1 in favour of PGE2. The reason for this is that greater amounts of GLA shift fatty acid metabolism toward PGE1 formation in favour of AA and PGE2, since the synthetic desaturase enzyme of the latter step has a low activity. There is also evidence from rat studies that a second enzyme involved in conversion of linoleic acid to GLA shows reduced activity with age and therefore can lead to less PGE1 formation.28
Certain fatty acids may also exert their effects via growth hormone. In addition to acting as a promoter of bone formation, growth hormone can also stimulate the breakdown of body fat stores leading to a raised serum free fatty acid concentration. A diet rich in fats may have a negative feedback effect on growth hormone secretion, which was found experimentally in humans given high fat meals over 28 weeks,29 and so may in turn lead to reduction in bone formation, especially as its levels are already low in the elderly.
There are two potential mechanisms by which PUFAs may promote bone health:
- Directly through influencing the metabolism of prostanoid precursors involved in bone homeostasis
- Indirectly via growth hormone action
However, the situation is further complicated by the fact that different types of PUFA have different effects, with effects relating to age and development of the subject. Therefore, more research is necessary to determine which fatty acids and the n-3: n-6 ratio that is necessary for maximal benefit as a nutraceutical supplement.

Conjugated linoleic acid
Conjugated linoleic acid (CLA) refers to a family of geometric isomers of linoleic acid. The main dietary sources of CLA are meats and dairy products derived from grazing animals that have stomachs containing the bacteria that modify dietary linoleic acid.
In vivo rat studies have investigated dietary supplements of CLA on bone mineralisation and markers of bone turnover. Overall, results showed an increase in overall bone mineral content measured as body ash content.30
It has been proposed that, as with the case of n-3 PUFAs, CLA may affect the synthesis of prostaglandins by competing with n-6 PUFA substrates for COX-2, with reduction in formation of detrimental PGE2. A study on human, in vitro, osteoblast-like cell lines confirmed significantly reduced PGE2 and raised osteocalcin secretion,31 although this effect depended on which specific isomeric form of CLA was administered. Reasons for differences in response to different isomers may relate to how they are metabolised and transported in the blood, therefore influencing how readily they can be taken up by bone tissue.
In a study feeding rats with CLA it was shown to enhance calcium absorption in bone, and this effect was enhanced if it was co-administered with n-3 PUFAs.32 It therefore appears that age, dietary PUFA ratios and the isomeric form of CLA may be important factors in its effectiveness and therefore its use as a nutraceutical.
In a randomised, double-blind, intervention trial of CLA versus a placebo in 60 healthy adult males (aged between 39 and 64 years), results showed no significant effect on a wide range of serum and urinary markers of bone metabolism. The lack of any meaningful findings was suggested as being due to use of supa less active isomeric form of CLA, or that the doses used were much lower than those used in the above animal studies.
A double blind study of adult, type 2 diabetes patients who were taking either CLA or n-6 PUFAs (derived from safflower) found a decrease in serum concentrations of the hormone leptin after eight weeks.33 This is a neuroendocrine factor, secreted by the central nervous system, that has a central role in appetite and lipid metabolism. It is proposed as a local regulatory factor in bone-promoting osteoblast activity.
More studies are necessary to determine if CLA has any potential effect on osteoblast or osteoclast functioning and whether it is of any value in bone health.
Phytoestrogens
The use of phytoestrogens for treating menopausal symptoms, including osteoporosis has been recently reviewed,34 and several large-scale epidemiological studies and clinical trials have been carried out for this group.
The major examples of phytoestrogens are derived from a large number of plant sources. These include:
- Isoflavones — found in chickpeas, red clover and soybean. Soy is the richest source. The main isoflavones that have been extracted and purified are genistein (60 per cent of the isoflavone content in soy),35 daidzein (25 per cent of the isoflavone content in soy) and glycitein (15 per cent of the isoflavone content in soy), and are found in their glycosylated (glycone) form within plant tissues.
- Lignans found in various legumes, grains and flaxseed. Examples of isolated compounds include secoisolariciresinol diglucoside, isolariciresinol, and pinoresinol.
They are thought to be most promising in their role as supplements in preventing and treating postmenopausal osteoporosis, because of their similarity in structure to estradiol and therefore as potential replacements for oestrogen deficiency. As a result, most studies with phytoestrogens have been restricted to female subjects during or after menopause.
These compounds are able to act as weak agonists at oestrogen receptors, specifically the beta type located on osteoblast membranes. As they are lipophilic molecules they can readily traverse cell membranes and bind to cytoplasmic oestrogen receptors, which go on to act as transcription factors. This leads to increased gene expression of alkaline phosphatases and osteocalcin (bone formation factors), osteoprotegerin (an inhibitor of osteoclast activity) and IGF (a promoter of osteoblast activity) resulting in reduced bone resorption and increased bone formation. Phyto-oestrogens are particularly favourable over other forms of oestrogen replacement because they have no activity at the alpha form of oestrogen receptors located in the breast and uterus, so have none of the adverse effects on these tissues associated with standard HRT.36
Experiments with cultures of osteoblastlike cells have found evidence of direct responses to administered isoflavones, although at levels much greater than would be obtained in normal diets. Examples of effects noted include increased alkaline phosphatase and collagen synthesis activity in osteoblasts from mice administered with genestein or daidzein.37 The positive effects of soy isoflavones in reducing loss of bone mineral density as well as promoting bone formation over resorption have been reviewed.38 Similar studies with female rats have shown that providing flax lignans in the diet promoted bone strength early in development.39
Epidemiological studies have shown strong evidence that soy isoflavones have a positive effect on bone mineral density. These were borne out by the fact that far lower rates of osteoporosis and fractures have been observed in oriental women compared with their western counterparts, even when height and weight have been accounted for. Soy has been part of oriental diets far longer than in the West, with daily soy protein consumption being significantly greater (55g daily noted in Japanese women).35
A prospective cohort study of over 24,000 postmenopausal Chinese women, aged 40–70 years, used evidence of clinical fracture (considered more appropriate than markers of bone formation for ascertaining potential value as a supplement) as its main measure against soy consumption.40 The main findings after a four-year follow-up were that high soy intakes correlated with a significantly reduced risk of postmenopausal fractures, particularly in the first few postmenopausal years. This is of interest as this is where bone loss rates are thought to be greatest and therefore the optimal time to use soy supplements.
Clinical intervention trials have also been carried out using isoflavone extracts and have demonstrated varying levels of significance. Reduction in loss of bone mineral density and change in biochemical markers was variably reported.34
Most intervention studies focused on soy isoflavones, but one study attempted to compare soy isoflavones with flaxseed lignan supa plementation in a group of 46 postmenopausal women over 16 weeks. They were randomly assigned a soy, flax or wheat (placebo) muffin daily.41 Results found that only flax appeared to have significant effects on serum alkaline phosphatase concentrations. This may be explained by the fact that flaxseed oil is also a rich source of alphalinolenic acid, one of the beneficial n-3 PUFAs associated with bone health described above.
Studies have attempted to determine which isoflavones, in particular, are likely to have the greatest effect in preventing bone loss, with many proposing genistein. However, an in vivo study using rats has shown evidence that daidzein may be the most active.42 Its action may, however, be mediated via the way the body metabolises it. Following ingestion there is initial hydrolysis of daidzein’s sugar moiety by enzymes from the intestinal brush border, enabling its ready absorption into the blood stream. Within peripheral tissues daidzein is enzymatically converted to a metabolite known as equol, which has been demonstrated as having twice the oestrogenic action in rats compared with its parent molecule.43
In addition to the isoflavones contained in soy protein, soy can replace animal sources in the diet, thus providing added benefit. As described above, animal proteins interfere with the process of calcium reabsorption by the kidneys and thus, indirectly, lead to a reduction in bone formation.
Further studies are therefore needed to determine beneficial doses of phytoestrogens, with current recommendations being 40–100mg of isoflavones daily,35 based on a standard oriental dietary intake. There has been some suggestion that phytoestrogens may have adverse effects on the uterus and fertility in animals due to their oestrogen-like actions, although at the doses suggested they are thought to be safe.44 Overall, phytoestrogens appear to show the most promise in all of the nutraceuticals so far described, although there has been limited investigation into their use in pre and postmenopausal osteoporosis.
Conclusion
Due to the shortcomings of current treatments for osteoporosis, there is a strong argument in favour of patients taking a more active role to prevent the onset of osteoporosis through healthy diet and exercise from a young age, or the use of individualised nutraceutical therapies for prevention and treatment. Although there is some promise being generated from the compounds considered in this article, prevention of osteoporosis still appears likely to be better than cure.
References
- Ferguson N. Osteoporosis in focus. London: Pharmaceutical Press, 2004.
- Tanna N. Osteoporosis and its treatment. The Pharmaceutical Journal 2005;275:521–2.
- World Health Organization Scientific Group on the Prevention and Management of Osteoporosis. Prevention and management of osteoporosis: report of a World Health Organization scientific group. Geneva: WHO, 2003.
- Goldring SR. Inflammatory mediators as essential elements in bone remodelling. Calcified Tissue International 2003;73:97–100.
- Watkins BA, Li Y, Lippman HE, Feng S. Modulatory effect of omega-3 polyunsaturated fatty acids on osteoblast function and bone metabolism. Prostaglandins Leukotrienes and Essential Fatty Acids 2003;68:387–98.
- National Osteoporosis Society. What is osteoporosis? Available at: www.nos.org.uk/about.htm. Accessed 28 June 2006.
- Rapport L, Lockwood B. Nutraceuticals. London: Pharmaceutical Press, 2002.
- Cardinali DP, Ladizesky MG, Boggio V, Cutrera RA, Mautalen C. Melatonin effects on bone: experimental facts and clinical perspectives. Journal of Pineal Research 2003;34:81–7.
- Conti A, Conconi S, Hertens E, Skwarlo-Sonta K, Markowska M, Maestroni JM. Evidence for melatonin synthesis in mouse and human bone marrow cells. Journal of Pineal Research 2000;28:193–202.
- Tan DX, Manchester LC, Reiter RJ, Qi WB, Zhang M, Weintraub ST, et al. Identification of highly elevated levels of melatonin in bone marrow: its origin and significance. Biochimica et Biophysica Acta 1999;1472:206–14.
- Roth JA, Kim BG, Lin WL, Cho MI. Melatonin promotes osteoblast differentiation and bone formation. Journal of Biological Chemistry 1999;274:22041–7.
- Fernandes G, Lawrence R, Sun D. Protective role of n-3 lipids and soy protein in osteoporosis. Prostaglandins Leukotrienes and Essential Fatty Acids 2003;68:361–72.
- Koyama H, Nakade O, Takada Y, Kaku T, Lau KH. Melatonin at pharmacologic doses increases bone mass by suppressing resorption through down-regulation of the RANKL-mediated osteoclast formation and activation. Journal of Bone and Mineral Research 2002;17:1219–29.
- Suzuki N. Hattori A. Melatonin suppresses osteoclastic and osteoblastic activities in the scales of goldfish. Journal of Pineal Research 2002;33:253–8.
- Ladizesky MG, Boggio V, Albornoz LE, Castrillon PO, Mautalen C, Cardinali DP. Melatonin increases estradiol-induced bone formation in ovariectomised rats. Journal of Pineal Research 2003;34:143–51.
- Forsling ML. Wheeler MJ. Williams AJ. The effect of melatonin administration on pituitary hormone secretion in man. Clinical Endocrinology 1999;51:637–42.
- Colucci S, Mori G, Vaira S, Brunetti G, Greco G, Mancini L, et al. L-Carnitine and isovaleryl L-carnitine fumarate positively affect human osteoblast proliferation and differentiation in vitro. Calcified Tissue International 2005;76:458–65.
- Chiu KM, Keller ET, Crenshaw TD, Gravenstein S. Carnitine and dehydroepiandrosterone sulfate induce protein synthesis in porcine primary osteoblast-like cells. Calcified Tissue International 1999;64:527–33.
- Corwin RL. Effects of dietary fats on bone health in advanced age. Prostaglandins Leukotrienes and Essential Fatty Acids 2003;68:379–86.
- Kato I, Toniolo P, Zeleniuch-Jacquotte A, Shore RE, Koenig KL, Akhmedkhanov A, et al. Diet, smoking and anthropometric indices and postmenopausal bone fractures: a prospective study. International Journal of Epidemiology 2000;29:85–92.
- Umegaki K, Hashimoto M, Yamasaki H, Fujii Y, Yoshimura M, Sugisawa A, et al. Docosahexaenoic acid supplementationincreased oxidative damage in bone marrow DNA in aged rats and its relation to antioxidant vitamins. Free Radical Research 2001;34:427–35.
- Coetzer H, Claasen N, van-Papendorp DH, Kruger MC. Calcium transport by isolated brush border and basolateral membrane vesicles: role of essential fatty acid supplementation. Prostaglandins Leukotrienes and Essential Fatty Acids 1994;50:257–66.
- Marie PJ. Cellular and molecular alterations of osteoblasts in human disorders of bone formation. Histology and Histopathology 1999;14:525–38.
- Weiler HA, Fitzpatrick-Wong SC. Modulation of essential (n-6):(n-3) fatty acid ratios alters fatty acid status but not bone mass in piglets. Journal of Nutrition 2002;132:2667–72.
- Watkins BA, Li Y, Allen KG, Hoffmann WE, Seifert MF. Dietary ratio of (n-6)/(n-3) polyunsaturated fatty acids alters the fatty acid composition of bone compartments and biomarkers of bone formation in rats. Journal of Nutrition 2000;130:2274–84.
- Sakaguchi K, Morita I, Murota S. Eicosapentaenoic acid inhibits bone loss due to ovariectomy in rats. Prostaglandins Leukotrienes and Essential Fatty Acids 1994;50:81–4.
- Lorenzini A, Bordoni A, Spano C, Turchetto E, Biagi PL, Hrelia S. Age-related changes in essential fatty acid metabolism in cultured rat heart myocytes. Prostaglandins Leukotrienes and Essential Fatty Acids 1997;57:143–7.
- Bhathena SJ, Berlin E, Judd JT, Kim YC, Law JS, Bhagavan JN, et al. Effects of n3 fatty acids and vitamin E on hormones involved in carbohydrate and lipid metabolism in men. American Journal of Clinical Nutrition 1991;54:684–88.
- Park Y, Albright KJ, Liu W, Storkson JM, Cook ME, Pariza MW. Effect of conjugated linoleic acid on body composition in mice. Lipids 1997;32:853–8.
- Cusack S, Jewell C, Cashman KD. The effect of conjugated linoleic acid on the viability and metabolism of human osteoblast-like cells. Prostaglandins Leukotrienes and Essential Fatty Acids 2005;72:29–39.
- Kelly O, Cusack S, Jewell C, Cashman KD. The effect of polyunsaturated fatty acids, including conjugated linoleic acid, on calcium absorption and bone metabolism and composition in young growing rats. British Journal of Nutrition 2003;90:743–50.
- Doyle L, Jewell C, Mullen A, Nugent AP, Roche HM, Cashman KD. Effect of dietary supplementation with conjugated linoleic acid on markers of calcium and bone metabolism in healthy adult men. European Journal of Clinical Nutrition 2005;59:432–40.
- Belury MA, Mahon A, Banni S. The conjugated linoleic acid (CLA) isomer, t10c12-CLA, is inversely associated with changes in body weight and serum leptin in subjects with type 2 diabetes mellitus. Journal of Nutrition 2003;133:257S–260S.
- Kotecha N, Lockwood B. Soy-relieving the symptoms of menopause and fighting osteoporosis. Pharmaceutical Journal 2005;275:483–7.
- Jones M. Soya and osteoporosis. NutraCos 2003;26–28.
- Chen YM, Ho SC, Lam SS, Ho SS, Woo JL. Soy isoflavones have a favorable effect on bone loss in Chinese postmenopausal women with lower bone mass: a doubleblind, randomized, controlled trial. Journal of Clinical Endocrinology and Metabolism 2003;88:4740–7.
- Dang ZC, Papapoulos S, Lowik C. Phytoestrogens enhance osteogenesis and concurrently inhibit adipogenesis. Journal of Nutrition 2002;132:617S.
- Setchell KD, Lydeking-Olsen E. Dietary phytoestrogens and their effect on bone: evidence from in vitro and in vivo, human observational, and dietary intervention studies. American Journal of Clinical Nutrition 2003;78:593S–609S.
- Ward WE, Yuan YV, Cheung AM, Thompson LU. Exposure to purified lignan from flaxseed (Linum usitatissimum) alters bone development in female rats. British Journal of Nutrition 2001;86:499–505.
- Zhang X, Shu XO, Li H, Yang G, Li Q, Gao YT, et al. Prospective cohort study of soy food consumption and risk of bone fracture among postmenopausal women. Archives of Internal Medicine 2005;165:1890–5.
- Brooks JD, Ward WE, Lewis JE, Hilditch J, Nickell L, Wong E, et al. Supplementation with flaxseed alters estrogen metabolism in postmenopausal women to a greater extent than does supplementation with an equal amount of soy. American Journal of Clinical Nutrition 2004;79:318–25.
- Picherit C, Coxam V, Bennetau-Pelissero C, Kati-Coulibaly S, Davicco MJ, Lebecque P, et al. Daidzein is more efficient than genistein in preventing ovariectomy-induced bone loss in rats. Journal of Nutrition 2000;130:1675–81.
- Setchell KD. Absorption and metabolism of soy isoflavonesfrom food to dietary supplements and adults to infants. Journal of Nutrition 2000;130:654S–5S.
- Barnes S. Phyto-oestrogens and osteoporosis: what is a safe dose? British Journal of Nutrition 2003;89:S101–8.


