Shutterstock.com
Broadly defined as a difficulty initiating or maintaining sleep, insomnia disorder (or insomnia), is an often neglected diagnosis. This may be owing to its high subjectivity, its perception as a condition of low importance or even that it is not a medical condition at all[1],[2],[3],[4]
. At least one in three people will experience insomnia that can cause sleeplessness or waking without feeling refreshed, during their lifetime. This ultimately impairs daytime function causing tiredness, impaired concentration and mood disturbance[5],[6],[7]
.
Sleep affects all body systems and deleterious effects to the immune, cardiovascular, endocrine and neuronal systems are seen when sleep deprivation occurs[8]
. Sleep deprivation has been shown to impair cognitive and motor function to the same level as alcohol intoxication — Cappuccio et al. showed that injury-involved car accidents were six times more likely to happen in drivers who had less than six hours’ sleep the previous night[9],[10]
.
The bi-directional relationship between sleep and health makes sleep disorders — both diagnosed and undiagnosed — a significant global health burden. In addition, poor sleep is a common predictor of cardiovascular, metabolic, neurological and psychiatric ill health[11]
. This highlights the important role pharmacists and healthcare professionals have in exploring sleep with their patients during routine consultations.
This article — the first of two focusing on insomnia — aims to provide an overview of sleep and the disorders associated with it, and considers how pharmacists and other healthcare professionals can recognise, diagnose and potentially prevent this disorder. The second article (part 2) will focus on the pharmacotherapies, both over-the-counter and prescribed, for insomnia and how cognitive behavioural therapy for insomnia (CBT-I) can be incorporated into pharmacy consultations.
Normal sleep
To understand insomnia, it is important to first understand the process of sleep and the patient’s expectations.
Brainwaves
Sleep is a restorative physiological process characterised by an alteration in brainwave activity, viewed via an electroencephalograph (EEG). This reading shows that brainwave frequency slows as the sleeper drifts from alertness towards deep sleep (N3 phase of sleep)[12]
. In addition, brain, eye, skeletal muscle and heart activity change throughout differing phases of sleep, as shown through polysomnography (PSG).
Clocks and rhythms
According to Alexander Borbély’s two process model, sleep is governed by two distinctly separate rhythms: the circadian rhythm (i.e. the intrinsic biological clock) and the homeostatic sleep drive (i.e. sleep pressure)[13],[14]
. The circadian rhythm is an endogenous cycle of around 24 hours, influenced by melatonin, and matched to environmental ‘zeitgebers’ (external or environmental cues that synchronise the biological rhythms to the earth’s 24-hour light/dark cycle) such as light, temperature and food (see Figure 1)[13]
.
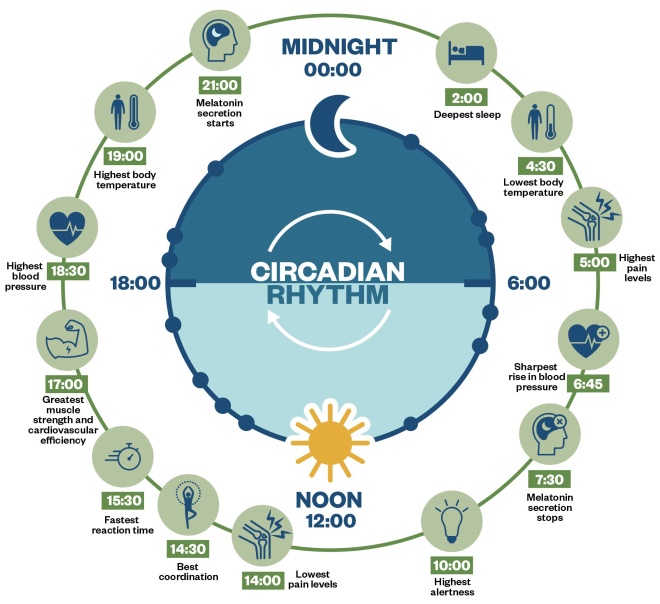
Figure 1: Circadian rhythm
Sleep pressure is governed by adenosine levels in the pre-frontal cortex of the brain: for every hour of waking, there is a steady accumulation of adenosine, increasing the urge to sleep. During sleep, adenosine is anabolised back to adenosine triphosphate (ATP). Together, sleep pressure and the circadian rhythm form the sleep–wake cycle. Optimising factors, such as food, exercise, temperature, light and noise improve the sleep–wake cycle and are generally referred to as ‘sleep hygiene’ (more detail will be covered in part 2)[15]
.
The sleep–wake cycle also includes another cycle: the ultradian rhythm, which this can be depicted as a graph that shows the changes in sleep stages (see Figure 2)[16]
.
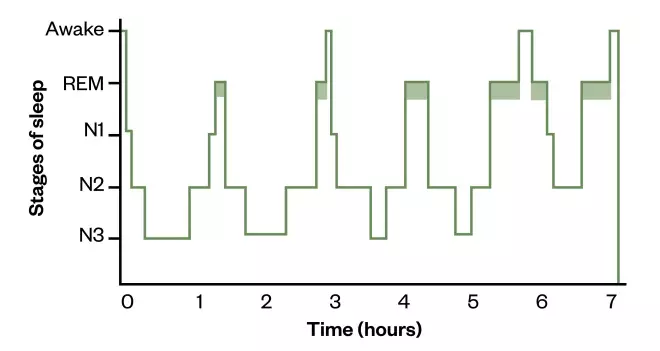
Figure 2: Hypnogram depicting normal adult sleep
On a hypnogram, a sleep cycle is usually around 90 minutes and there are four to six cycles of rapid eye movement (REM) sleep/non-REM sleep stages that occur during a major period of sleep. Most slow-wave sleep occurs in the first one or two cycles; this is the deepest period of sleep. The second half of the sleeping period contains most REM sleep and little or no slow-wave sleep, and may contain brief periods of wakefulness which can be recorded but are not usually perceived. The stage that occurs before waking is normally REM sleep.
N1: light sleep; N2: deeper, memory forming sleep; N3: deep, restorative sleep; REM: rapid eye movement
Managing expectations of normal sleep
Even in healthy adults, the need for sleep decreases as age increases, with the time spent asleep and the structure of this sleep, or ‘sleep architecture’, changing[17]
. As sleep latency (i.e. the time taken to fall asleep) increases, more time is spent in stages 1 and 2 of non-rapid eye movement sleep and less time is spent in rapid eye movement (REM) sleep stages[17]
.
Managing patient expectations around normal sleep length and quality through ageing is important. For example, no matter the measures taken, it is unlikely that an individual aged over 65 years will be able to routinely sleep like an adolescent again. Consequently, it is possible that in advanced age, those requiring less sleep are unnecessarily prescribed medicines to aid sleep (e.g. hypnotics) to try to reach unrealistic sleep targets, leading to over-sedation and potentially increasing the risk of falls. In addition, cognitive impairment as a side effect of hypnotics can also lead to false dementia diagnoses[18]
. Comorbidity and polypharmacy is more common in advanced age, and the impact of additional disease states and medicines can further disturb sleep[19]
.
Insomnia disorder
Although the International Classification of Diseases 10th edition manual and Diagnostic and Statistical Manual of Mental Disorders 5th edition (DSM-V) definitions of insomnia differ slightly, it is described in both as a ‘difficulty initiating and maintaining sleep’[1],[20]
.
The International Classification for Sleep Disorders (ICSD-3) outlines three diagnostic criteria for insomnia:
- A report of sleep initiation or maintenance problems;
- Despite adequate opportunity and circumstances to sleep;
- Resulting in daytime consequences[5]
.
Chronic insomnia is differentiated from insomnia by ICSD-3 when these criteria last for more than three months and occur on three or more nights of the week[5]
.
However, the National Institute for Health and Care Excellence differentiates acute or short-term insomnia from long-term insomnia at the four-week time point[6]
. This figure relates to the maximum length stipulated on the product licence of many hypnotics, whereas the ICSD-3 definition aims to differentiate persistent insomnia from self-limiting insomnia, which may not require specialist input. Regardless of the duration of symptoms, insomnia is a clinical predictor of depression and should be treated if it is distressing to the patient[21]
. Critically, the presentation of insomnia is subjective and pharmacists should be open-minded during consultations.
Spielman’s model of insomnia
Factors including age, disease state and lifestyle can predispose people to insomnia. Spielman’s model is useful when interpreting insomnia[22]
. It considers precipitating factors, such as a bereavement or exacerbation of a disease state, that can trigger insomnia and how perpetuating insomnia continues long-term once the original precipitating factor has been resolved. This type of insomnia is difficult to treat as its perpetuating nature can be a result of cognitive and behavioural factors, rather than external causes and as a result may require a level of cognitive restructuring, likely through CBT-I (see part 2)[23]
,[24]
.
This model can be used to:
- Take steps to reduce factors predisposing patients to insomnia;
- Discern between disease states which are not insomnia, but may precipitate it (e.g. restless leg syndrome and sleep apnoea);
- Identify perpetuating insomnia, which tends to be largely psychological in nature and caused by behaviours developed in response to the original precipitating insomnia[25]
.
Risk factors
Broadly, risk factors can be considered as those that contribute to having insomnia disorder, but also how insomnia can be a risk factor for other conditions. As these are not always mutually exclusive, it is appropriate to group these together.
Up to 50% of older people experience insomnia, although this may be owing to a decline in sleep efficiency and unrealistic expectations of sleep duration at this stage of life[5],[26],[27],[28],[29],[30]
. However, this could also be a result of comorbidities that increase the likelihood of developing insomnia[31]
. Women are more likely to experience insomnia than men, as are individuals who are unemployed, or have low levels of education or financial income[5],[26],[27],[28],[31]
,[32]
.
The bi-directional relationship between insomnia and other health conditions is well established, with the prevalence of insomnia in adults with two or more illnesses being over 50%[26],[33]
. In addition, insomnia is a risk factor for the development or exacerbation of chronic health conditions, which in turn can worsen insomnia. Cardiovascular, gastrointestinal and pulmonary diseases are more prevalent in patients with insomnia than those without[5],[27],[34]
.
Often, multiple underlying pathologies can lead to symptoms that disrupt sleep, such as nocturnal polyuria can be present in several disease states, including: diabetes, prostate enlargement, restless leg syndrome and obstructive sleep apnoea[6],[29],[35]
.
In 1987, a historical study of male participants (n=4,064) illustrated an increased prevalence of insomnia symptoms in subjects with obesity and untreated hypertension, COPD, rheumatism or diabetes[36]
. This is reflected in the findings of a larger literature study published in 2009 that showed similar relationships in both genders (n=25,579)[34]
.
Insomnia is also a risk factor for anxiety, depression and an increased perception of pain[30]
. Insomnia and mental health conditions are frequently concurrent in the general population, with epidemiological evidence suggesting that a third of patients who were diagnosed with a mental health disorder, and also showed symptoms of insomnia or expressed dissatisfaction with sleep, did not have their insomnia diagnosed as a condition in its own right[28],[34]
. Although conversations around sleep may provide an appropriate segue into discussions around mental health, in practice, the opportunity to treat patients holistically may be being missed.
Diagnosis
Typical daytime symptoms of insomnia include fatigue, irritability and poor concentration. Some patients develop increased anxiety around bedtime, worrying about their likelihood of poor sleep, leading to an underlying state of hyperarousal, intrusive thoughts and a racing mind[37]
. This creates a vicious cycle of maladaptive, compensatory sleep behaviours, which may perpetuate insomnia.
Considering the psychological component in pain can help pharmacists and healthcare professionals understand its large impact in insomnia. While there are objective parameters that can be measured, such as sleep efficiency, patient-reported symptoms are largely subjective and, as such, the account of a bed partner, if present, can be useful. Additionally, maintaining a sleep diary for a minimum of two weeks can provide useful insight into the extent and nature of the sleep disturbance[5],[6]
. Sleep diaries can identify common problems and pharmacists can often provide helpful advice, such as:
- Excessive caffeine consumption — advise to reduce number of drinks and switch to decaffeinated options (up to six hours before bedtime);
- Long sleep latency (period between getting into bed and falling asleep) — advise to start a ‘screen-free’ wind down period prior to bed and only getting into bed when feeling sleepy;
- Waking periods in the night — advise that sleep cycles are roughly 90 minutes in length and that it is natural to wake when one ends. If awake for more than 15 minutes, advise the patient to leave the bed and engage in a wind down activity, such as reading a book, or having a light carbohydrate snack, and only returning to bed when sleepy;
- Waking periods associated with intrusive thoughts — advise patients to keep a notepad or journal next to the bed to write down any worries keeping them awake to address in the morning.
PSG, or a sleep study, requires referral to secondary care, but is not required for diagnosis[34]
. PSG may be useful if other sleep disorders are suspected, or if there is a discrepancy between the subjective symptoms the patient reports and the reality of their sleep pattern.
To help patients begin to track their sleeping pattern and address any bad sleep habits (see part 2), pharmacists should take a sleep history and start patients on sleep diaries (see Box 1)[38],[39]
. As part of the consultation pharmacists can also conduct a ‘STOPBang’ questionnaire to screen for sleep apnoea[39]
. If this or other sleep disorders are suspected, the patient should be referred to a sleep clinic.
Box 1: Questions to help reveal a patient’s sleep history
- How often do you find it difficult to get to sleep or stay asleep?
- Does this occur most nights?
- How does this affect your daytime activities?
- Consider if their answer fits the diagnostic criteria of insomnia or if another sleep disorder is possible.
- Have you noticed any patterns or triggers? Do you know how long this has occurred for?
- Do you snore heavily?
- It may be appropriate to screen for obstructive sleep apnoea — if they answer ‘yes’, complete the STOP-Bang questionnaire (a tool to identify sleep apnoea).
- When you try to relax in the evening or sleep at night, do you ever have unpleasant, restless feelings in your legs that can be relieved by walking or movement?
- This is a rapid screening question for restless leg syndrome (RLS) approved by the European Journal of Neurology. More information on RLS can be found at:
rls-uk.org/diagnosis.
- This is a rapid screening question for restless leg syndrome (RLS) approved by the European Journal of Neurology. More information on RLS can be found at:
- Can you tell me what over-the-counter or prescribed medicine you take? How often do you drink alcohol and caffeine? Do you smoke or use any nicotine replacement therapy?
- Talk me through your average day, hour by hour.
- When was the last time you napped during the day? Is this a common occurrence?
- Tell me about your exercise routines? Do you feel exercise during the day could help your sleep? Do you ever exercise late in the day and feel it negatively affects your sleep?
- I think a sleep diary could be beneficial in understanding your sleep problems; would you be interested in starting one?
- A sleep diary and other resources, including an overview video, can be found at: cntw.nhs.uk/smartersleep.
- A sleep diary and other resources, including an overview video, can be found at: cntw.nhs.uk/smartersleep.
Scales and indexes for insomnia
There are two tools available to help assess sleep quality and understand a patient’s experience of insomnia diagnosis that have been validated against the DSM-V criteria for insomnia. The insomnia severity index is a self-reporting index relating to patient concerns and perception of symptoms of insomnia over the previous two weeks, and incorporates a seven-item questionnaire linked to DSM-V. The sleep condition indicator is a two-item version of this questionnaire comprising of a 0–4 Likert rating scale and is useful for screening for insomnia in primary care[40],[41],[42]
. These tools can be useful to record changes to sleep quality over time[43]
.
Other scales and indexes that may be useful for assessing the levels of sleepiness and depression respectively, both of which are associated with insomnia, include:
- The Epworth sleepiness scale, which is more useful in diagnosing other sleep disorders that disturb sleep (e.g. obstructive sleep apnoea); and
- The Beck depression inventory, which can be useful for monitoring the effect of improved sleep on depression[43]
.
Figure 3 outlines the insomnia diagnosis pathway as per the British Association for Psychopharmacology consensus statement[32]
. The flowchart can be used by diagnosticians and indicates several interventions that could be made to address factors that may cause sleep disturbances, and those that could be reversed to resolve the symptoms of insomnia.
If sleep habits (e.g. watching TV or exercising before bed) and sleep scheduling (i.e. waking and sleeping times) cannot be optimised, then the intake of stimulating substances (e.g caffeine, nicotine or medicine) should be reviewed. If all of these factors have been optimised and symptoms persist, then it is likely that a diagnosis of insomnia is appropriate and referral for treatment should be considered.
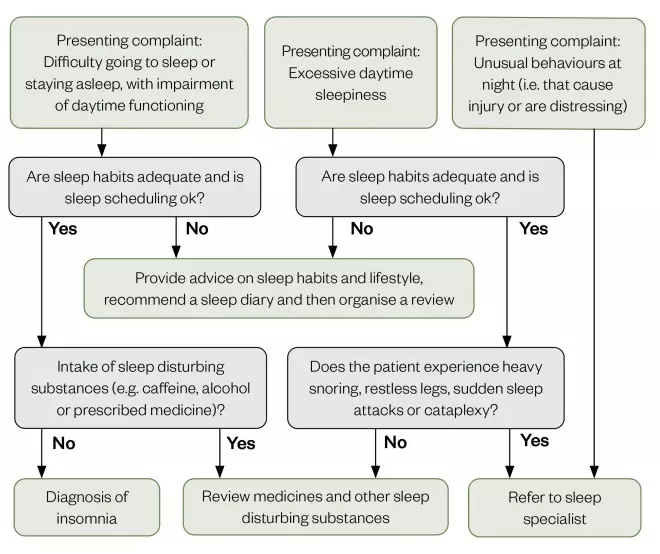
Figure 3: Algorithm to determine diagnosis of insomnia
Source: Adapted from J Psychopharmacol
[32] with permission
Sleep diaries
A useful way of recording symptoms can be through completion of a sleep diary. Patients are asked to complete a chart on which they record the times into and out of bed; time asleep; time they woke; and any night-time awakenings[44]
.
Factors that may affect sleep, such as smoking or caffeine, alcohol and food (e.g. late meals or chocolate) intake, should also be recorded. The diary should be completed over a minimum of two weeks in order to provide good insight into the nature of the patient’s experience of sleep disorder[45],[46]
. Figure 4 shows the typical appearance of insomnia within a diary, characterised by long sleep latency and frequent night-time awakenings.
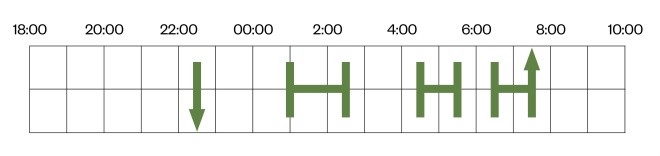
Figure 4: Typical appearance of insomnia disorder within a sleep diary
The typical appearance of insomnia within a diary, characterised by long sleep latency — the downwards arrow indicates that the patient went to bed at 22:30, but did not fall asleep until 1:00 — and experienced frequent night-time awakenings (e.g. gap recorded between 2:30–4:30 and 5:30–6:30) — before waking up at 7:30.
Sleep efficiency
This is a useful parameter that can be calculated from sleep diaries. It takes into account the time asleep and the total time in bed, and can be calculated using the formula in Figure 5.
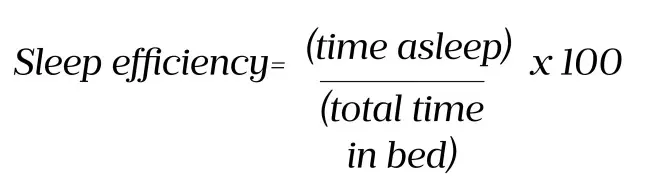
Figure 5: Formula for calculating sleep efficiencÂy
A sleep efficiency greater than 85% is consistent with normal sleep. A sleep efficiency less than 50% indicates severe insomnia.
The formula can be used to calculate the sleep efficiency of the patient from Figure 4. The patient went to bed at 22:30 and had night-time awakening, before waking up for the day at 7:30: the total time in bed was 9 hours, with a total time asleep of 3.5 hours. Using the sleep efficiency formula of ‘time asleep’ divided by ‘total time in bed’ multiplied by 100 indicates a sleep efficiency of 39%. Therefore, in this example, the patient can be said to have sleep efficiency that would be described as severe insomnia.
It is clear that sleep efficiencies can vary largely night-to-night, so it is best practice to average these findings over a period of at least two weeks[45]
. These data can also be used to track patient response to pharmaceutical care interventions by observing sleep efficiency over time (see Table)[40],[41]
.
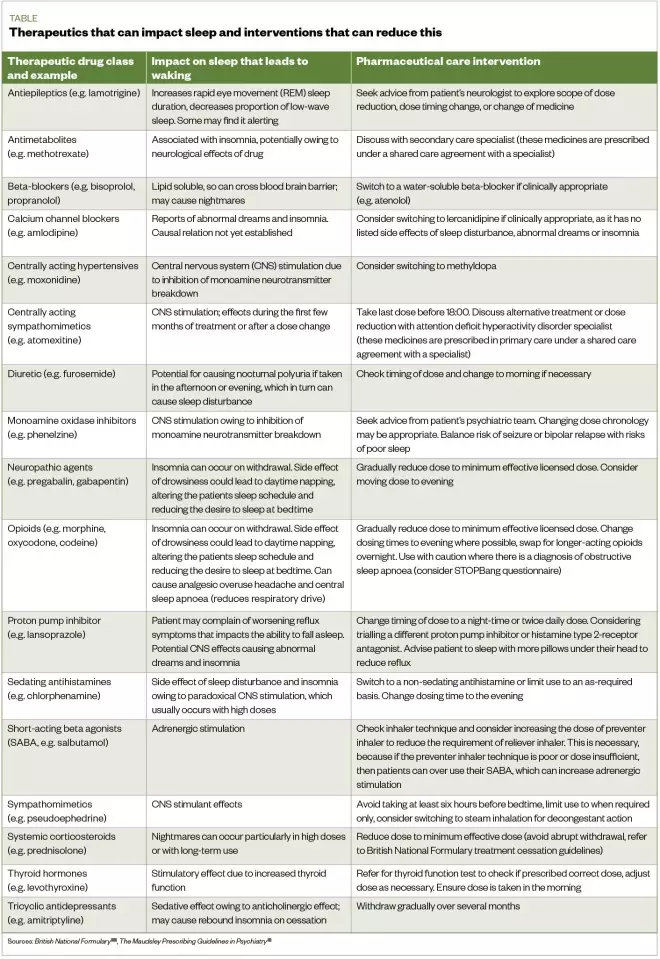
Table: Therapeutics that can impact sleep and interventions that can reduce this
Sources: British National Formulary
[40], The Maudsley Prescribing Guidelines in Psychiatry
[41]
Opportunities for insomnia intervention
Inconsistent approaches to diagnosis and lack of awareness often lead to insomnia remaining undiagnosed
[2]. This is in part owing to a lack of education for both the general population and healthcare professionals around sleep and sleep disorders, highlighting an important role for pharmacists in the identification and consequent treatment of patients with insomnia in community, general practice and hospital settings[42]
.
Many patients purchase over-the-counter medicines — or collect their prescribed medicines relating to sleep — from community pharmacies, providing an opportunity for pharmacists to engage patients in discussions regarding healthy sleep behaviours. Pharmacists working in general practices may spot a potential undiagnosed case of insomnia — during chronic disease management or medication review consultations — for referral to colleagues in general practice for further investigations and formal diagnosis.
Pharmacists who lack confidence in this area may build their confidence by following up with patients to identify outcomes — for example, by placing a note on the patient’s records to speak to the patient to see how they responded to proposed treatments, or speaking to another healthcare professional looking after the patient to monitor progression and management. Although finding time to do this may be challenging, following up on recommendations will help develop practice.
Pharmacists working in hospitals in secondary or tertiary care organisations may care for patients who have problems sleeping as a direct result of being an inpatient, and who may be prescribed hypnotics, such as zopiclone, temazepam or melatonin. If a patient is discharged on hypnotics, ward staff should advise them (and their colleagues in primary care) in their discharge letter that continuation of these medicines requires rapid review after leaving hospital.
The opportunities described above demonstrate areas where pharmacists can contribute to the identification and management of insomnia.
Protecting people from poor sleep
Many causes of sleep disruption are preventable; therefore, issues with sleep can be prevented before insomnia occurs. Poor sleep can be improved by modifying factors, such as stress, excitement, anticipation, pain, illness, changes in sleep schedule, daytime napping, change of time zone or altitude, medicine, noise and light[5]
.
There are three simple steps every pharmacist can take to understand and protect a patient’s sleep:
- Ask the question “How are you sleeping?” — especially when talking to patients with chronic disease;
- Review medicines and consider whether any are stimulating or sedating at the inappropriate times (see Table);
- Educate patients about what is considered normal sleep and how their sleep can be improved.
Although patient-directed information about this may be limited, the examples below outline the effect of lifestyle and medicines on sleep, and highlight important changes that should be made:
- Eating a high-calorie meal within one hour of bedtime slows down the onset of sleep owing to increased gastric volume causing discomfort[46]
. For patients experiencing dyspepsia (indigestion) that keeps them awake, taking a proton pump inhibitor at night may aid sleep. For patients who consume chocolate and soft drinks, the high sugar and caffeine content can also disrupt sleep; - Exercise during the day improves sleep; however, exercise just before bedtime can lead to difficulties getting to sleep[47],[48]
; - Inactivity can cause insomnia, particularly for those remaining indoors. This can create negative cycles, as daytime fatigue owing to insomnia can create a lack of motivation to do things[47],[49],[50],[51]
; - Nicotine can cause insomnia through its stimulant action, and overnight withdrawal symptoms, which decrease N3 sleep[52]
. Smoking cessation can precipitate or exacerbate insomnia[53]
; - Caffeine (in tea and coffee) temporarily reduces homeostatic sleep pressure, increases sleep latency and decreases N3 sleep[32],[54]
. Drinking 5–6 cups of coffee per day can create a constant level of caffeine in the bloodstream and drinking caffeine up to six hours before bedtime can cause sleep disruption[54]
; - Alcohol helps induce sleep owing to its initial sedative action[55],[56]
. However, later in the night it causes a dose-dependent disruption to sleep by delaying the onset of, and fragmenting REM sleep. This results in frequent awakening and a greater proportion of N1 sleep (i.e. the translational stage of sleep), which is not as restorative[51]
. The following night ‘REM rebound’ occurs, to restore the REM deficit. This suggests there is a homeostatic control mechanism associated with REM sleep[56]
. This REM rebound causes more restorative sleep, but increases the risk of parasomnias (i.e. unwanted experiences, such as movements, behaviours or dreams) occurring; - Medicine use, such as taking stimulants late at night, or sedative medicines during the day, can disturb the sleep–wake cycle.
Summary
This article highlights why it is important to understand a patient’s sleep habits and the role of the pharmacist in helping modify these. However, even when sleep habits are understood, some patients may still require treatment to help modify their sleep. The pharmacological and non-pharmacological options available are described in a second article, to be published in July 2020; in particular, it will outline how cognitive behavioural therapy for insomnia can be utilised by pharmacists in their consultations to better manage patients with sleep disorders.
About the authors
Rebecca Richardson is clinical pharmacist at Garden Park Surgery, Howden, UK; Alistair Paterson is clinical pharmacist at Northumbria, Tyne and Wear NHS Foundation Trust, UK; Adam Rathbone is lecturer in social and clinical pharmacy at Newcastle University, UK; Julia Blagburn is integrated care pharmacist, Newcastle upon Tyne Hospitals NHS Foundation Trust, UK and Andy Husband is professor of clinical pharmacy at the school of pharmacy, Newcastle University, UK. Correspondence to:
adam.rathbone@newcastle.ac.uk
Financial and conflicts of interest disclosure
The authors have no relevant affiliations or financial involvement with any organisation or entity with a financial interest in or financial conflict with the subject matter or materials discussed in this manuscript. No writing assistance was used in the production of this manuscript.
References
[1] World Health Organization. ICD-10: International Statistical Classification of Diseases and Related Health Problems: Tenth revision. Geneva: World Health Organization; 2011
[2] Brass SD, Li C-S & Auerbach S. The underdiagnosis of sleep disorders in patients with multiple sclerosis. J Clin Sleep Med 2014;10(9):1025–1031. doi: 10.5664/jcsm.4044
[3] Araújo T, Jarrin DC, Leanza Y, Vallières A & Morin CM. Qualitative studies of insomnia: Current state of knowledge in the field. Sleep Med Rev 2017;31:58–69. doi: 10.1016/j.smrv.2016.01.003
[4] Coveney C, Williams SJ & Gabe J. Medicalisation, pharmaceuticalisation, or both? Exploring the medical management of sleeplessness as insomnia. Sociol Health Illn 2019;41(2):266–284. doi: 10.1111/1467-9566.12820
[5] Sateia M. International Classification of Sleep Disorders. 3rd edn. Chest 2014;146(5):1387–1394. doi: 10.1378/chest.14-0970
[6] National Institute for Health and Care Excellence. Insomnia. 2020. Available at: https://cks.nice.org.uk/insomnia (accessed June 2020)
[7] Ancoli-Israel S & Roth T. Characteristics of insomnia in the United States: Results of the 1991 National Sleep Foundation Survey. I. Sleep 1999;22 Suppl 2:S347–353. PMID: 10394606
[8] Colrain IM. Sleep and the Brain. Neuropsychol Rev 2011;21(1):1–4. doi: 10.1007/s11065-011-9156-z
[9] Williamson AM & Feyer AM. Moderate sleep deprivation produces impairments in cognitive and motor performance equivalent to legally prescribed levels of alcohol intoxication. Occup Environ Med 2000;57(10):649–655. doi: 10.1136/oem.57.10.649
[10] Herman J, Kafoa B, Wainiqolo I et al. Driver sleepiness and risk of motor vehicle crash injuries: a population-based case control study in Fiji (TRIP 12). Injury 2014;45(3):586–591. doi: 10.1016/j.injury.2013.06.007
[11] Cappuccio FP, Miller MA & Lockley SW. Sleep, Health and Society: From aetiology to public health. Oxford: Oxford Scholarship Online; 2011
[12] Dement W & Kleitman N. Cyclic variations in EEG during sleep and their relation to eye movements, body motility, and dreaming. Clin Neurophysiol 1957;9(4):673–690. doi: 10.1016/0013-4694(57)90088-3
[13] Borbely AA. A two process model of sleep regulation. Human Neurobiol 1982;1(3):195–204. PMID: 7185792
[14] Anderson K. How to treat insomnia and other sleep problems. Newcastle upon Tyne: Regional Sleep Service. 2018. Available at: http://www.neurone.org.uk/wp-content/uploads/Teaching-slides-for-2018-sleep-training-day.pdf (accessed June 2020)
[15] Stepanski EJ & Wyatt JK. Use of sleep hygiene in the treatment of insomnia. Sleep Med Rev 2003;7(3):215–225. doi: 10.1053/smrv.2001.0246
[16] Reading P. Sleep disorders: diagnosis. Clinical Pharmacist 2015;7(9). doi: 10.1211/CP.2015.20069423
[17] Ohayon MM, Carskadon MA, Guilleminault C & Vitiello MV. Meta-analysis of quantitative sleep parameters from childhood to old age in healthy individuals: developing normative sleep values across the human lifespan. Sleep 2004;27(7):1255–1273. doi: 10.1093/sleep/27.7.1255
[18] Shih H-I, Lin C-C, Tu Y-F et al. An increased risk of reversible dementia may occur after zolpidem derivative use in the elderly population: a population-based case-control study. Medicine (Baltimore) 2015;94(17):e809. doi: 10.1097/MD.0000000000000809
[19] Suzuki K, Miyamoto M, Hirata K. Sleep disorders in the elderly: Diagnosis and management. J Gen Fam Med 2017;18(2):61–71. doi: 10.1002/jgf2.27
[20] Substance Abuse and Mental Health Services Administration. Table 3.36, DSM-IV to DSM-5 insomnia disorder comparison, impact of the DSM-IV to DSM-5 changes on the National Survey on Drug Use and Health. Available at: https://www.ncbi.nlm.nih.gov/books/NBK519704/table/ch3.t36/ (accessed June 2020)
[21] Baglioni C & Riemann D. Is chronic insomnia a precursor to major depression? Epidemiological and biological findings. Curr Psychiatry Rep 2012;14(5):511–518. doi: 10.1007/s11920-012-0308-5
[22] Spielman AJ, Caruso LS & Glovinsky PB. A Behavioral Perspective on Insomnia Treatment. Psychiatr Clin North Am 1987;10(4):541–553. PMID: 3332317
[23] Williams J, Roth A, Vatthauer K & McCrae CS. Cognitive behavioral treatment of insomnia. Chest 2013;143(2):554–565. doi: 10.1378/chest.12-0731
[24] Morin CM, Blais F & Savard J. Are changes in beliefs and attitudes about sleep related to sleep improvements in the treatment of insomnia? Behav Res Ther 2002;40(7):741–752. doi: 10.1016/s0005-7967(01)00055-9
[25] Ebben MR & Spielman AJ. A practical guide to insomnia assessmnet. In: Pagel J & Pandi-Perumal S. Primary Care Sleep Medicine. New York: Springer; 2014
[26] Bixler EO, Kales A, Soldatos CR, Kales JD & Healey S. Prevalence of sleep disorders in the Los Angeles metropolitan area. Am J Psychiatry 1979;136(10):1257–1262. doi: 10.1176/ajp.136.10.1257
[27] Klink M & Quan SF. Prevalence of reported sleep disturbances in a general adult population and their relationship to obstructive airways diseases. Chest 1987;91(4):540–546. doi: 10.1378/chest.91.4.540
[28] Ohayon MM. Epidemiology of insomnia: what we know and what we still need to learn. Sleep Med Rev 2002;6(2):97–111. doi: 10.1053/smrv.2002.0186
[29] Lindam A, Kendall BJ, Thrift AP et al. Symptoms of Obstructive Sleep Apnea, Gastroesophageal Reflux and the Risk of Barrett’s Esophagus in a Population-Based Case-Control Study. PLoS One 2015;10(6):e0129836. doi: 10.1371/journal.pone.0129836
[30] Morphy H, Dunn KM, Lewis M, Boardman HF & Croft PR. Epidemiology of insomnia: a longitudinal study in a UK population. Sleep 2007;30(3):274–280. PMID: 17425223
[31] Terzano MG, Parrino L, Cirignotta F et al. Studio Morfeo: insomnia in primary care, a survey conducted on the Italian population. Sleep Med 2004;5(1):67–75. doi: 10.1016/j.sleep.2003.09.006
[32] Wilson S, Anderson K, Baldwin D et al. British Association for Psychopharmacology consensus statement on evidence-based treatment of insomnia, parasomnias and circadian rhythm disorders: An update. J Psychopharmacol 2019;33(8):923–947. doi: 10.1177/0269881119855343
[33] Mellinger GD, Balter MB & Uhlenhuth EH. Insomnia and its treatment. Prevalence and correlates. Arch Gen Psychiatry 1985;42(3):225–232. doi: 10.1001/archpsyc.1985.01790260019002
[34] Ohayon MM & Reynolds CF, 3rd. Epidemiological and clinical relevance of insomnia diagnosis algorithms according to the DSM-IV and the International Classification of Sleep Disorders (ICSD). Sleep Med 2009;10(9):952–960. doi: 10.1016/j.sleep.2009.07.008
[35] Goessaert A-S, Krott L, Walle JV & Everaert K. Exploring nocturia: gender, age, and causes. Neurourol and Urodyn 2015;34(6):561–565. doi: 10.1002/nau.22638
[36] Gislason T & Almqvist M. Somatic diseases and sleep complaints. an epidemiological study of 3,201 Swedish men. Acta Medica Scandinavica 1987;221(5):475–481. PMID: 3496735
[37] Narisawa H. Anxiety and its related factors at bedtime are associated with difficulty in falling asleep. Tohoku J Exp Med 2013;231(1):37–43. doi: 10.1620/tjem.231.37
[38] Ferri R, Lanuzza B, Cosentino FII et al. A single question for the rapid screening of restless legs syndrome in the neurological clinical practice. Eur J Neurol 2007;14(9):1016–1021. doi: 10.1111/j.1468-1331.2007.01862.x
[39] British Snoring & Sleep Apnoea Association. STOPBang questionnaire. 2014. Available at: https://britishsnoring.co.uk/stop_bang_questionnaire.php (accessed June 2020)
[40] Joint Formulary Committee. British National Formulary. London: BMJ Group and Pharmaceutical Press; 2020
[41] Taylor DM, Barnes TR, Young AH. The Maudsley Prescribing Guidelines in Psychiatry. New Jersey: John Wiley & Sons; 2018
[42] Rosen RC, Rosekind M, Rosevear C, Cole WE & Dement WC. Physician Education in Sleep and Sleep Disorders: A National Survey of U.S. Medical Schools. Sleep 1993;16(3):249–254. doi: 10.1093/sleep/16.3.249
[43] Omachi TA. Measures of sleep in rheumatologic diseases: Epworth sleepiness scale (ESS), functional outcome of sleep questionnaire (FOSQ), insomnia severity index (ISI), and Pittsburgh sleep quality index (PSQI). Arthritis Care Res (Hoboken) 2011;63(Suppl 11):S287–S296. doi: 10.1002/acr.20544
[44] Lawrence G & Muza R. Assessing the sleeping habits of patients in a sleep disorder centre: a review of sleep diary accuracy. J Thorac Dis 2018;10(Suppl 1):S177–S183. doi: 10.21037/jtd.2017.12.127
[45] Wohlgemuth WK, Edinger JD, Fins AI & Sullivan RJ, Jr. How many nights are enough? The short-term stability of sleep parameters in elderly insomniacs and normal sleepers. Psychophysiology 1999;36(2):233–244. PMID: 10194970
[46] Crispim CA, Zimberg IZ, dos Reis BG et al. Relationship between food intake and sleep pattern in healthy individuals. J Clin Sleep Med 2011;7(6):659–664. doi: 10.5664/jcsm.1476
[47] Lira FS, Pimentel GD, Santos RV et al. Exercise training improves sleep pattern and metabolic profile in elderly people in a time-dependent manner. Lipids Health Dis 2011;10:113. doi: 10.1186/1476-511X-10-113
[48] Chennaoui M, Arnal PJ, Sauvet F & Leger D. Sleep and exercise: a reciprocal issue? Sleep Med Rev 2015;20:59–72. doi: 10.1016/j.smrv.2014.06.008
[49] Montgomery P & Dennis J. Physical exercise for sleep problems in adults aged 60+. Cochrane Database Syst Rev 2002(4):CD003404. doi: 10.1002/14651858.CD003404
[50] Passos GS, Poyares D, Santana MG et al. Effects of moderate aerobic exercise training on chronic primary insomnia. Sleep Med 2011;12(10):1018–1027. doi: 10.1016/j.sleep.2011.02.007
[51] Haario P, Rahkonen O, Laaksonen M et al. Bidirectional associations between insomnia symptoms and unhealthy behaviours. J Sleep Res 2013;22(1):89–95. doi: 10.1111/j.1365-2869.2012.01043.x
[52] Jaehne A, Unbehaun T, Feige B et al. How smoking affects sleep: a polysomnographical analysis. Sleep Med 2012;13(10):1286–1292. doi: 10.1016/j.sleep.2012.06.026
[53] Fucito LM, Redeker NS, Ball SA et al. Integrating a behavioural sleep intervention into smoking cessation treatment for smokers with insomnia: A Randomised Pilot Study. J Smok Cess 2014;9(1):31–38. doi: 10.1017/jsc.2013.19
[54] Drake C, Roehrs T, Shambroom J & Roth T. Caffeine effects on sleep taken 0, 3, or 6 hours before going to bed. J Clin Sleep Med 2013;9(11):1195–1200. doi: 10.5664/jcsm.3170
[55] Stein MD & Friedmann PD. Disturbed sleep and its relationship to alcohol use. Subst Abus 2005;26(1):1–13. doi: 10.1300/j465v26n01_01
[56] Roehrs T & Roth T. Sleep, sleepiness, and alcohol use. Alcohol Res Health 2001;25(2):101–119. PMID: 11584549