MAG / The Pharmaceutical Journal
Ciclosporin (cyclosporine), an anti-inflammatory treatment, is also being increasingly used to manage various forms of dry eye, particularly in patients for whom artificial tears and other over-the-counter treatments are ineffective. It can help patients to produce more, healthier tears (rather than replacing them, as with artificial tears). A common side effect of ciclosporin is a burning sensation in the eyes.
Advise patients using ciclosporin that the effect is not immediate. Patients using ciclosporin must use it as directed for at least 90 days before any effect is likely to be noticed.
Non-steroidal anti-inflammatory drops can also help to relieve the inflammation associated with dry eye disease, and may be used alone or in combination with ciclosporin.
Corticosteroid eye drops and ointments may also be prescribed in severe cases of dry eye disease. However, these medicines have common side effects, including cataracts and raising the pressure within the eye. Patients using these medicines should be monitored regularly at a specialist eye clinic.
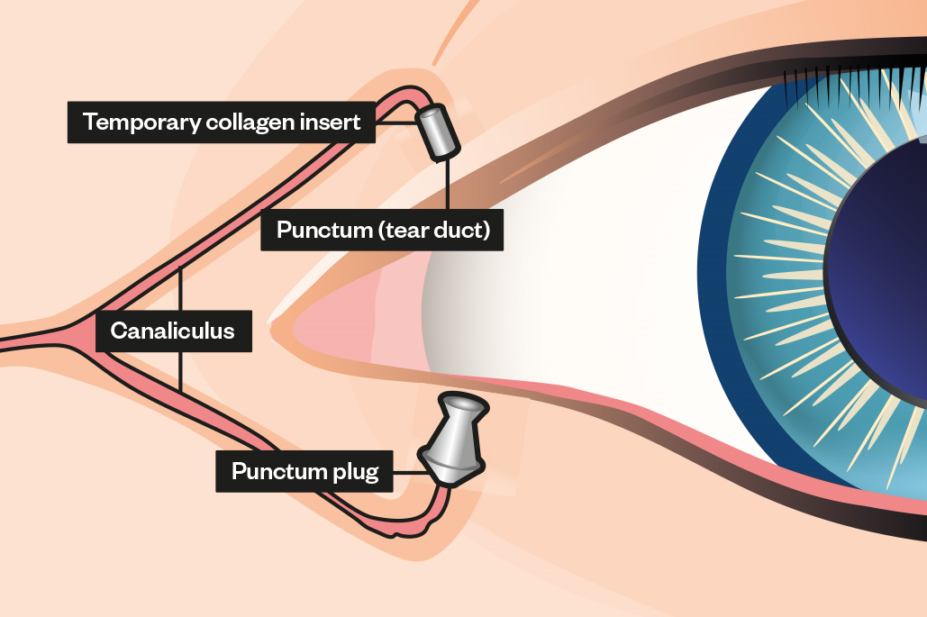
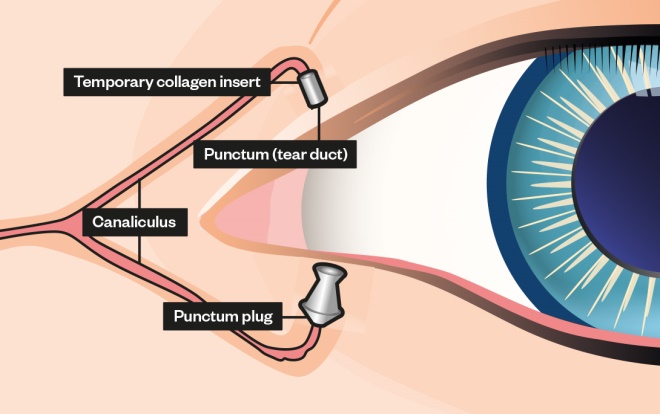
Punctal plugs are tiny devices inserted into tear ducts prevent tears from draining, increasing the eye’s tear film and surface moisture[1]
. The two general types of tear duct plugs are:
- Soluble: made of materials such as collagen that the body absorbs;
- Semi-permanent: made of long-lasting materials such as silicone.
Punctal plugs
Source: MAG / The Pharmaceutical Journal
References
[1] Not A Dry Eye. Symptoms. Available at: www.notadryeye.org/all-about-dry-eye-syndrome/symptoms-of-dry-eye-syndrome-and-related-conditions/ (accessed 8 July 2017)



