
DR P. MARAZZI/SCIENCE PHOTO LIBRARY
Usually described as a ‘psychotic illness’, schizophrenia is a chronic and debilitating neurodevelopmental disorder that affects thinking, emotions and behaviour. It typically develops in adolescence or early adulthood, with a lifetime prevalence of around 1% of the population worldwide[1]
. Psychosis can occur in people with schizoÂphrenia, as well as in conditions such as dementia, stroke, bipolar disorder and depression. It can cause people to perceive or interpret things differently from those around them. Psychosis can also caused by medicines, including anti-Parkinson’s drugs, corticosteroids and illicit sÂubstances.
Historically, many patients with diagnosed schizophrenia will have spent some time under inpatient psychiatric care; however, since the 1990s, there has been a shift in policy to reduce institutionalisation and increase the amount of people who are able to be treated in their own homes, even in periods of acute illness[2]
.
People with schizophrenia are consistently shown to have a significantly shorter life expectancy (by an average of 15–20 years) than that observed in the general population[3],[4]
. Although the cause of this is multifactorial, including higher rates of alcohol and substance misuse, suicide and smoking, further deaths from cardiovascular disease (CVD) is thought to play a significant role.
Some medicines used to manage schizophrenia may cause side effects, such as weight gain and diabetes. These not only add to the risk of CVD, but cause significant morbidity and can affect whether patients will continue taking medicines in the longer term. Stigma and discrimination are commonly experienced by those who are diagnosed with schizophrenia and can affect all aspects of life, including reducing access to appropriate healthcare.
Pharmacists can ensure patients are safely treated and monitored, and help patients access appropriate advice and treatment to help them stay healthier for longer (see Box 1).
Box 1: Role of the pharmacist
As a pharmacist, it is important to:
- Know the local mental health trust — be aware of what they provide, their contact details, crisis line, medicines helpline and pharmacy supply details;
- Know what else is available in the local area (e.g. support groups, carer groups and community centres for smoking cessation, exercise, weight loss and healthy eating);
- Ensure patient records are up to date with medicines provided elsewhere (e.g. depots and clozapine) and ensure that all medicines are checked for interactions;
- Check that patients are being monitored appropriately and encourage them to be involved in their treatment plan;
- Communicate with other healthcare professionals when there are concerns;
- Identify major adverse effects and refer the patients appropriately to their team or GP.
Aetiology
Schizophrenia is thought to have multifactorial aetiology, with genetic, biological and environmental factors all contributing[5]
. The high degree of heritability has led to renewed interest in genetic variants linked with schizophrenia[6]
. Although no single gene has been implicated, several variants have been linked to the development of schizophrenia[6],[7]
. Genetics alone cannot account for its development and there is thought to be a link with environmental stressors, particularly those that occur early in life[8]
. People who have risk factors, such as a family history or those exhibiting symptoms of psychosis, have a substantial risk of developing schizophrenia[9]
.
Research examining potential biomarkers for schizophrenia, to establish which are relevant, reproducible and useful in individualising and developing treatments, is ongoing[10],[11]
.
The use of certain substances, such as cannabis, cocaine, lysergic acid diethylamide (LSD) or amphetamines, can trigger symptoms in susceptible individuals. Patients presenting with psychotic symptoms and are distressed, or have a decline in function, should be referred to a specialist psychiatric service for assessment.
Diagnosis and symptoms
Schizophrenia normally involves periods of acute illness where symptoms can be intense and distressing, followed by more settled periods where symptoms can subside[12]
. Patients exhibit substantial cognitive deficit that can significantly impair function, including problems in speed of processing information, attention/vigilance, working memory, verbal learning, visual learning, reasoning and problem solving, and social cognition[13],[14]
,[15]
. Cognitive effects may impact the patient’s ability to remember to take medicine or attend appointments, therefore, support should be given to help patients manage these difficulties.
Diagnosis usually involves a detailed assessment of the patient, including their current symptoms and full history, often referred to as a ‘mental state examination’[16]
. Psychiatrists and other members of the multidisciplinary team may undertake multiple assessments over a period of around a month, but these could take longer depending on the available history of the patient and their presentation. Younger adolescent patients in particular can take many months to formally diagnose. These assessments help to differentiate schizophrenia from other disorders, such as psychosis related to an acute illness or substance. Diagnostic tests and tools, such as the International Classification of Diseases (ICD-10), are normally only carried out if other organic causes of psychosis are suspected and to aid decisions about the monitoring of treatment (see Box 2)[17]
.
Box 2: Using the International Classification of Diseases (ICD-10) to aid diagnosis of schizophrenia
Schizophrenia is diagnosed according to International Classification of Diseases (ICD-10) where a mixture of symptoms — including at least one symptom from the first three below — are present most of the time for at least one month:
- Hallucinations;
- Delusions;
- Specific thought disorder (e.g. thought insertions or withdrawals, or thought broadcasting);
- At least one symptom from two of the following:
- Catatonic symptoms (e.g. reduced speech and movement, unresponsiveness, agitation or confusion);
- Negative symptoms (e.g. social withdrawal, isolation, apathy or self neglect);
- Thought disorder (e.g. breaks in train of thought, neologisms [i.e. a newly coined word or expression]).
The same tool can be used to further categorise the condition into subtypes, based on the type and predominance of symptoms that the patient is presenting with[1],[9]
. For example:
- Paranoid schizophrenia;
- Hebephrenic schizophrenia, also called disorganised schizophrenia, which involves predominant changes in expressed mood or emotion with inappropriate responses to situations and disorganised thought and speech. Negative symptoms often predominate with fleeting delusions and hallucinations;
- Catatonic schizophrenia, which involves significant changes in movement, from reduced movement to hyperactivity and agitation;
- Unspecified.
Positive symptoms indicative of schizophrenia are characterised by something being added that is not normally there (e.g. hallucinations, delusions and specific thought disorder), whereas negative symptoms are those that involve a reduction in functioning, such as apathy, self-neglect, withdrawal and lack of drive[1],[15]
.
Both positive and negative symptoms can be exaggerated during periods of acute psychosis and, as patients can put themselves at risk and end up in crisis, it is important that they are evaluated and treatment is started promptly[16]
. Patients should be helped to self-identify the signs of their psychosis and educated about what to do in a crisis; however, it is also important that other members of their care team, friends or family are able to help identify when someone may be becoming mentally unwell.
Many people with schizophrenia will experience a prodromal period before a diagnosis is made[18],[19]
. This may be a period where negative symptoms dominate and patients may become isolated and withdrawn. This may be confused with depression — may be confused with depression — particularly in younger people — although depression and anxiety disorders themselves may also be prominent and worthy of treatment[16]
. It is hoped that future treatments aimed at earlier stages of schizophrenia could help reduce progression of the disease.
People who experience more frequent or longer episodes of untreated schizophrenia can become irreversibly impaired, experiencing long-term problems with cognition and residual symptoms; therefore, it is paramount that patients remain on effective treatments and in remission for as long as possible[20]
.
Symptoms that overlap with other psychotic disorders
In bipolar disorder, people experience periods of elevated mood (mania or hypomania) and periods of depression, and symptoms indicative of schizophrenia (e.g. delusions and hallucinations) can develop. In schizoaffective disorder, patients experience psychotic symptoms similar to schizophrenia, as well as the changes in affect or mood seen in bipolar disorder.
Many symptoms overlap the boundaries of traditional diagnoses and many of the treatments are the same, regardless of the diagnosis, as they aim to manage the symptoms. While this article focuses on schizophrenia, much of the information regarding antipsychotics would be applicable to other conditions in which they are used.
Children and younger people
Schizophrenia is rarely diagnosed in children, although evidence suggests that early signs may develop in this population group. It is usual for the first few episodes of psychosis to occur in teenagers and early adulthood, with peaks of diagnoses in the mid-20s. After this age, the rate of new diagnoses starts to fall, with few occurring after people reach their late-40s[21]
.
As diagnoses in children and younger people can be more complicated, particularly if they are in the early stages of such an illness, it is important that they are appropriately and quickly referred to a child and adolescent mental health service or an early intervention service. These patients may need longer periods of observation before an accurate diagnosis can be made[22]
.
Pharmacological treatment
Antipsychotics are the main treatment strategy for schizophrenia and are used to treat both the acute positive and negative symptoms and maintain remission (see Figure). However, it is common for patients to have significant adverse effects and fail to respond to treatment, resulting in many patients stopping treatment and being inadequately treated[23],[24]
.
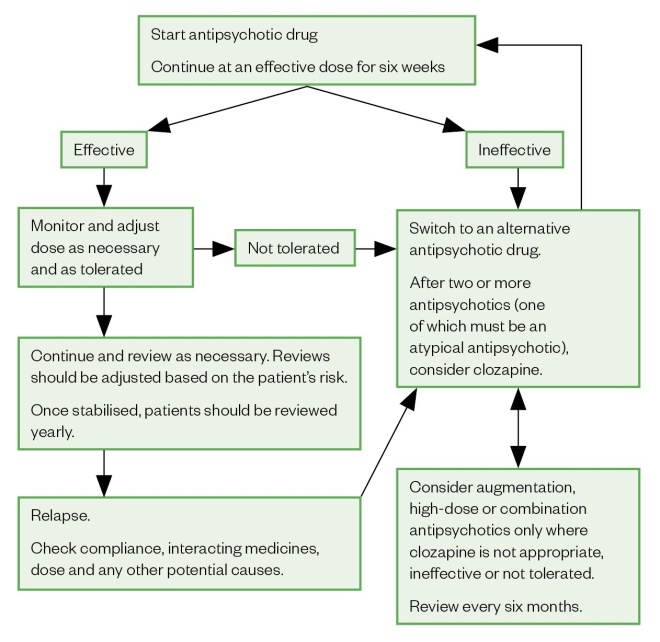
Source: Algorithm for the treatment of schizophrenia
Because overactivity in the dopaminergic pathways is thought to be involved in the development of schizophrenia, antipsychotics mainly target the dopamine D2 receptor. As well as being involved in mood and psychosis, dopaminergic neurones have many other functions that can be affected by blocking the D2 receptors, such as movement and the regulation of prolactin, leading to drug-induced movement disorders (extrapyramidal side effects [EPSEs]) and hyperprolactinaemia. Hyperprolactinaemia often leads to menstrual changes in women, including amenorrhea, reduced fertility, sexual dysfunction and breast changes (enlargement and lactation).
Antipsychotics are often referred to as typical or atypical, and first-, second- or third-generation. Properties they display can overlap and there is no evidence of any significant benefit of one class over the other (see Table 1)[25]
. However, there is wide variance in the side effects they can cause[15]
. It is, therefore, essential to understand what side effects are most important to the patient and to know which antipsychotic drug is more likely to cause these adverse effects. As adverse effects are frequently the cause of nonadherence, it is important to identify and deal with any potential problems to try and prevent patients from stopping medicine without support[20]
.
| Typical or first-generation antipsychotic drugs | Atypical or second and third-generation antipsychotics |
|---|---|
|
|
| Source: British National Formulary [25] | |
Many of the newer antipsychotics are serotonin 5-hydroxytryptamine receptor 2A (5HT2A) antagonists and affect dopamine receptors to a lesser and varying extent[26]
. The action at the 5HT2A receptors is thought to be one of the reasons why the atypical antipsychotics have less propensity to cause EPSEs and also may be involved in antipsychotic activity[26]
. Antipsychotics can also affect a range of other receptors that are often involved in the manifestation of adverse effects (see Table 2)[26],[27],[28]
,[29]
.
| Receptor | Main effect of antipsychotic | Adverse effects | Antipsychotics most likely to cause this effect | Antipsychotics least likely to cause this effect |
|---|---|---|---|---|
| Alpha 1 adrenergic receptor | Antagonist | Dizziness Postural hypotension Sedation | Clozapine Chlorpromazine Risperidone | Amisulpride Aripiprazole |
| Alpha 2 adrenergic receptor | Antagonist | Hypertension | Risperidone | Amisulpride Aripiprazole |
| Dopamine D2 | Antagonist | Extrapyramidal side effects Hyperprolactinaemia | Amisulpride First-generation antipsychotics Risperidone | Aripiprazole* Clozapine Olanzapine Quetiapine |
| Histamine H1 receptor | Antagonist | Sedation Weight gain | Clozapine Chlorpromazine Olanzapine Quetiapine | Amisulpride Aripiprazole Lurasidone |
| Muscarinic acetylcholine receptor | Antagonist | Blurred vision Confusion Constipation Dry mouth Urinary retention | Chlorpromazine Clozapine | Amisulpride Aripiprazole Lurasidone |
| 5-hydroxytryptamine 2A receptor | Antagonist | Sedation | Clozapine Lurasidone Olanzapine Risperidone | Amisulpride |
| * Aripiprazole is a partial agonist at the D2 receptor Source: The Maudsley Prescribing Guidelines in Psychiatry
| ||||
Glutamate hyperactivity and altered immune function have been shown to have a potential role in the development of schizophrenia[30],[31],[32]
. Ketamine, an N-methyl-D-aspartate (NMDA) antagonist, has been shown to elicit psychotic symptoms similar to those seen in schizophrenia[33],[34]
. Antipsychotics may already be having an effect on these systems despite not being their major targets[35]
. Future treatments that target these systems and other neurotransmitters are in development and, if they prove to be successful, could provide the first truly different treatment option for people with schizophrenia since clozapine was reintroduced in 1990[30],[32],[36]
. Deficits in cognitive function are not well treated by current antipsychotics and, while cognitive remediation has been shown to be a promising option, antipsychotics that improve cognition are also an important area for research[37],[38]
.
All major guidelines, such as the National Institute for Health and Care Excellence (NICE), the Scottish Intercollegiate Network (SIGN) and the British Association of Psychopharmacology (BAP), base the choice of initial treatment on the patient’s preference, taking into account the range of side effects, the patient’s physical health and other medicines, and any existing comorbidities[1],[15],[39]
. When choosing subsequent antipsychotic drugs, it is important to consider previous treatments and the reasons they were stopped or changed, as this can help guide further choices by exploring options with different tolerability profiles.
Antipsychotic drug dosages are generally used in line with the British National Formulary, with low doses used in young people (aged <18 years) and older people (>65 years), and patients with comorbidities. Male patients often require higher doses, and smokers may need doses towards the upper range for medicines, such as haloperidol, olanzapine and clozapine as the metabolism of these drugs is induced by smoking[40],[41],[42]
. Doses at a higher strength can be used initially, particularly in an acutely unwell patient, but these can be adjusted once symptoms are under control.
With the exception of clozapine, most antipsychotics are able to be used cautiously in patients with other comorbidities and medicines[25]
,[27],[28]
. Caution is required when prescribing antipsychotics to patients with epilepsy, CVD, severe respiratory disease, blood dyscrasias or liver disease, with dose adjustments and/or closer monitoring normally required.
Because of the risk of agranulocytosis with clozapine, it is contraindicated with other high-risk medicines (such as carbimazole, carbamazepine or disease-modifying antirheumatic drugs) that should generally be avoided or used with very close monitoring[25]
. Clozapine is also linked with higher risk of epilepsy, bowel obstruction, sedation and certain cardiovascular changes; as such, it has several specific contraindications[25]
. However, clozapine is specifically licensed for the treatment of psychosis related to Parkinson’s disease and also shows particular efficacy in reducing the risk of suicide in those with schizophrenia[43],[44],[45]
.
Not all antipsychotics are licensed for use in children and formulation availability may vary, which may make some antipsychotics more suitable for use in younger patients. In addition, the following medicines are licensed for schizophrenia from specific ages: haloperidol at 12 years, sulpiride at 14 years, aripiprazole at 15 years and clozapine at 16 years. Where possible, antipsychotics with a licence in children should be used, but in some cases there may be more suitable antipsychotics available that are not specifically licensed in children.
Long-acting antipsychotics
Patient compliance or concordance with prescribed medicines is a challenge across all patient groups, including those with chronic and severe physical and mental health illnesses, such as schizophrenia. Non-concordance is associated with relapse and this can occur rapidly after medicine is discontinued.
Some antipsychotics are formulated as long-acting injections (LAIs), and commonly used for patients where oral formulations are not suitable, such as those who struggle to remember to take medicines every day or where LAIs are more convenient for the patient. There are currently four atypical (olanzapine, risperidone, paliperidone, aripiprazole) and four typical (haloperidol, zuclopenthixol, flupentixol and fluphenazine) antipsychotic LAIs available in the UK, although the typical depot fluphenazine was discontinued at the end of 2018. LAIs are administered between once weekly and every three months and there is little difference in effectiveness of the antipsychotic LAIs. Side effects are similar to their oral equivalents. The main differences are related to their pharmaceutical formulations and kinetics. Long-acting antipsychotics are often administered by community mental health teams and it is important to ensure they are recorded on patient medication records.
Switching antipsychotics
It is important to consider medicines that have similar side effects when switching antipsychotics, such as those that are very sedative or cause EPSEs, because these effects can be additive. LAIs remain in the body for long periods after the medicines are stopped, which can also lead to additive adverse effects — particularly when switching to an oral antipsychotic. In many cases, there will be a period of cross-tapering to avoid leaving the patient without medicine, that could lead to relapse. Therefore, patients may need extra support to help them make a smooth transition from one antipsychotic to another.
Treatment-resistant schizophrenia
This is generally defined as the failure to respond to or tolerate two sequential antipsychotics, where one is a second-generation antipsychotic or an atypical antipsychotic given at an adequate dose for an adequate length of treatment[1],[15],[39]
.The definition of two antipsychotics is somewhat arbitrary; however, the aim is to encourage the use of clozapine in patients who are not responding well to initial treatments, whereas in practice the use of clozapine may be delayed. Clozapine is uniquely indicated in treatment-resistant schizophrenia[39]
.
While there is no clear choice for initial treatment of schizophrenia, NICE, SIGN and BAP guidelines all recommend that clozapine should be offered to patients who fail to respond to or tolerate two antipsychotic drugs, one of which should be a second-generation antipsychotic[1],[15],[39]
. Clozapine is the only antipsychotic with consistent evidence of superiority in comparison to other antipsychotics. However, its use is restricted owing to the risk of serious haematological adverse effects[46],[47],[48]
.
Where there is inadequate response to clozapine alone, combinations of medicines, such as mood stabilisers, other antipsychotics and antidepressants, can be used to augment clozapine or as an alternative to clozapine[27],[28],[49]
.
Where clozapine has failed to be effective or is not tolerated, patients can be tried on doses of single or combined antipsychotics that exceed the licensed maximum doses. This is often referred to as high-dose antipsychotic therapy and, to ensure the patient is treated safely, extra monitoring is normally undertaken. It is important to consider that the evidence for the effectiveness of high dose antipsychotic therapy is limited and there has been a shift away from using such high doses.
In acute episodes, other medicines — particularly benzodiazepines and hypnotics — may be used to manage symptoms of agitation and aggression, and these should ideally be reviewed regularly and stopped once the patient is no longer acutely unwell.
Psychological treatment
Some patients may find psychological treatments helpful in understanding how to cope with their illness[50]
. Psychological treatment should ideally be used alongside medicine, and some patients may need to start the treatment when they are not acutely unwell in order to gain the most benefit.
Cognitive behavioural therapy (CBT) is a commonly used evidence-based method that aims to challenge unhelpful thinking patterns and help patients understand their symptoms and illness. Patients are normally offered a course of one-to-one sessions with a healthcare professional, which typically involve practical activities aimed at changing the way the person manages their problems[51]
. The evidence supports the use of CBT in addition to antipsychotics where it can be beneficial, but it is not suitable as a standalone treatment in schizophrenia because it does not prevent relapse[52],[53]
.
Family therapy can help families come together to deal with a diagnosis by looking at causes of stress and finding ways to address these in a positive way. The number and frequency of the sessions can vary[54]
. Arts therapies, such as music, painting/drawing or dance offer different ways for people to express and work through their problems[55]
.
Cognitive remediation, a therapy developed in the 2000s, is defined as the therapeutic process of increasing or improving an individual’s capacity to process and use incoming information, allowing increased functioning in everyday life. It has shown promise in helping patients overcome cognitive deficits[56],[57]
.
There are often support groups, carer groups and charities available locally that can provide advice and support for both patients and families living with schizophrenia. Patients may need support with other activities, such as with housing, benefits, education, training and work.
Care programme approach
Many patients with schizophrenia with ongoing needs are managed under the ‘Care programme approach’ (CPA), where they have one person who co-ordinates their care and is responsible for ensuring they have an up-to-date care plan[58]
. It is a multidisciplinary approach that aims to bring together and address a person’s healthcare and social care needs, and is managed by the patient’s community mental health team. Not all patients with schizophrenia will require a CPA because it is generally used for those with more complex needs.
Monitoring and follow up
It is important to carry out baseline monitoring because it is difficult to ascertain the effects of medicines. Patients should be reviewed frequently for both clinical effects and adverse effects. Formal rating scales, such as the Glasgow Antipsychotic Side-effect Scale (GASS) or GASS-Clozapine, can be used to help patients rate how they perceive their side effects and can be used to monitor changes[59]
. The patient should be evaluated for signs of psychotic symptoms, including any changes as some patients will have some residual symptoms. A full assessment of the carer’s views is required as patients may not always fully articulate their symptoms.
The patient’s ability to function at the pre-illness level should be the goal for successful treatment. Some patients with chronic illness may remain symptomatic; therefore, their success may be graded on their ability to successfully manage everyday activities[60]
. They should also be free from any distressing symptoms or adverse effects. Patients who remain symptomatic should be referred to their mental health team to see if their treatment can be optimised.
Antipsychotics have been associated with metabolic syndrome, where the highest risk is with olanzapine, and the lowest risk is generally clozapine with aripiprazole[27]
. Antipsychotics have also been associated with weight gain (particularly clozapine and olanzapine), diabetes and high cholesterol[61]
. Weight is monitored weekly for the first 6 weeks and then with cholesterol and glucose levels at 12 weeks and yearly[49]
.
Weight gain tends to start early on in treatment with antipsychotics, which is not thought to be dose-related and tends to slow down or plateau with continued therapy[62],[63]
. It is a common side effect and can interfere with a patient’s life to the extent that they no longer want to take medicine[43],[64]
. Therefore, it should be highlighted that stopping antipsychotics does not reverse weight gain. Treatments are available, such as metformin, aripiprazole and orlistat, to help patients with weight loss[61]
. It is important to discuss this with patients before they start treatment so that it can be recognised, offering the patient help to reduce the weight gain.
It is advised that healthcare professionals monitor both glycosylated haemoglobin and glucose levels to ensure that both acute and chronic changes in glucose are identified, as the rise in blood glucose can be quite sudden. Increases observed in these tests should prompt an evaluation of the patient’s diet and lifestyle, and referral to dieticians or weight management programmes can be considered. Annual liver and kidney function tests are also required.
Monitoring clozapine
This can cause potentially fatal myocarditis and cardiomyopathy that tend to occur early on in treatment. Symptoms include fever, chest pain, flu-like symptoms, tachycardia and shortness of breath, which should prompt a referral for further examination.
Clozapine is only started by consultant psychiatrists who, along with the pharmacy team, dispense the clozapine. They must be registered with a clozapine-monitoring service as this medicine requires specific blood monitoring owing to the risk of neutropenia and agranulocytosis. The monitoring frequency is dependent on the length of time the patient takes clozapine (see Table 3)[43]
. Patient’s full blood counts (FBC) are taken, but the results of importance are the white blood cell (WBC) count, including neutrophil and eosinophil counts separately, and platelet counts, which are recorded in the clozapine-monitoring service[43]
.
| Length of time patient is taking clozapine | Frequency of testing |
|---|---|
| 0–18 weeks | Weekly |
| 18–52 weeks | Fortnightly |
| >52 weeks | Four-weekly |
| Abnormal results | |
| Amber result (white blood cell [WBC] count 3.0×109–3.5×109/L and/or neutrophil count 1.5×109–2.0×109/L) | Twice-weekly until green (until WBC count >3.5×109 and neutrophil count >2.0×109) |
| Red result (WBC count <3.0×109/L and/or or neutrophil count <1.5×109/L) | Daily until green |
Source: eMC[43] | |
The patient’s eosinophil levels are monitored in case they develop eosinophilia (cell counts above 3.0×109/L), in which case clozapine is discontinued[43]
. Clozapine should also be discontinued if platelet counts fall below 50×109/L (normal 150–400 x109/L)[43]
, but can be continued for patients who experience an amber result (WBC count 3.0×109–3.5×109/L and/or neutrophil count 1.5×109–2.0×109/L). However, clozapine must be stopped immediately if a patient experiences a red result (WBC count <3.0×109/L and/or or neutrophil count <1.5×109/L). Daily FBCs must be taken with close monitoring of the patient until the FBC normalises.
Patients are required to adhere to blood test monitoring to be able to continue with clozapine, as well as to monitor for serious haematological adverse effects. If they do not consent to having regular blood tests, they will be unable to start or continue clozapine. During titration, blood pressure, temperature and pulse are monitored before and after each dose until the dose is stable[43],[65]
. Once stable, monitoring of these occurs when the patient attends for their blood test.
Testing of the patient’s clozapine plasma level is not mandatory, but it is recommended to be completed annually or when clinically indicated[65]
. Reasons for undertaking this test include:
- Non-response to current dose;
- Concerns about non-concordance;
- Change in smoking habits;
- Concerns about toxicity.
Clozapine plasma levels are taken as a trough level and at least 12 hours post-dose where clozapine plasma levels are indicated. Where a patient is taking twice-daily clozapine they should not take their morning dose until the level has been taken[65]
. Both clozapine and norclozapine (a major metabolite of clozapine) levels are monitored and the ratio between the two can provide information about compliance and potential saturation of the metabolic pathway[27],[28]
.
Where clozapine is missed for two or more consecutive days, it will generally need to be retitrated. Consideration needs to be given to patients who struggle to take clozapine consistently owing to this need to retitrate, because it can lead to the patient being undertreated for long periods of time. Poor adherence was seen as the greatest reason why patients were not offered clozapine[66]
.
Unless there is a serious adverse effect, such as a red result or bowel obstruction where clozapine should be stopped immediately, it is usual to slowly reduce and stop clozapine to reduce the risk of a rebound psychosis (see Table 4)[67],[68]
.
| Adverse effect | Advice and management for the patient |
|---|---|
| Agranulocytosis | Any patient with an unexplained fever, sore throat or illness should be advised to see their GP. Advise patients to attend their regular blood test monitoring (this may vary from patient to patient). |
| Constipation | Offer healthy dietary advice and advise an increase of fluid intake and exercise. If there are any signs of serious constipation or it is not improving with simple measures, advise the patient to speak to their GP because constipation can lead to bowel obstruction or paralytic ileus, which can be fatal. Initial treatment should be with a stimulant laxative, but combination of stimulant and osmotic laxative or stool softeners are often required. |
| Diabetes | This can occur in relation to weight gain but also on its own in the absence of weight gain. It can occur early on in treatment where plasma glucose should be measured, rather than haemoglobin A1c. It is important to provide advice on healthy eating and exercise and refer the patient to their team for further monitoring and advice. |
| Hypersalivation | If this is a concern for the patient, advise them to speak to their prescriber as medicine (e.g. hyoscine hydrobromide) can be given. |
| Postural hypotension | Advise patients to take care standing up if feeling dizzy and speak to their doctor if they are concerned. |
| Sedation | It is possible to adjust twice-daily dosing so that a greater proportion is taken at night time. Advise the patient to speak to their prescriber. |
| Seizures | Any patient who experiences seizures needs to be assessed by a doctor. Seizures are more common at high clozapine levels, but can occur at lower levels in susceptible individuals. |
| Tachycardia | Rapid heartbeat is common at the start of treatment, but tends to improve with continued use. Patients should speak to their doctor or psychiatry team to have their heart rate checked. In rare cases, it can be a sign of more serious conditions, such as myocarditis (inflammation of the heart muscle). |
| Weight gain | This is common with clozapine and often occurs early on in treatment. It is important to provide advice on healthy eating and exercise and refer the patient to their team for further monitoring and advice. |
| Source: The Maudsley Prescribing Guidelines in Psychiatry [27] ; Psychotropic Drug Directory [28] ; eMC[43] | |
Discontinuing treatment
For the majority of patients, treatment with antipsychotics will need to be lifelong as discontinuation leads to a high risk of relapse — the risk increases quickly once the medicine is removed from the patient’s system[69]
. Withdrawal of longer-acting medicine, or discontinuations that are carried out very slowly, tend to delay relapse because it takes some time for the medicines to be removed.
Patients may want to discontinue medicine for several reasons, including side effects, problems with monitoring, or poor understanding of their illness[24],[64]
. Where possible, when patients have benefitted from treatment, steps should be taken to optimise therapy, such as:
- Reducing the dose to create a balance between effects and adverse effects;
- Reviewing other medicines that may be causing additional adverse effects, such as anticholinergics or sedative medicines;
- Considering options to manage adverse effects;
- Improving the patient’s understanding of their medicine.
Unless there is an immediate risk, most antipsychotics should be stopped gradually with monitoring to ensure any potential relapse can be identified quickly and managed efficiently.
Useful resources
- Mind: www.mind.org.uk
- Rethink Mental Illness: www.rethink.org
- Mental Health Foundation: www.mentalhealth.org.uk
- British Association for Psychopharmacology: https://bap.org.uk
- Royal College of Psychiatrists: www.rcpsych.ac.uk
- Choice and Medication: www.choiceandmedication.org
- Young Minds: https://www.headmeds.org.uk
Financialand conflicts of interest disclosure
The authors have no relevant affiliations or financial involvement with any organisation or entity with a financial interest in or financial conflict with the subject matter or materials discussed in this manuscript. No writing assistance was used in the production of this manuscript.
Peer-reviewed article
This article has been peer reviewed by relevant subject experts prior to acceptance for publication. The reviewers declared no relevant affiliations or financial involvement with any organisation or entity with a financial interest in or in financial conflict with the subject matter or materials discussed in this article.
References
[1] National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: prevention and management. Clinical guideline [CG178]. 2014. Available at: https://www.nice.org.uk/guidance/cg178 (accessed February 2020)
[2] Mental Health Taskforce. The five year forward view for mental health. 2016. Available at: https://www.england.nhs.uk/wp-content/uploads/2016/02/Mental-Health-Taskforce-FYFV-final.pdf (accessed February 2020)
[3] Buck D, Maguire D; The King’s Fund. Inequalities in life expectancy: changes over time and implications for policy. 2015. Available at: https://www.kingsfund.org.uk/sites/default/files/field/field_publication_file/inequalities-in-life-expectancy-kings-fund-aug15.pdf (accessed February 2020)
[4] Laursen TM. Life expectancy among persons with schizophrenia or bipolar affective disorder. Schizophr Res 2011;131(1–3):101–104. doi: 10.1016/j.schres.2011.06.008
[5] Tsoi DT-Y, Hunter MD & Woodruff PWR. History, aetiology, and symptomatology of schizophrenia. Psychiatry 2008;7(10):404–409. doi: 10.1016/j.mppsy.2008.07.010
[6] Insel TR. Rethinking schizophrenia. Nature 2010;468(7321):187–193. doi: 10.1038/nature09552
[7] Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature 2014;511(7510):421–427. doi: 10.1038/nature13595
[8] van Os J, Kenis G & Rutten BPF. The environment and schizophrenia. Nature 2010;468(7321):203–212. doi: 10.1038/nature09563
[9] Fusar-Poli P, Rutigliano G, Stahl D et al. Development and validation of a clinically based risk calculator for the transdiagnostic prediction of psychosis. JAMA Psychiatry 2017;74(5):493–500. doi: 10.1001/jamapsychiatry.2017.0284
[10] Boksa P. A way forward for research on biomarkers for psychiatric disorders. J Psychiatry Neurosci 2013;38(2):75–77. doi: 10.1503/jpn.130018
[11] Comes AL, Papiol S, Mueller T et al. Proteomics for blood biomarker exploration of severe mental illness: pitfalls of the past and potential for the future. Transl Psychiatry 2018;8(1):160. doi: 10.1038/s41398-018-0219-2
[12] Harvey PD, Loewenstein DA & Czaja SJ. Hospitalization and psychosis: influences on the course of cognition and everyday functioning in people with schizophrenia. Neurobiol Dis 2013;53:18–25. doi: 10.1016/j.nbd.2012.10.022
[13] Green MF. Cognitive impairment and functional outcome in schizophrenia and bipolar disorder. J Clin Psychiatry 2006;67 Suppl 9:3-8; discussion 36–42. PMID: 16965182
[14] Green MF. Impact of cognitive and social cognitive impairment on functional outcomes in patients with schizophrenia. J Clin Psychiatry 2016;77(Suppl 2):8–11. doi: 10.4088/JCP.14074su1c.02
[15] Scottish Intercollegiate Guidelines Network. SIGN 131: management of schizophrenia. 2013. Available at: https://www.sign.ac.uk/assets/sign131.pdf (accessed February 2020)
[16] Byrne P. Managing the acute psychotic episode. BMJ 2007;334(7595):686–692. doi: 10.1136/bmj.39148.668160.80
[17] World Health Organization. The ICD-10 classification of mental and behavioural disorders: clinical descriptions and diagnostic guidelines. 2016. Available at: https://www.who.int/classifications/icd/en/bluebook.pdf (accessed February 2020)
[18] White T, Anjum A & Charles Schulz S. The Schizophrenia Prodrome. Am J Psychiatry 2006;163(3):376–380. doi: 10.1176/appi.ajp.163.3.376
[19] Klosterkötter J, Hellmich M, Steinmeyer EM & Schultze-Lutter F. Diagnosing schizophrenia in the initial prodromal phase. Arch Gen Psychiatry 2001;58(2):158–164. doi: 10.1001/archpsyc.58.2.158
[20] Seppala J, Miettunen J, Jääskeläinen E et al. Definition, epidemiology, clinical course and outcomes in treatment-resistant schizophrenia. Eur Psychiatry 2017;41(Suppl):S67. doi: 10.1016/j.eurpsy.2017.01.070
[21] Pedersen CB, Mors O, Bertelsen A et al. A comprehensive nationwide study of the incidence rate and lifetime risk for treated mental disorders. JAMA Psychiatry 2014;71(5):573–581. doi: 10.1001/jamapsychiatry.2014.16
[22] National Institute for Health and Care Excellence. Psychosis and schizophrenia in children and young people: recognition and management. Clinical guideline [CG155]. 2016. Available at: https://www.nice.org.uk/guidance/cg155 (accessed February 2020)
[23] Ackenheil M & Weber K. Differing response to antipsychotic therapy in schizophrenia: pharmacogenomic aspects. Dialogues Clin Neurosci 2004;6(1):71–77. PMID: 22034253
[24] Ascher-Svanum H, Nyhuis AW, Stauffer V et al. Reasons for discontinuation and continuation of antipsychotics in the treatment of schizophrenia from patient and clinician perspectives. Curr Med Res Opin 2010;26(10):2403–2410. doi: 10.1185/03007995.2010.515900
[25] Joint Formulary Committee. British National Formaulary. London: Pharmaceutical Press; 2019
[26] Stahl SM. Stahl’s Essential Psychopharmacology: neuroscientific basis and practical application. 4th edn. Cambridge: Cambridge University Press; 2014
[27] Taylor D, Barnes TRE & Young AH. The Maudsley Prescribing Guidelines in Psychiatry. 13th edn. New Jersey: Wiley-Blackwell; 2018
[28] Bazire S. Psychotropic Drug Directory. Dorset: Lloyd-Reinhold Communications; 2018
[29] Procyshyn RM. Clinical Handbook of Psychotropic Drugs. 23rd edn. Oxford: Hogrefe; 2019
[30] Müller N. Immunological aspects of the treatment of depression and schizophrenia. Dialogues Clin Neurosci 2017;19(1):55–63. PMID: 28566947
[31] Schobel SA, Chaudhury NH, Khan UA et al. Imaging patients with psychosis and a mouse model establishes a spreading pattern of hippocampal dysfunction and implicates glutamate as a driver. Neuron 2013;78(1):81–93. doi: 10.1016/j.neuron.2013.02.011
[32] Stone JM. Glutamatergic antipsychotic drugs: a new dawn in the treatment of schizophrenia? Ther Adv Psychopharmacol 2011;1(1):5–18. doi: 10.1177/2045125311400779
[33] Lahti A, Weiler MA, Tamara Michaelidis BA et al. Effects of ketamine in normal and schizophrenic volunteers. Neuropsychopharmacology 2001;25(4):455–467. doi: 10.1016/S0893-133X(01)00243-3
[34] Krystal JH, Karper LP, Seibyl JP et al. Subanesthetic effects of the noncompetitive NMDA antagonist, ketamine, in humans. Psychotomimetic, perceptual, cognitive, and neuroendocrine responses. Arch Gen Psychiatry 1994;51(3):199–214. doi: 10.1001/archpsyc.1994.03950030035004
[35] Egerton A, Bhachu A, Merritt K et al. Effects of antipsychotic administration on brain glutamate in schizophrenia: a systematic review of longitudinal 1H-MRS studies. Front Pychiatry 2017;8:66. doi: 10.3389/fpsyt.2017.00066
[36] Dunlop J & Brandon NJ. Schizophrenia drug discovery and development in an evolving era: are new drug targets fulfilling expectations? J Psychopharmacol 2015;29(2):230–238. doi: 10.1177/0269881114565806
[37] Green MF. Impact of cognitive and social cognitive impairment on functional outcomes in patients with schizophrenia. J Clin Psychiatry 2016;77(Suppl 2):8–11. doi: 10.4088/JCP.14074su1c.02
[38] Young J & Geyer M. Developing treatments for cognitive deficits in schizophrenia: the challenge of translation. J Psychopharmacol 2015;29(2):178–196. doi: 10.1177/0269881114555252
[39] Barnes T; Schizophrenia Consensus Group of the British Association of Pyschopharmacology. Evidence-based guidelines for the pharmacological treatment of schizophrenia: recommendations from the British Association for Psychopharmacology. J Psychopharmacol 2011;25(5):567–620. doi: 10.1177/0269881110391123
[40] Bigos KL, Pollock BG, Coley KC et al. Sex, race and smoking impact olanzapine exposure. J Clin Pharmacol 2008;48(2):157–165. doi: 10.1177/0091270007310385
[41] Melkersson KI, Hulting AL & Rane AJ. Dose requirement and prolactin elevation of antipsychotics in male and female patients with schizophrenia or related psychoses. Br J Clin Pharmacol 2001;51(4):317–324. doi: 10.1046/J.1365-2125.2001.01352.x
[42] Pharmaceutical Press. Stockley’s Drug Interactions. London: Pharmaceutical Press; 2019
[43] eMC. Leyden Delta BV. Zaponex 25 and 100 mg Tablets. 2019. Available at: http://www.medicines.org.uk/emc/medicine/30938 (accessed February 2020)
[44] Patchan KM, Richardson C, Vyas G & Kelly DL. The risk of suicide after clozapine discontinuation: cause for concern. Ann Clin Psychiatry 2015;27(4):253–256. PMID: 26554366
[45] Meltzer HY & Okayli G. Reduction of suicidality during clozapine treatment of neuroleptic-resistant schizophrenia: impact on risk-benefit assessment. Am J Psychiatry 1995;152(2):183–190. doi: 10.1176/ajp.152.2.183
[46] Leucht S, Cipriani A, Spineli L et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet 2013;382(9896):951–962. doi: 10.1016/S0140-6736(13)60733-3
[47] Tandon R, Belmaker RH, Gattaz WF et al. World Psychiatric Association Pharmacopsychiatry Section statement on comparative effectiveness of antipsychotics in the treatment of schizophrenia. Schizophr Res 2008;100(1–3):20–38. doi: 10.1016/j.schres.2007.11.033
[48] Lieberman JA, Stroup S, McEvoy JP et al. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 2005;353(12):1209–1223. doi: 10.1056/NEJMoa051688
[49] National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: treatment and management. Clinical Guideline [CG178]: Recommendations. 2014. Available at: https://www.nice.org.uk/guidance/cg178/chapter/1-Recommendations (accessed February 2020)
[50] Johnson KC, LaPrairie JL, Brennan PA et al. Prenatal antipsychotic exposure and neuromotor performance during infancy. Arch Gen Psychiatry 2012;69(8):787–794. doi: 10.1001/archgenpsychiatry.2012.160
[51] Sinclair D & Adams CE. Treatment resistant schizophrenia: a comprehensive survey of randomised controlled trials. BMC Psychiatry 2014;14:253. doi: 10.1186/s12888-014-0253-4
[52] Morrison AP, Law H, Carter L et al. Antipsychotic drugs versus cognitive behavioural therapy versus a combination of both in people with psychosis: a randomised controlled pilot and feasibility study. Lancet Psychiatry 2018;5(5):411–423. doi: 10.1016/S2215-0366(18)30096-8
[53] Jauhar S, Mckenna PJ, Radua J et al. Cognitive-behavioural therapy for the symptoms of schizophrenia: systematic review and meta-analysis with examination of potential bias. Br J Psychiatry 2014;204(1):20–29. doi: 10.1192/bjp.bp.112.116285
[54] Pilling S, Bebbington P, Kuipers E et al. Psychological treatments in schizophrenia: I. Meta-analysis of family intervention and cognitive behaviour therapy. Psychol Med 2002;32(5):763–782. doi: 10.1017/S0033291702005895
[55] Richardson P, Jones K, Evans C et al. Exploratory RCT of art therapy as an adjunctive treatment in schizophrenia. J Ment Heal 2007;16(4):483–491. doi: 10.1080/09638230701483111
[56] Medalia A & Saperstein AM. Does cognitive remediation for schizophrenia improve functional outcomes? Curr Opin Psychiatry 2013;26(2):151–157. doi: 10.1097/YCO.0b013e32835dcbd4
[57] Barlati S, Deste G, De Peri L et al. Cognitive remediation in schizophrenia: current status and future perspectives. Schizophr Res Treatment 2013;2013:156084. doi: 10.1155/2013/156084
[58] Kingdon D. Care programme approach. Psychiatr Bull 1994;18(02):68–70. doi: 10.1192/pb.18.2.68
[59] Hynes C, Keating D, McWilliams S et al. Glasgow antipsychotic side-effects scale for clozapine — development and validation of a clozapine-specific side-effects scale. Schizophr Res 2015(1–2);168:505–513. doi: 10.1016/j.schres.2015.07.052
[60] Kerwin RW & Bolonna A. Management of clozapine-resistant schizophrenia. Adv Psychiatr Treat 2005;11(2):101–106. doi: 10.1192/apt.11.2.101
[61] Cooper SJ, Reynolds GP, Barnes T et al. BAP guidelines on the management of weight gain, metabolic disturbances and cardiovascular risk associated with psychosis and antipsychotic drug treatment. J Psychopharmacol 2016;30(8):717–748. d oi: 10.1177/0269881116645254
[62] Umbricht DS, Pollack S & Kane JM. Clozapine and weight gain. J Clin Psychiatry 1994;55(Suppl B):157–160. PMID: 7961563
[63] Maayan L & Correll CU. Management of antipsychotic-related weight gain. Expert Rev Neurother 2010;10(7):1175–200. doi: 10.1586/ern.10.85
[64] Legge SE, Hamshere M, Hayes RD et al. Reasons for discontinuing clozapine: a cohort study of patients commencing treatment. Schizophr Res 2016;174(1–3);113–119. doi: 10.1016/j.schres.2016.05.002
[65] Central and North West London NHS Foundation Trust. Clozapine: clinical and operational guidelines. 2018. Available at: https://staff.cnwl.nhs.uk/application/files/2815/4781/6192/Clozapine_clinical_and_operational_guidelines.pdf (accessed February 2020)
[66] Swinton M & Ahmed AG. Reasons for non-prescription of clozapine in treatment-resistant schizophrenia. Crim Behav Ment Health 1999;9(3):207–214. doi: 10.1002/cbm.313
[67] Witharana D & Basu A. Rebound psychosis following withdrawal of clozapine. Psychiatrist 2011;35:155. doi: 10.1192/pb.35.4.155
[68] Moncrieff J. Does antipsychotic withdrawal provoke psychosis? Review of the literature on rapid onset psychosis (supersensitivity psychosis) and withdrawal-related relapse. Acta Psychiatrica Scandinavica 2006;114(1):3–13. doi: 10.1111/j.1600-0447.2006.00787.x
[69] Weiden PJ, Kim E, Bermak J et al. Does half-life matter after antipsychotic discontinuation? J Clin Psychiatry 2017;78(7):e813-e820. doi: 10.4088/JCP.16m11308


