This content was published in 2008. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
The human body contains about 50mg/kg of iron. Of this, approximately 60 per cent is in the haemoglobin of red blood cells and 5 per cent is in the myoglobin of muscle cells. A further 30 per cent is found in body stores as ferritin (20 per cent) and haemosiderin (10 per cent), which are the two major iron-storage proteins and are found mainly in the liver. The remaining 5 per cent or so of the body’s iron is distributed among various enzymes. A small amount (<0.1 per cent) is in transit in the circulation bound to transferrin, the main iron transport protein in the body.
Iron has several functions. It is:
- Necessary for the formation of haemoglobin and hence for the transport of oxygen and cell respiration
- Responsible for oxygen storage in the muscle (as a component of myoglobin)
- A component of cytochromes (which are critical for the electron transport chain) and, therefore, involved in energy production (by virtue of its redox activity)
- A component of enzymes necessary for immune function (Iron deficiency may compromise immune function while iron overload may predispose to infection because iron is an essential growth factor for microorganisms.)
Iron intake
Iron intake is required to replace normal losses from the body. Each day, 0.5-1mg of iron is lost in the faeces, urine and sweat, and menstruating women lose an additional 0.5-0.7mg of iron per day (averaged over a month). Heavy menstrual blood loss can result in iron deficiency (see below).
During pregnancy, iron is needed to form additional haemoglobin (equivalent to 1-2mg of iron per day) and, during childhood, intake is needed to develop stores for growth (equivalent to about 0.6mg of iron per day).
Reference nutrient intakes (RNIs) for Iron (amounts judged to be sufficient for most of the population at different ages) are shown in Panel 1.
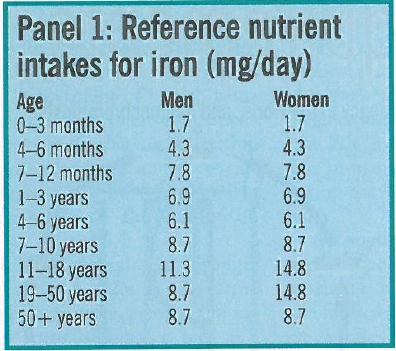
However, mean iron intake in adult men and women in the UK is 13.2 mg/day and 10mg/day, respectively1 and men and women aged 19-34 years have significantly lower iron intakes than those aged 35-64 years. Moreover, 92 per cent of women aged 19-49 have an iron intake below their RNI. Dietary iron exists in two forms: haem iron (ie, iron in haemoglobin), found in meat, and non-haem iron (mainly in the form of iron salts), found in grains, fruit and vegetables. Liver and kidney are the richest sources of haem iron (10mg/100g) followed by red meat (2-3mg/100g), chicken and turkey (0.5-1.0mg/100g) and fish (0.5-1mg/100g).
Foods rich in non-haem iron include wholemeal bread (2mg/two slices), fortified breakfast cereals (2-9mg/bowl), beans and pulses (2-3mg/serving), nuts and dried fruit (1-2mg/small handful), green vegetables (1.5mg/100g) and blackberries and blackcurrants (1mg/ 100g).
Between 15 and 35 per cent of haem iron is absorbed, depending on iron status. Absorption is more effective than that from non-haem iron and is little influenced by the composition of a meal. Conversely, the absorption of non-haem iron is strongly influenced by other dietary constituents, some of which promote iron abscrption (eg, ascorbic acid, meat, citric acid) and some of which inhibit absorption (eg, phytates found in unrefined grains, tannins in tea and calcium). Nonhaem iron absorption can vary from less than 1 per cent to more than 90 per cent.
To enable optimal absorption of iron from the diet it is, therefore, important for everyone (particularly those who do not eat red meat) to consume a dietary source of vitamin C with as many meals and snacks as possible.
The mechanism by which iron is absorbed from the gut is not clearly understood, but recent research has uncovered the existence of a number of genes coding for proteins in the control of iron absorption. Most iron is absorbed in the duodenum and jejunum and, following solubilisation in the stomach, it is transported, mainly by active transport, into the mucosal cells of the duodenum. It then passes into the bloodstream where it is transported by transferrin.
Iron is transported to the bone marrow and other tissues where it is used for metabolic processes. Any absorbed iron in excess of need is stored (as ferritin or haemosiderin) in the liver, spleen or bone marrow from where it can be released in times of high need (eg, in pregnancy). Any unwanted iron is excreted in the faeces.

Iron deficiency anaemia
Iron deficiency is the main remaining nutritional deficiency in Europe. Estimates suggest that over two billion people worldwide suffer from iron deficiency anaemia and many more suffer from depleted iron stores and are at risk of anaemia.
Iron deficiency anaemia develops when there is inadequate iron for haemoglobin synthesis (see Figure 1).

A normal haemoglobin level is maintained for as long as possible after the body’s iron stores are depleted. The period following depletion of these stores and preceding the development of iron deficiency anaemia is described as one of “latent iron deficiency”.
The World Health Organization defines iron deficiency anaemia as a haemoglobin below 13g/dL in men over 15 years of age, below 12g/dL in non-pregnant women and below 11g/dL in pregnant women.2 According to WHO estimates, iron deficiency anaemia is present in 20 per cent of preschool children, 6 per cent of children aged 5-14 years, 10 per cent of adult women (aged 15-59 years), 22 per cent of pregnant women, 4 per cent of adult men (aged 15-59 years) and 12 per cent of people aged over 60 years.2
Using anaemia as an indirect indicator, the WHO has estimated that 30-40 per cent of pre-school children and pregnant women in developed countries are iron-deficient.2 People who are at risk of iron deficiency (through increased requirements or high losses) are listed in Panel 2.

Symptoms
Signs and symptoms of iron deficiency anaemia include pallor, fatigue, restlessness, headaches and faintness, all of which are non-specific and common to other conditions. Severe anaemia is associated with weakness, breathlessness, palpitations, impaired effort tolerance and, eventually, heart failure. Epithelial changes induced by lack of iron in the cells, such as brittle nails, spoon-shaped nails (koilonychia), atrophy of the papillae of the tongue, angular stomatitis and brittle hair, are well documented but occur infrequently.
Iron deficiency anaemia limits work performance and studies have linked it to reduced productivity.7 In children, iron deficiency anaemia is associated with impaired psychomotor development. In pregnancy, there is evidence to suggest that iron deficiency anaemia is associated with premature births, low birth weight and increased perinatal mortality.
Diagnosis
The diagnosis of iron deficiency anaemia partly relies on a taking good clinical history. This should include questions about:
- Dietary intake
- Use of non-steroidal anti-inflammatory drugs (which may lead to gastrointestinal bleeding)
- Presence of blood in the faeces (a sign of haemorrhoids or bowel cancer)
- In women, the duration and heaviness of periods (eg, the use of up to five tampons or sanitary towels per day is normal) and occurrence of clots
Currently, there is no single test to determine, with accuracy, iron status. A combination of several different laboratory methods must, therefore, be used to diagnose iron deficiency anaemia correctly. The most commonly used tests are listed in Panel 3.

Iron deficiency anaemia is usually diagnosed by a haemoglobin level below the cut-off level for age and sex, plus at least two other abnormal iron status measurements. The most commonly used are probably low serum ferritin, high protoporphyrin and, more recently, high serum transferrin receptor.
Treatment
A key issue in the management of iron deficiency anaemia is to find and treat the underlying cause. Iron must also be given to correct the anaemia. With treatment, haemoglobin concentrations should increase by about 1-2g/L every day or 20g/L over three to four weeks. Treatment should be continued for three months after normalisation of haemoglobin levels in order to replenish iron stores.
Oral iron
The treatment of choice for iron deficiency anaemia is an oral preparation (see Panel 4), of which the most cost-effective is ferrous sulphate. Other ferrous salts, such as the fumarate and the gluconate, have equivalent rates of absorption, differing mainly in the amount of elemental iron released.

Slow release and enteric coated preparatons are promoted on the basis of fewer side effects and once daily administration but they dissolve slowly and can bypass the proximal duodenum into an area of the gut where absorption is poor. Cost and side effects are the main determinants of choice for an iron preparation. The recommended oral dose for iron deficiency anaemia in adults is 100-200mg of elemental iron daily (usually ferrous sulphate 200mg tds).
Parenteral iron
If necessary, iron can be given parenterally as iron dextran or iron sucrose. With the exception of patients with severe renal failure receiving haemodialysis, parenteral iron does not produce a faster increase in haemoglobin levels than oral iron provided that the oral iron preparation is taken as directed and absorption is adequate. However, iron stores are replaced much faster with parenteral iron.
Parenteral iron is indicated where there is genuine intolerance to oral iron, poor compliance with oral treatment, continuing blood loss or documented malabsorption. It is commonly used in patients with iron deficiency and active inflammatory bowel disease. It is not recommended for use in children. The total dose is calculated according to body weight and iron deficit, using a standard formula. (Product literature should be consulted.)
There are two parenteral iron preparations:
- Iron dextran (Cosmofer) is given by deep intramuscular injection, slow intravenous injection or intravenous infusion. It carries a small risk of anaphylaxis so a test dose should be given before every dose and the patient observed for 60 minutes after the first test dose and for 15 minutes after each subsequent test dose. Facilities for cardiopulmonary resuscitation must be available.
- Iron sucrose complex (Venofer). The risk of anaphylactic reactions with this preparation is lower than with iron dextran, but the British National Formulary recommends observation of the patient for 15 minutes following a test dose.
Side effects
Side effects of iron include:
- Nausea and epigastric pain (dose-related)
- Altered bowel habit, either constipation or diarrhoea (The relationship with dose is less clear. Constipation can be severe, particularly in older patients, leading to faecal impaction. Iron can exacerbate diarrhoea in inflammatory bowel disease and extra care should be taken with these patients.)
- Anaphylactic reactions (parenteral iron)
Patients should be counselled on the best way of minimising gastrointestinal side effects because this maximises compliance so, although iron preparations are best absorbed on an empty stomach, taking them with meals reduces gastrointestinal side effects.
The incidence of side effects due to ferrous sulphate is no greater than with other iron salts when compared on the basis of equivalent amounts of iron. If side effects occur, the dose can be reduced. Alternatively, starting with 200mg ferrous sulphate od or bd and building up to tds may reduce the occurence of side effects.
In large amounts, iron (as a prospective pro-oxidant) can cause the generation of free radicals, with possible increased risk of disease (eg, cardiovascular disease, cancer).
Iron preparations are a common cause of accidental overdose in children. Symptoms include nausea, abdominal pain, diarrhoea, haematemesis, rectal bleeding and hypotension. Liver damage and coma may occur later.
The child should be taken to hospital immediately and, if possible, the stomach emptied within one hour of ingesting the excessive iron. Specific therapy is with desferrioxamine, to chelate the iron.
Interactions
Iron interacts with several medicines, reducing their absorption. Iron can reduce the absorption of quinolone antibiotics, tetracyclines, bisphosphonates, penicillamine, thyroid hormones, zinc, entacapone, mycophenolate and, possibly, levodopa. Conversely, some medicines (oral magnesium salts [including antacids], tetracyclines, calcium salts, trientine and zinc) reduce the absorption of iron.
Prophylaxis
According to the BNF, prophylaxis with an iron preparation (eg, ferrous sulphate 200mg od or bd, which is also the dose suggested for “mild iron deficiency”) is justifiable in individuals who have risk factors for iron deficiency. It may be appropriate in:
- Pregnancy (however, routine prophylaxis in pregnancy is not recommended — if iron stores are replete, iron is of no benefit)
- Patients with malabsorption conditions, such as Crohn’s disease or ulcerative colitis
- Patients who have had subtotal or total gastrectomy
- Haemodialysis patients
- The management of low birthweight infants, such as pre-term neonates
Over-the-counter products
Several OTC items, mostly multivitamin and mineral preparations, contain iron. Adult products contain recommended daily doses of 10-14mg, with some products for people over the age of 50 years containing lower amounts (eg, 4-6mg). Equivalent products for children contain 2-5mg per daily dose. These products are, therefore, not intended for the treatment of iron deficiency anaemia but for bridging dietary gaps. The Expert Vitamin and Mineral group9 has suggested a guidance safe total daily intake of iron from supplements alone of 17mg for use on a long-term basis. Most supplements sold in UK pharmacies would not contain iron in excess of this amount and are safe to use for people who want to supplement their diets.
People most likely to benefit from such supplements are women who suffer from heavy periods. Those who avoid red meat, particularly when a change is rapid and follows many years of an omnivorous diet, coould also benefit. However, if recommending supplements containing iron, pharmacists should be aware that one in 200 people of northern European descent has a genetic predisposition to the iron-loading disease haemochromatsis. In this inherited condition, iron absortion is inappropriately high and results in an accumulation of the element in tissue mainly the liver, but also the pancreas, heart, endocrine glands and joints.
Symptoms include chronic fatigue, weainess, lethargy, enlarged liver, cirrhosis, abnormal liver function tests, arthritis, cardiomyopathy, psychiatric disorders. Some cases are not diagnosed until the patient presents with clinical problems associated with organ failure (usually around 40 to 50 years of age). Treatment is removal of excess body iron by phlebotomy.
A low iron diet is not sufficient to prevent or treat haemochromatosis. Iron fortified breakfast cereals and large doses of vitamin C should be avoided and consumption of offal and red meat should be reduced.

References
- Henderson L, Irving K, Gregory 1, Bates CJ, Prentice A, Parks 1 et al. National diet and nutrition survey: adults aged 19 to 64 years. Volume 3. Vitamin and mineral intake and urinary analysis. London: HMSO; 2003.
- World Health Organisation. Iron deficiency anaemia. Assessment, prevention and control. A guide for programme managers. Available at http://whqlibdoc.whoint (accessed 6 May, 2008).
- Office of Population Censuses and Surveys Social Survey Division and Department of Health. The National Diet and Nutrition Survey: children aged 1.5 to 4,5 years. London: HMSO, 1995.
- Office for National Statistics Social Survey Division. National diet and nutrition survey: young people aged 4 to 18 years. London: HMSO; 2000.
- Ministry of Agriculture Fisheries and Food. National diet and nutrition survey: people aged 65 years and over. Report of the diet and nutrition survey. London: HMSO; 1998.
- Cardenas VM, Mulla ZD, Ortiz M, Graham DY. Iron deficiency and Helicobacter pylon infection in the United States. Amercan Journal of Epidemiology 2006;163:127-34.
- MacPhail P Iron. In: Essentials of Human Nutrition, edited by Mann 1 and Truswell AS, 3rd edition. Oxford University Press, 2007, pp125-37.
- OTC Directory 2007/2008. Treatments for common ailments. Proprietary Association of Great Britain.
- Expert Vitamin and Mineral Group. Safe upper levels of vitamins and minerals. Available from: http://www.food.gov.uk (accessed 6 May 2008).