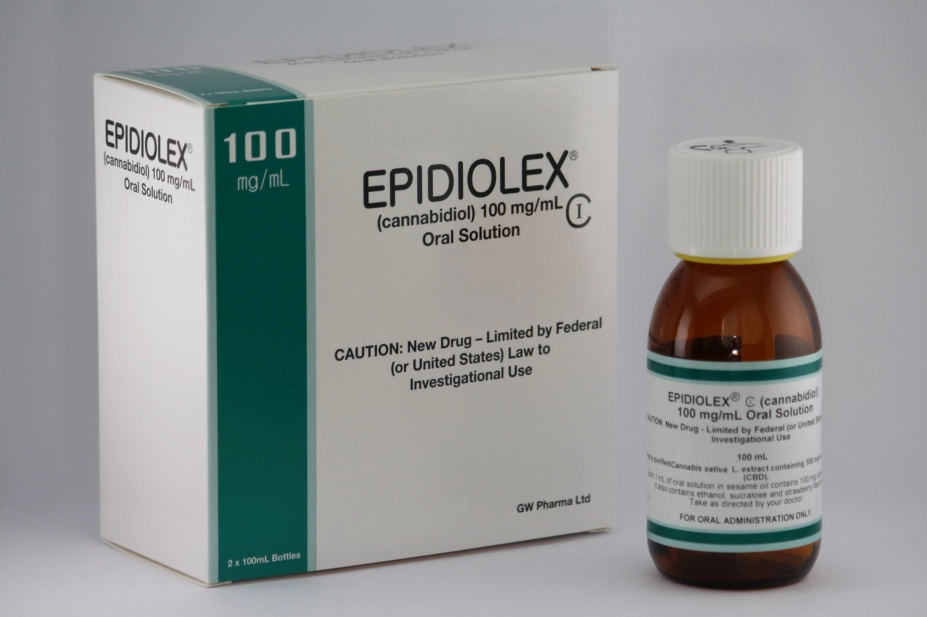
GW Pharmaceuticals
Epidyolex, a cannabidiol medicine for severe epilepsy in children, has been approved for use on the NHS in Scotland.
The decision was announced by the Scottish Medicines Consortium (SMC) on 7 September 2020.
Epidyolex is approved as an adjunct therapy, alongside clobazam, for seizures associated with Lennox-Gastaut syndrome or Dravet syndrome in children aged two years or under.
The SMC said the medicine was considered for NHS use through its patient and clinician engagement (PACE) process, which is used for medicines to treat end-of-life and very rare conditions.
“Through PACE, patient groups and clinicians spoke of the devastating impact Lennox-Gastaut syndrome has on the lives of patients, their family and carers,” the SMC said in its announcement, noting that the PACE programme also discussed the “debilitating and severe” Dravet syndrome.
“We know from the powerful testimony given by patients and clinicians in our PACE meetings that our decisions on cannabidiol for both Lennox-Gastaut syndrome and Dravet syndrome will be welcomed, and hopefully provide some relief for patients and their families,” said Mark MacGregor, chair of the SMC.
Epidyolex is manufactured by GW Pharmaceuticals. Chris Tovey, GW’s chief operating officer, said the SMC’s decision is “proof that cannabis-based medicines can successfully go through extensive randomised placebo-controlled trials and a rigorous SMC evaluation process to reach those patients in need.
“I am especially proud that this is the first time a medicine of this kind has achieved routine reimbursement in Scotland.”
A spokesperson for Epilepsy Scotland said the charity was “delighted”, adding that from “participating in the [SMC’s] PACE process, it is clear that they have listened to patients and clinicians about the profound impact of these severe and difficult-to-treat epilepsy syndromes, and the long-standing need for a specifically approved treatment option”.
Louise Cousins, director of external affairs at the charity Epilepsy Action, said the SMC’s decision was “very welcome”, adding that “new treatment options for severe epilepsies do not come very often”.
However, Cousins said there was “still more work to do”.
“Much-needed clinical trials and alternative access routes should be prioritised so that those people with epilepsy who could benefit from cannabis-based medicines can access them safely on the NHS,” she added.
In December 2019, the National Institute for Health and Care Excellence (NICE) recommended that the NHS pay for the medicine. The recommendation was made in a technology appraisal which applied to England and Wales. Shortly afterwards, NHS England announced it would fast-track funding for Epidyolex, making it available from 6 January 2019.
In June 2020, the medicine was reclassified from Schedule 2 to Schedule 5 of the Misuse of Drugs Regulations 2001, in a move that would “provide more flexibility in prescribing and dispensing of this medicine”, according to Gino Martini, the Royal Pharmaceutical Society’s chief scientist.