
Shutterstock.com
This article contains historic information taken from The Pharmaceutical Journal live blog, which provides updates on the ongoing COVID-19 pandemic.
For the latest information on how the UK pharmacy sector is responding to the COVID-19 pandemic, visit: https://bit.ly/PJCOVIDLive
Friday 3 April
17:00
- Community pharmacists can now refer anyone they believe to be vulnerable for volunteer support during the COVID-19 pandemic. In a document published on 31 March 2020, NHS England and NHS Improvement said that priority for NHS Volunteer Responders would be given to the 1.5 million individuals identified as being most at risk from COVID-19 and asked to self-isolate at home for an extended period, as well as those who health practitioners and local authorities “consider to be vulnerable”. NHS Volunteer Respondents can be asked to help individuals with tasks such as delivering medicines from pharmacies and driving patients to appointments. NHS England and NHS Improvement started matching volunteers with tasks on 1 April 2020.
16:15
- New guidance on personal protective equipment (PPE) was published by Public Health England on 2 April 2020. The guidance states that, for community pharmacy staff, if social distancing of two metres is not maintained, sessional use of fluid repellent surgical masks is recommended. Sessional use means for the duration of duties in a specific clinical care setting or exposure environment.
- However, a table accompanying the guidance appears to give a narrower definition of when a face mask should be worn, specifying that pharmacy staff only need to wear one if they are working in an area with possible or confirmed cases – such as individuals with a new, continuous cough or high fever – and are unable to maintain social distancing of two metres. Although it does add that “sessional use should always be risk assessed and considered where there are high rates of community cases”. Read more here.
15:00
- Community pharmacies in England “will need to be open” over the Easter bank holidays, an NHS England and NHS Improvement (NHSE&I) Primary Care Bulletin has said. The bulletin, sent on 1 April 2020, said this would be “part of the wider NHS response to the pandemic”, with general practices also asked to treat the bank holiday’s “as ordinary working days”. However, the Pharmaceutical Services Negotiating Committee (PSNC) said in a statement on 3 April 2020 that it was still “pressing NHSE&I for urgent confirmation on this matter” as the “ambiguity is unhelpful” given the pressures pharmacies are under.
- Simon Dukes, chief executive of the PSNC, said: “It is clear from our latest conversations that pharmacies will be expected to open for a few hours on both Good Friday and Easier Monday”. He added that the PSNC “will be pressing for funding to cover the additional costs that opening for additional hours – on such short notice and on bank holidays – will bring for contractors who are already struggling to make ends meet through this public health crisis”.
12:20
- Using hydroxychloroquine and azithromycin in combination to treat COVID-19 could increase the risk of a heart attack, cardiologists have warned. The paper, published in Cardiology Magazine on 29 March 2020, says that clinicians who treat COVID-19 patients with both hydroxychloroquine and azithromycin should consider monitoring those patients for ventricular arrhythmia, which can lead to cardiac arrest in those who are critically ill.
- “Until we have clinical outcome data supporting the benefit or harm of these medications, I would advocate for a cautious approach in using the combination of hydroxychloroquine and azithromycin,” said Eric Stecker, associate professor of medicine at the Oregon Health & Science University School of Medicine in Oregon, USA and lead author of the paper. “Any medications that increase the risk of cardiac risk require consideration of both risks and benefits, and right now we do not have evidence that benefits outweigh risks for use of hydroxychloroquine or chloroquine.”
9:20
- The Department of Health and Social Care has published a statement on the £300m advance funding for community pharmacies that was previously announced by the Pharmaceutical Services Negotiating Committee earlier this week. The statement says the advance payment will “will support pharmacies to provide critical services to protect community health, including supplying medicines and providing medical advice to patients, during a period of unprecedented demand”. The statement noted that the government has also reduced the number of services that community pharmacies are required to deliver, including suspending the piloting of new services. Jo Churchill, pharmacy minister, said the government “hugely values the role of the community pharmacy, which is why we are backing them with £300m of advanced funding”.
- Community pharmacies will however be expected to repay the money in 2020/2021, as was previously announced.
Thursday 2 April
16:15
- Independent pharmacies need an “urgent large cash injection” to help them through the COVID-19 pandemic, two West London MPs have said. In a letter to health secretary Matt Hancock, Rupa Huq and Gareth Thomas, Labour MPs for Ealing Central and Acton and Harrow West respectively, highlighted how one of their local community pharmacies was struggling with finances during the pandemic. The MPs’ letter also called for access to proper personal protective equipment, compensation for extra working hours and a reversal of recent pharmacy cuts. The MPs said that current coronavirus business support packages do not help community pharmacies, who are “on the front line helping vulnerable patients stay well”. Community pharmacies “have made a massive difference to the NHS during this crisis — they need our support, and urgently,” the letter says.
13:58
- Pharmacists could be allowed to supply some controlled drugs without a prescription during the COVID-19 pandemic under proposed emergency legislation, the government has said. Details of the proposed legislation were announced in a letter dated 1 April 2020 from Home Secretary Priti Patel to Owen Bowden-Jones, chair of the Advisory Council on the Misuse of Drugs (ACMD). The legislation would allow pharmacists to supply medicines in Schedule 2, 3 and Part 1 Schedule 4 of the Misuse of Drugs Regulations 2001 if the patient had been receiving them as part of ongoing treatment. The proposals would also allow the same controlled substances to be supplied under a Serious Shortage Protocol, so alternative products could be supplied. Patel said these measures would “help secure access to controlled drugs within the healthcare system in a pandemic and where there is a serious risk to human health”, and she asked the ACMD to advise on the potential risk of introducing these measures by 3 April 2020.
12:00
- NHS Pathways carried out 259,069 triages for Covid-19 from phone calls to NHS 111 or 999 in England between 18 March and 1 April 2020, according to figures compiled by NHS Digital. The number of Covid-19 assessments completed online via the NHS 111 site for the same period was 1,565,782. The average number of telephone triages per 100,000 population was 427. NHS Luton Clinical Commissioning Group (CCG) recorded the highest rate of 762.2 per 100,000 while North Bath and North East Somerset recorded the lowest – 232.7 per 100,000. The average number of completed 111 online assessments was 2,797.2 per 100,000 population. NHS Bedfordshire recorded the highest rate of 4,537.9 while NHS Croydon CCG had the lowest rate – 1,193.7.
- A Bristol pharmacy has been gifted a set of 3D-printed face shields, after tweeting concerns about pharmacy staff morale over inadequate personal protective equipment. Knowle West Media Centre, an arts centre and charity based in South Bristol, saw the tweet and made the face shields in their digital fabrication and product design space. Ade Williams, superintendent pharmacist at Bedminster Pharmacy, told The Pharmaceutical Journal that each team member at his pharmacy had now been given one of the shields. The pharmacy is now working with the charity, and other local 3D-printers, to get similar products made for other frontline health care providers. Receiving the shields, Williams, said was an indication that: “community pharmacy is a treasured part of the NHS. Your community will look out for you because you have looked after them.”
Amazing act of kindness from our South Bristol Community. Protective face shields have now been 3D printed for all the @Bedminsterpharm team! Challenge to make available to all HCP and Social care teams also accepted. Help is coming from unexpected sources.#WeAreInThisTogether
pic.twitter.com/GzvFVd5xiy— Ade Williams (@adewilliamsnhs) March 31, 2020
8:15
- The community pharmacy sector in Wales will receive advance payments of £50m by 7 April 2020 to help “alleviate the immediate cash flow issue” caused by demand for prescriptions during the COVID-19 outbreak. A letter from Andrew Evans, chief pharmaceutical officer for Wales, sent to all community pharmacies on 27 March 2020, said the one-off increase in advance payments “will be in addition to the advance paid at the end of March”, adding that the value of each additional payment will match their end of March advance payment. Pharmacies are expected to repay the advance “over a ten-month period in equal instalments starting on 1 June,” the letter said. Judy Thomas, director of contractor services at Community Pharmacy Wales, told The Pharmaceutical Journal that the payment was “welcome”, adding that “feedback from the network is that it met their immediate concerns in respect of the cash flow issues caused by the significant increase in prescription volume”.
Wednesday 1 April
16:50
- The Pharmaceutical Journal has confirmed that a hospital pharmacist has died from COVID-19. East Sussex Healthcare NHS Trust confirmed that Pooja Sharma, who lived in Eastbourne and worked at Eastbourne District General Hospital in East Sussex, has died. The West London Coroners Court confirmed that Sharma’s death was related to COVID-19.
15:45
- “Urgent discussions” are being held over what hours pharmacies should open during the Easter bank holidays, the Pharmaceutical Services Negotiating Committee (PSNC) has said. The negotiator said it was “keen to ensure that neither too few nor too many pharmacies are open”, and was seeking an agreement from NHS England that existing arrangements would suffice. GP surgeries have been asked to open on Good Friday and Easter Monday and NHS England now has the option, under new regulations, of requiring community pharmacies to open for a set number of hours on these days.
- GP surgeries in Scotland have also been asked to open on the Easter holidays. Matt Barclay, director of operations at Community Pharmacy Scotland (CPS), said the organisation would speak with its board of directors, and other relevant parties, and decide what service level is needed from pharmacy.
14:00
- The new Scottish NHS Pharmacy First service, which was scheduled to launch in community pharmacies on 22 April 2020, has been postponed because of the COVID-19 pandemic. In a video update published on 31 March 2020, Matt Barclay, director of operations at Community Pharmacy Scotland (CPS), said that it was “in no-one’s interest” that the service be rolled out on the originally intended date, and that it would instead be introduced later in the year. CPS was, Barclay said, working with the Scottish government to agree on the best time to launch the service.
- Pharmacies across Spain have held a minute’s silence to remember five pharmacists who have died from COVID-19 in the country. The remembrance took place at 12:00 on 1 April 2020. The International Pharmaceutical Federation (FIP) headquarters also observed one minutes’ silence, in solidarity with colleagues in Spain.
Las farmacias sevillanas han guardado a las 12 un minuto de silencio por los 5 boticarios muertos en España por #coronavirus. Desde hoy además colaboran con la @guardiacivil para ayudar a los más vulnerables. Farmacia Cervantes. Mairena del Alcor pic.twitter.com/By0kWPTrRv
— inmaculada jimenez (@inmajimsev) April 1, 2020
12:00
- No medicine has yet demonstrated efficacy in treating COVID-19 the European Medicines Agency (EMA) has said. In a statement published on 31 March 2020, the EMA said it had been in contact with developers of around 40 therapeutic medicines. Clinical trials to assess drugs’ safety and efficacy against COVID-19 include remdesivir (investigational); lopinavir/ritonavir (anti-HIV medicine); and chloroquine and hydroxychloroquine (treatments against malaria and autoimmune diseases). But the EMA said that on the basis of preliminary data no drug has showed efficacy in treating the disease. The EMA also said it was also in discussion with developers of 12 potential COVID-19 vaccines, two of which have entered phase I clinical trials.
- All prescription charges should be suspended during the COVID-19 pandemic, the Royal Pharmaceutical Society (RPS) in England has said. The society has co-signed a letter from the Prescription Charges Coalition sent to health ministers, and the RPS said it was also raising the issue directly during weekly meetings with pharmacy minister Jo Churchill. Claire Anderson, chair of the RPS English Pharmacy Board, said the pandemic had exacerbated the need for some patients to choose between buying medicines, heating their homes or buying food. “We hope to see an emergency waiver on prescription charges as soon as possible,” she said.
For many years we’ve campaigned to abolish Rx charges for people with LTCs as part of @prescriptionCC Find out why we all believe now is the time to scrap Rx charges #rpscovid19
https://t.co/wgoET5XM3q
pic.twitter.com/DvYPcqCfRx— Royal Pharmaceutical Society (@rpharms) March 31, 2020
9:00
- Community pharmacies will be given £300 to pay for screens or other physical barriers to help them ensure social distancing is maintained in the pharmacy during the COVID-19 pandemic. The money was announced in the latest briefing sent to community pharmacists from chief pharmaceutical officer for England, Keith Ridge, on 31 March 2020. The letter also asked pharmacies to restrict sales of formula milk to “sensible quantities” for individual customers to prevent shortages.
- In his letter, Ridge also said he was aware of the demand from pharmacies for posters and other signs that would tell patients whether they could enter the pharmacy or not. He said NHS England was “working on” a poster to warn the public that abuse of pharmacy staff would not be tolerated, and he said they were “looking at” a system that would provide professionally printed posters for pharmacies “in the near future”.
Tuesday 31 March
16:50
- Pharmacies will be given cash advances totalling £300m to help them deal with the COVID-19 pandemic, but no extra funding has been agreed so far, pharmacy negotiators have said. The Pharmaceutical Services Negotiating Committee (PSNC) said that pharmacy contractors will receive a total of £200m as an ‘uplift’ to their January payments, which should be paid on 1 April 2020, or soon after. They will get an extra £100m at the end of April, or in early May, along with their payment for work carried out in February. The payments will need to be “reconciled at a later date”, the PSNC said. The negotiator said it had been pressing the case to government ministers and NHS England for “urgent cashflow assistance” to help pharmacies for a number of weeks. It is still in negotiations over an increase in overall funding to help deal with an increase in prescriptions, staffing costs, and rising drugs bills that have been caused by the pandemic.
- PSNC chief executive Simon Dukes said he had told ministers that community pharmacy was at a “critical point”, with many pharmacies now not financially viable. “This funding gesture alone is not enough,” he said. “We have informed HM Government that it simply will not be sufficient to help many contractors to meet the rapidly increasing costs that they are facing as a result of this pandemic.”

12:10
- A webinar giving hospital pharmacists an overview of critical care has been put together by the Royal Pharmaceutical Society (RPS) and the UK Clinical Pharmacy Association (UKCPA). Mark Borthwick, consultant pharmacist in critical care and Greg Barton, critical care and burns pharmacist, responded to questions from pharmacists who are new to critical care, but who may be drafted in to critical care units during the COVID-19 pandemic. The webinar covered medicines reconciliation, sedation and acute kidney injury in critical care, with a focus on scenarios likely to be encountered when caring for patients with COVID-19.
- Beth Ward, head of education at the RPS, told registrants that the webinar was “just the start of our package on critical care”, and that more support would soon be made available. A recording is available on the RPS website.
11:45
- NHS Scotland has launched an online recruitment system for healthcare staff who have recently stopped practising but are willing to come back to work during the “current public health emergency”. Pharmacists and pharmacy technicians who left the General Pharmaceutical Council register within the past three years are among those who are being asked to help fight the COVID-19 pandemic. Scottish Chief Pharmaceutical Officer, Rose Marie Parr, has written to the pharmacy profession appealing to eligible individuals to sign up for temporary emergency registration in order to “keep our workforce healthy” and continue to provide services.
- Pharmacy bodies have signed a joint letter calling on pharmacies to close entrances in order to control people coming into the pharmacy to maintain social distancing advice of two metres. The General Pharmaceutical Council, Royal Pharmaceutical Society, and Association of Pharmacy Technicians UK were among the signatories to the advice designed to “to protect staff and users of the pharmacy”. Such measures do not negate the need for all pharmacy teams to have access to appropriate personal protective equipment, it added.
09:15
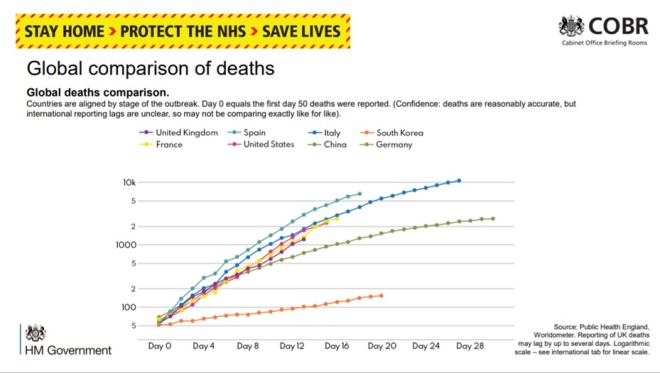
- The social distancing measures put in place by the Government appear to be working, the government’s chief scientific officer said last night. Patrick Vallance said that it was “premature” to put a time on how long these measures might last, but that their prediction was that the measures put in place were enough to see transmission of COVID-19 start to decrease.
- He said: “We think that has had the effect that is desired, that takes 2 or 3 weeks to feed through into the number of people appearing in hospital, a little longer in terms of those seriously ill or who might die, so we expect to see a lag phase before you see these curves changing.” You can see the latest data presented by Vallance here.
Monday 30 March
15:30
- Operating a pharmacy without a responsible pharmacist is still unlawful and should not become “normalised”, even during the COVID-19 pandemic, the General Pharmaceutical Council (GPhC) has warned. Earlier in March 2020 the regulator said that during the current crisis if a responsible pharmacist had to leave a pharmacy premises urgently, and where no locum cover could be secured, it would be in patients’ best interest to supply medicines that have already been dispensed and checked rather than withhold them. Another pharmacist should be remotely available by phone or video.
- But in a letter sent to pharmacies on 27 March 2020, the GPhC said it was concerned that some pharmacies were standardising these emergency arrangements. The regulator said it was “illegal and professionally unacceptable” to normalise a procedure that it expected to happen rarely and in exceptional circumstances.
- A personalised digital ID card to help members confirm their pharmacist status during the COVID-19 crisis has been produced by the Pharmacists’ Defence Association (PDA). In a statement the PDA said: “There are unfortunately still reports of some schools and childcare, supermarkets and even some authorities not being clear about the key role pharmacists perform and challenging pharmacists’ status”.

Source: Pharmacists’ Defence Association
12:15
- “Urgent discussions” over a temporary exemption from signing prescription forms, are taking place between the Pharmaceutical Services Negotiating Committee (PSNC) and the government, the PSNC has said. The PSNC said it was one of a number of proposed changes to pharmacy operations that it was discussing with the Department of Health and Social Care (DHSC), and NHS England & NHS Improvement (NHSE&I) as part of its COVID-19 response. The PSNC has also reminded pharmacy contractors that they must dispense legally valid paper prescriptions during the COVID-19 pandemic. The reminder came after it reported that NHSE&I had recorded several cases of pharmacy contractors refusing to dispense paper prescriptions, including those from dentists and hospital outpatient departments.
08:32
- The Prime Minister singled out the “wonderful” pharmacy profession for particular praise last night in a video posted to Twitter, in which he gave a brief update on the government’s work on the COVID-19 pandemic. Boris Johnson thanked pharmacists for their role in “dispensing vital medicines” and for providing “reassurance for customers they interact with”. The full video is below:
Thanks to everyone who has been staying at home.
By delaying the spread of the disease we can reduce the pressure on our NHS, and that’s how we hope to save many thousands of lives.#StayHomeSaveLives
pic.twitter.com/kxdqItMYSE— Boris Johnson #StayHomeSaveLives (@BorisJohnson) March 29, 2020
Friday 27 March
16:38
- Rose Marie Parr, Scotland’s chief pharmaceutical officer, has sent a message of thanks to Scotland’s pharmacy teams. In the video, Parr said she wanted to add her thanks to those of the First Minister, Nicola Sturgeon, and the cabinet secretary for health, Jeane Freeman. “We can see in these really difficult and challenging times how pharmacy has become the front line”, she said, adding that pharmacy “is now showing itself in the really professional and important light that it always has been, but is now much more obvious to the public”.
14:10
- In Scotland, pharmacy teams and their families will be included in key healthcare worker testing for COVID-19 in order to get self-isolating staff back to work as quickly as possible. However, Harry McQuillan, chief executive of Community Pharmacy Scotland (CPS), said that expectations would need to be managed. “You’ll get a test if we can and if they’re available because there’ll be limited numbers,” he said. “It’s NHS board specific and the CPS team are going to try and collate that on the website as we think that’s a game changer for the workforce.”
- In England, chief executive of the Pharmaceutical Services Negotiating Committee, Simon Dukes, said at a press conference that the pharmacy minister, Jo Churchill, had assured him that community pharmacy staff were on the list of priority people to be tested. But on the question of when the testing would take place, he added: “I don’t know the answer to that but then I don’t think the NHS does either right now.”
- In a House of Commons health and social care committee meeting on 26 March 2020, Yvonne Doyle, medical director at Public Health England, said that pharmacists involved in the COVID-19 response would be considered in the “first tranche” of testing. “Pharmacists are considered very much part of the solution to elements of how we get through this epidemic,” she said.

Source: Alamy Stock Photo
The General Pharmaceutical Council has temporarily registered 3,332 pharmacists and 2,909 pharmacy technicians
12:46
- More than 6,200 pharmacy professionals have been given temporary registration by the General Pharmaceutical Council (GPhC) so that they can to return to work during the COVID-19 pandemic, if they wish to do so. The figure includes 3,332 pharmacists and 2,909 pharmacy technicians. Those given temporary registration had, in the last three years, either voluntarily removed themselves from the GPhC register or were removed for non-renewal, with no fitness to practise issues. All those temporarily registered will be required to meet the GPhC’s standards for pharmacy professionals.
- Duncan Rudkin, chief executive of the GPhC, said that many had already been in touch to say they wanted to return to work in this time of national emergency. “I would like to thank each one of them for considering returning to practice during these very difficult times; their contribution will make a significant difference in the response to this pandemic”, he said.
12:18
- The Royal Pharmaceutical Society (RPS) has announced the cancellation of its National Pharmacy Board elections due to the COVID-19 pandemic. In an announcement, the RPS said that with pharmacists currently devoting all their energy towards supporting patients and the public, “an election would be a diversion from the key priority of the nation and the profession”. Board members whose term was due to end in June will be granted a one-year extension to their term. If those members successfully stand for election in 2021, the RPS said that “it is anticipated they would chose to step down from office one year early in 2023 (when their term of office would have come to an end had they been re-elected in 2020) rather than serving the full three years”.
11:15
- Community Pharmacy Scotland (CPS) has published a letter verifying that pharmacy staff are key workers after it was made aware that pharmacy team members, and delivery drivers, were being stopped and questioned by police officers about their movements. Police have been on patrol in Scotland to enforce government advice on staying at home during the COVID-19 pandemic. The letter, which the CPS has been told will be accepted by police officers, is to act as identification in lieu of a standardised NHS ID card, which are not held by pharmacy professionals. In a statement on its website, the CPS said the letters should be filled out with pharmacists’ details and branch-stamped. “Each member of the team should carry a copy to speed up the process if stopped, and we are told that this will be accepted by officers,” the CPS said.
08:58
- The Royals were clapping for the pharmacy profession last night, as were many others #Clapforpharmacy
To all the doctors, nurses, carers, GPs, pharmacists, volunteers and other NHS staff working tirelessly to help those affected by #COVID19: thank you.#ClapForOurCarers
#ClapForNHS
pic.twitter.com/XnaUPJyDoX— Kensington Palace (@KensingtonRoyal) March 26, 2020
It’s very true that we are dependent on our local pharmacies during this pandemic and we need to give them support and appreciation while they are working under such immense pressures #ClapforPharmacy
https://t.co/DRbOWbqnAo— Keith Ridge (@keithridge1) March 26, 2020
Thursday 26 March
16:48
- The Royal Pharmaceutical Society (RPS) has responded to the General Pharmaceutical Council’s decision to postpone the pre-registration assessments in June and September 2020. Gail Fleming, the director for education at the RPS, said it “fully supports” the postponement as it “recognises the unprecedented and challenging circumstances that many pre-registration pharmacist trainees are in and will enable them to focus on their own wellbeing whilst at the same time providing care to patients.” Fleming added that the RPS also supports “the concept of provisional registration, which will enable trainees to progress without adversely affecting workforce numbers”.
16:15
- The Centre for Postgraduate Pharmacy Education (CPPE) has now launched an online course for pharmacy professionals returning practice in response to the COVID-19 pandemic. After completing a self-assessment framework, pharmacy professionals can refresh their knowledge in nine areas including clinical knowledge, community pharmacy contractual requirements and legislation and professionalism.
13:45
- The pharmacy pre-registration assessments for June and September 2020 have been postponed, the General Pharmaceutical Council (GPhC) has confirmed. In a joint statement published on 26 March 2020, Duncan Rudkin and Trevor Patterson, chief executives of the GPhC and the Pharmaceutical Society of Northern Ireland (PSNI) respectively, said they were looking to reschedule the dates towards the end of 2020, or early in 2021, “once the situation is clearer”.
- The regulators said they are now making plans for the period between now and the rescheduled assessments, which could include “the possibility of a form of provisional registration for current pre-registration trainees”. The GPhC and PSNI also thanked pre-registration trainees “for their patience and professionalism at this time”.
12:50
- Antibody testing kits for COVID-19 are not likely to be available as soon as had been hoped, the Chief Medical Officer for England has said. Answering questions as part of the Prime Minister Boris Johnson’s daily briefing on 25 March 2020, Chris Whitty said the home-testing kits were not likely to be ordered “next week”. Whitty also said that “the right people” should get the tests first so that they could go to work. Earlier on 25 March 2020 Sharon Peacock, director of the National Infection Service at Public Health England had said the tests could be ordered “in days”. Giving evidence to MPs on the House of Commons Science and Technology committee, Peacock said that 3.5 million antibody tests were being evaluated to ensure they work “this week”.
12:35
- ‘NHS volunteer responders’ will be asked to deliver medicines to people’s homes on behalf of community pharmacy. In a webinar, Jill Loader, deputy director of pharmacy commissioning at NHS England and Improvement, told community pharmacists that volunteers who signed up to help the NHS through the Good Sam app would be given advice about delivering medicines and will not be allowed to carry controlled drugs. Read the full story here.
11:50
- The Royal Pharmaceutical Society (RPS) has written to police force s asking for greater protection for pharmacy staff during the COVID-19 outbreak. In a letter to the National Police Chiefs’ Council, the RPS asked them to take a “zero-tolerance approach” to aggressive behaviour from members of the public. They’ve also asked for pharmacy staff to be given more support and reassurance from police. On 23 March 2020 The General Pharmaceutical Council said it was receiving an increasing number of reports that pharmacy staff were experiencing “abuse, disorder and even violence” from members of the public. Robbie Turner, RPS director of pharmacy and member experience, said it was “disappointing to see that some members of the public are behaving poorly towards pharmacy teams. This is not acceptable and will not be tolerated”.
8:35
- A traffic light recording system has been set up for pharmacies to report how likely they are to have to close because of the COVID-19 pandemic. The Pharmaceutical Services Negotiating Committee (PSNC) has developed a template with Pinnacle Health that allows pharmacies to record their operational status as either red, amber, or green. The system provides automatic feedback to local pharmaceutical committees and allows pharmacies to record information required for NHS England and Improvement if the pharmacies hit amber status, which means they have planned closures, or red status, if they have to close. The system was announced as part of joint guidance from the PSNC, National Pharmacy Association, Company Chemists’ Association, and the Association of Independent Multiple Pharmacies on resilience guidance for pharmacy teams.
Wednesday 25 March
16:25
- Home testing kits for COVID-19 could be available to order in a matter of days according to Sharon Peacock, director of the National Infection Service at Public Health England. Giving evidence to MPs on the House of Commons Science and Technology committee on 25 March 2020, Peacock said that 3.5 million antibody tests are being evaluated to ensure they work “this week” before being distributed into the community through an Amazon order or from “somewhere like Boots because it requires a blood prick”. She added that these would be “not just for key workers [but] for the general population”. In addition to these tests, she said “further millions” were being ordered “today”.
- Experts in vaccine development also gave evidence at the session, saying that 30 different vaccine candidates were being worked on across the world. Andrew Pollard, professor of paediatric infection and immunity at the University of Oxford and Melanie Saville, director of vaccine research and development at the Coalition for Epidemic Preparedness Innovations both agreed that although the majority of vaccine candidates would not be available for another year to 18 months – a vaccine could be available within six months. Although Pollard said this would need “a lot of things to fall in place in order for that to happen”.
14:45

Source: Shutterstock.com
On 25 March 2020, Duncan Rudkin, chief executive of the General Pharmaceutical Council (GPhC), assured trainees that the regulator understood that they were concerned about whether the prereg assessment would be going ahead.
- Pharmacy preregistration trainees have been urged to try not to worry about their preregistration exam and focus on “the important role they are playing in supporting patients during the COVID-19 pandemic”, by the pharmacy regulator. In a statement, Duncan Rudkin, chief executive of the General Pharmaceutical Council (GPhC) assured trainees that the regulator understood that they were concerned about whether the prereg assessment would be going ahead. The GPhC was, he said, working with student representative bodies, employers and universities to make decisions that are “fair to trainees, preserve the integrity of the register and which recognise the significant pressures that community pharmacies and hospitals are under”. A further update is expected early next week, he added.
12:50
- Sickness absence amongst the Madrid pharmacy workforce has risen to a reported 7%, according to a Tweet from the Pharmaceutical Group of the European Union (PGEU). The PGEU was quoting the Spanish publication Diariofarma, which reported that 14 pharmacies have closed in the Spanish capital as a consequence of the COVID-19 pandemic. The data was collected by El Colegio Oficial de Farmacéuticos de Madrid (COFM). Spain has been one of the worst affected countries by COVID-19 with reported deaths from the disease reaching 3,434, as at 25 March 2020.
In Madrid, 14 #pharmacies have closed due to #COVID19, with number of sick leaves representing 7% of #pharmacy
#workforce in the area. Another clear signal that #pharmacists need to be protected as they are at high risk & continuity of their services is vital for the population. https://t.co/FG2Z9eSmrs— PGEU (@PGEU) March 24, 2020
12:10
- Pharmacies saw a surge in the number of electronic prescription service (EPS) nominations in the week ending 20 March 2020, according to NHS Digital data. When taken collectively, all Boots pharmacies saw 62,697 more nominations in that week, with LloydsPharmacy branches seeing 41,798 more nominations and Rowlands pharmacies receiving 16,647 more nominations. In the previous week – ending 13 March 2020 – the large multiples saw just 19,162; 10,144; and 4,623 more nominations respectively.
- Online pharmacies have also seen large increases with 19,415 patients nominating Echo, the online repeat prescription service owned by Lloyds, during the week ending 20 March 2020. This was the largest increase in nominations for any individual pharmacy. Pharmacy2U saw the second highest increase with 9,809 nominations. The week before Echo and Pharmacy2u saw 5,576 and 1,815 more nominations respectively.
8:53

Source: Nick Treharne
On 25 March 2020, Vaughan Gething, the Welsh health minister, said: “I know that community pharmacies are facing unprecedented pressure, last week for example, one community pharmacy dispensed nearly 3,000 in one day, when normally they would only dispense 1,000 — three times as much in one day.”
- Vaughan Gething, the Welsh health minister has sent a message to pharmacies, saying that they are now allowed to open an hour later and earlier, and close for up to two hours during the day. “I know that community pharmacies are facing unprecedented pressure, last week for example, one community pharmacy dispensed nearly 3,000 in one day, when normally they would only dispense 1,000 — three times as much in one day.”
- He also encouraged patients not to request home deliveries of medicines and get a family member, friend or neighbour to collect them instead. “Just by doing this, you could make a significant contribution to the ability of our NHS to respond to this crisis. So please only request a delivery if you really do not have anybody else who can collect your prescription.” The full video is here:
A message from @WGHealthandCare to all community pharmacy teams and to the public. (In 2 parts) Thank you so much @vaughangething
pic.twitter.com/vZoM21zTgm— CommunityPharmWales (@CPWales) March 24, 2020
Tuesday 24 March
16:30
- Large pharmacy chains have announced that they are cutting their opening hours to help manage the COVID-19 pandemic. Rowlands and Lloyds pharmacies have begun opening an hour later at 10:00 across England, Scotland and Wales, with some stores closing earlier than previously. Both chains said they would also be closing for an hour at lunchtime, with a Rowlands spokesperson saying the closures would allow time for “exceptionally busy” staff to prepare prescriptions and to give staff some respite. A spokesperson for Boots said some of their pharmacies may close for short periods and may need to limit the number of people in the store during busy periods. Lloyds said it had introduced a two-person policy to maintain social distancing in its pharmacies. Tracey Clements, chief operating officer of Boots, said its stores were introducing markings on the floor to keep staff and patients safe and asking patients to put contact details on prescriptions so they can call or text when the prescription is ready for collection.
We have introduced new opening hours and measures to protect staff and customers due to Covid-19. Our Deputy Superintendent Pharmacist, Victoria Steele explains the changes. Check the opening hours of your local store here – https://t.co/zerjNIgRhC
#lloydspharmacy
#coronavirus
pic.twitter.com/Dohz79EdVv— LloydsPharmacy (@LloydsPharmacy) March 24, 2020
14:00
- Indemnity insurance during the COVID-19 pandemic will be extended to cover pharmacists working beyond their normal scope, or even the law, the Pharmacists’ Defence Association (PDA) has said. The PDA said it had agreed with insurance underwriters to extend cover to pharmacy professionals “in activities that are beyond the normal scope of their work or when they work beyond the law or the professional regulations”. The PDA also said it had been told that NHS Resolution would provide professional indemnity to support compensation payments for patients specifically for any errors that occur in situations related to the treatment or diagnosis of COVID-19.
- Free training for pharmacists returning to the register temporarily to help manage the COVID-19 pandemic is to be available from Thursday (26 March 2020). The Centre for Postgraduate Pharmacy Education is creating a new hub, which will be free to access and regularly updated. It will focus on patient safety and be available to both pharmacy professionals returning to work, and those who have not recently worked in patient-facing roles.
12:00
- Six new COVID-19 research projects have been given £10.5m funding by the National Institute for Health Research (NIHR) and UK Research and Innovation. The projects will focus on investigating potential vaccines; developing manufacturing processes to produce a vaccine at a million-dose scale; repurposing existing drugs to help those hospitalised with COVID-19; and collecting data on COVID-19 to improve knowledge of the disease. The government has announced a £20m fund for COVID-19 research, and health and social care secretary, Matt Hancock, said: “This investment will speed up globally recognised vaccine development capabilities and help us find a new defence against this disease.” The NIHR has announced that all other new clinical trials are being suspended to prioritise COVID-19 studies and enable the redeployment of clinical staff to frontline care.
10:45

Source: General Pharmaceutical Council
The pharmacy regulator has said that it is hearing increasing numbers of reports that pharmacy staff are experiencing “abuse, disorder and even violence” from members of the public
- The GPhC has said that it is hearing increasing numbers of reports that pharmacy staff are experiencing “abuse, disorder and even violence” from members of the public. It has issued a strong statement saying that: “Abuse of pharmacy staff is never acceptable. Pharmacy is a key part of the national response and its workforce are entitled to be treated with the same respect as other key healthcare professionals. This is an especially challenging time for the pharmacy profession and we condemn any abuse.”
- This is supported by a number recent posts on social media. The RPS has produced a poster that may help with communicating delays to patients (click here to print off a copy).
Absolutely horrified to see members of the public shouting at staff in a community pharmacy today . These guys have been under massive pressure and patients have no idea what they are going through. Give them a flipping (polite) break .
— Gillian Cameron (@Gill_Cameron23) March 20, 2020
We are working hard to support our customers in these challenging times. Please help us to help you! Therefore do not abuse our hard working staff, it’s not their fault if we have sold out of toilet rolls etc.
— Mills Pharmacy (@millsgosforth) March 24, 2020
Great to see our fantastic pharmacy teams being recognised. I have never known a week like this in the 25 years I have been a Pharmacist. But please, please could patients #bekind. Everything we do we do to keep our teams and you safe. Too many stories of abuse and aggression. https://t.co/EbPdEMBhhG— Yvonne Williams (@yewilliams73) March 21, 2020
Monday 23 March
16:47
- In a video posted by Community Pharmacy Scotland today, the national clinical director of the Scottish government Jason Leitch said that community pharmacy is one of the “building blocks” of the response to COVID-19. He added: “Community pharmacy is literally at the front line … I need you to step up and I need you to keep stepping up because we are not messing around here. This is this is going to be a long effort to save lives and I need community pharnacy to play a role in that.”
- He also explained that every health board in Scotland will soon roll out hubs and assessment centres for people who need to be tested for COVID-19 and/or be referred on. “The theory is that it should take the pressure off the really difficult hot spots, including community pharmacy,” he explained.
16:00
- The Pharmacy Schools Council (PSC) has said there are currently “no immediate plans to formally mobilise the student body” to help tackle the COVID-19 pandemic. It said the General Pharmaceutical Council (GPhC) had indicated that it was keen to maintain the supply of pharmacy graduates to the workplace. In a statement, the PSC said that if student pharmacists left their courses now “we would miss out on the opportunity of having a group of students who would, should they so choose, be in a position to assist, suitably qualified and healthy, after their exams in May [2020] and beyond when they could well be most needed.” The Guild of Healthcare Pharmacists has said it has worked with hospital trusts and Unite the Union to draw up a job description for MPharm undergraduates who did want to work in a temporary post.
- NHS England has published a survey for former pharmacy professionals considering a return to the register during the COVID-19 pandemic. It asks those considering a return to practice to outline their areas of experience, and to say in which capacity they’d prefer to work: for example, face-to-face, or over the phone. Details provided will be shared with NHS organisations to enable those responding to be placed in a suitable role. The survey also provides a list of frequently asked questions on how a temporary return to practice will work, including training, insurance and indemnity, and how long returning professionals may be needed for.
14:20
- The Royal Pharmaceutical Society (RPS) in England has written to health and social care secretary Matt Hancock expressing its “shock” at the lack of support for pharmacy during the COVID-19 pandemic. Claire Anderson, chair of the RPS English Pharmacy Board said many government communications have focused only on doctors and nurses, and she said many pharmacists were reporting that they were “exhausted and in need of greater support”.
Today the @rpharms writes to @MattHancock for greater recognition and support for pharmacy teams in their phenomenal response to helping fight the #covid19 pandemic.
More info here: https://t.co/75X8D16bep
Our asks: https://t.co/ohYHpfiZnl
#RPSCovid19
pic.twitter.com/dYrAFkoft0— Ravi Sharma (@RSharmaPharma) March 23, 2020
- The Competition and Markets Authority (CMA) has told pharmacies it will tackle the “minority of bad apples” in both the pharmacy and food and drinks sectors who are “charging unjustifiably high prices for essential goods or making misleading claims around their efficacy”. In an open letter, the CMA said “a minority of firms in your sector are seeking to capitalise on the current situation”, and that it would “use all of the powers available to us to ensure that markets continue to work well during the coronavirus outbreak”. The CMA said it wanted to hear of any price increases from wholesalers or suppliers, “so that we can investigate these issues further up the supply chain”.
- Packets of paracetamol are being sold via online bidding sites at increasingly inflated prices amid concerns around shortages resulting from the COVID-19 pandemic, The Pharmaceutical Journal has found. One vendor was selling tablets of paracetamol at over 500 times the retail price at Boots. Click here to read more.
- NHS England has struck a deal with the independent sector which means it will reallocate almost all its hospital capacity to the NHS including nearly 20,000 fully qualified staff, including around 750 pharmacists, technicians, and pharmacy support staff, 8,000 hospital beds and almost 1,200 ventilators. David Hare, chief executive of the Independent Healthcare Providers Network said: “This significant additional capacity across the country will be a major boost to the NHS’s efforts to treat those patients that need hospital care over the coming period and the independent sector stands ready to maintain that support for as long as needed.”
12:50
- Volunteers have been helping to reduce the immense pressure on pharmacies over the weekend, including one very familiar face (see below). If you get a chance, please do send us pictures of how you are coping with the weight of work at the moment and we will publish them. Email editor@phamaceutical-journal.com or tweet them to @pjonline_news.
Good to have some help today from @BruceWarner. Thanks. pic.twitter.com/tn0vlrUOuQ
— Martin (@wickerpharm) March 21, 2020
12:00

Source: Nic Bunce/Shutterstock.com
In an updated Standard Operating Procedure (SOP) for community pharmacy published on 22 March 2020, NHS England said pharmacies can close periodically “at the discretion of the responsible pharmacist” if pharmacies are facing “significant pressure”
- NHS England has given pharmacies permission to close their doors for up to two and a half hours per day to cope with the demand of COVID-19. In an updated Standard Operating Procedure (SOP) for community pharmacy published on 22 March 2020, NHS England said pharmacies can close periodically “at the discretion of the responsible pharmacist” if pharmacies are facing “significant pressure”. However, they must remain open “to the public between 10:00 and 12:00, and 14:00 and 16:00 as a minimum”.
- The SOP also noted that NHS England and PSNC are looking at how to support a medicine home delivery service following the government’s announcement that 1.5 million people are to be “shielded” from face-to-face contact. Community pharmacies, however, are not expected to meet this demand alone, with relatives, neighbours and friends, as well as volunteers “encouraged to collect and deliver medication on their behalf where they are fit and able to do so,” the SOP said.
- In a joint statement, the Pharmaceutical Services Negotiating Committee (PSNC), the Company Chemists’ Association, the National Pharmacy Association and the Association of Independent Multiple Pharmacies said the changes “should help to ensure that pharmacies can stay open to the public, and that staff can work safely.”
Friday 20 March
17:00
- In the midst of lockdown, these Madrid residents came out on their balconies to applaud their local pharmacists for their hard work and resilience during the pandemic. Daragh Connolly, president of the Irish Pharmacy Union, shared the uplifting video, which came from Jaime Acosta Gómez, a community pharmacist and member of the Executive Committee of FIP’s Community Pharmacy Section. The show of appreciation for healthcare workers goes far beyond Madrid. #AplausoSanitario has taken place all across Spain, and similar gratitude is being shown around the world, including Argentina, Turkey, the Netherlands and Switzerland.
Every evening @ 8 throughout Spain locals clap in appreciation of their healthcare professionals from their balconies
This is from my good friend and colleague @jaimeacosta_ as his staff respond to this fantastic gesture in Madrid@IrishPharmacy
@IPSA_Ireland
@FIP_CPS
pic.twitter.com/CDZnpYXizv— Daragh Connolly (@darconn) March 18, 2020
16:15
- A pharmacist in a small village in the north east of England has made his own version of cleansing hand gel and given it out for free to elderly and housebound customers. Yashwanth Koorakula, who owns J&J Pharmacy in Seaham, County Durham, told The Pharmaceutical Journal: “I was starting to see many customers in the pharmacy who had not come in for the last two or three years because they were wanting hand sanitiser. But we couldn’t provide it because there was no stock and I was aware that many of these customers were putting themselves at risk by coming into the pharmacy.”
- So he searched online and found World Health Organization guidance on how to make hand sanitiser using isopropyl alcohol, hydrogen peroxide, glycerol and sterile water. Koorakula sourced containers and they have now been filled and given away free of charge to local patients. “This is an extraordinary time and I just wanted to provide my elderly and housebound customers with some confidence and calm in a time like this,” he said.
15:20
- Submitting revalidation records for pharmacists is being postponed because of the COVID-19 pandemic. The General Pharmaceutical Council (GPhC) has said that any pharmacists with a submission date between 20 March 2020 and 31 August 2020 will not be asked to submit four CPD records, one peer discussion and one reflective account. They will, however, be expected to submit their full revalidation record by their submission due date in 2021. The GPhC said it had taken its decision “due to the challenges and pressures on the pharmacy sector at this time”. All pharmacists will be expected to renew their registration as normal in 2020.
14:00
- The government has banned the parallel export of more than 80 medicines, including adrenaline, insulin, paracetamol and morphine, out of the UK. The restrictions will help to ensure that there is an uninterrupted supply of medicines for NHS hospital intensive care units treating coronavirus patients. Health minister Lord Bethell said the bans aim “to protect patients in the UK and help ensure they can always get the treatments they need”. Companies that parallel export a medicine on the ban list may face enforcement action from the Medicines and Healthcare products Regulatory Agency and risk having their trading licence revoked for serious breaches. The full list of medicines can be found here.

Source: Shutterstock.com
On 20 March 2020, the Department of Health and Social Care has announced a ban on parallel export of more than 80 medicines, including adrenaline, insulin, paracetamol and morphine, out of the UK
13:30
- Regarding paracetamol, some pharmacists are already taking note.
Paracetamol production line at Newdays Pharmacy group! There may be little or no profit in it but there will be a lot of happy patients #paracetamol
pic.twitter.com/Nn8eF3c1YM— Olivier Picard (@OlivierPicardUK) March 20, 2020
13:00
- The National Pharmacy Association (NPA) has clarified that pharmacists are able to break down larger packs of paracetamol for retail sale to cope with supply issues. The NPA highlighted that while pharmacies are still able to procure prescription-only medicine packs of paracetamol, many are reporting shortages of paracetamol tablets 500mg as pharmacy and general sales list packs. After consulting with the Medicines and Healthcare products Regulatory Agency and professional indemnity advisors the NPA said pharmacists can “break down a larger pack to prepare a single supply of a non-prescription item for retail sale” under Regulation 4 of the Human Medicines Regulations 2012.
- The General Pharmaceutical Council (GPhC) echoed this advice, adding that pharmacy staff “should supply the paracetamol to the member of the public directly, with appropriate labelling and safety information supplied in a clear and understandable way” at the pharmacy’s medicine counter. Sandra Gidley, president of the Royal Pharmaceutical Society, said: “In these extraordinary times, you will find yourself in situations where the usual routine doesn’t work, and you need to do things differently. We are totally supportive of you using your professional judgement to help people.”
Following many enquiries from pharmacists regarding splitting packs of #paracetamol from 100 to smaller packs, please see official response from the @TheGPhC. Rule of thumb: as long as #pharmacists act in the best interest of #patients it is ok. pic.twitter.com/iyfTLSe3IN
— Leyla Hannbeck (@LeylaHannbeck) March 19, 2020
- Evidence published in the
New England Journal of Medicine
on 17 March 2020 has shown that the virus that causes COVID-19, SARS-CoV-2, can survive on some surfaces for a matter of hours and even days. The researchers found that SARS-CoV-2 remained viable in aerosols for up to three hours, on copper surfaces for up to 4 hours, cardboard for 24 hours and on plastic and stainless steel for up to 72 hours after application. The researchers said that their study could “provide information for pandemic mitigation efforts”.
12:22
- Pharmacies across the UK are closing intermittently throughout each day to maintain staff wellbeing and patient safety during the COVID-19 pandemic, The Pharmaceutical Journal can reveal. Nick Hunter, chief officer of Nottinghamshire, Rotherham and Doncaster Local Pharmaceutical Committees (LPCs) said he is advising and supporting local pharmacies to take measures, such as closing a few hours early or having a lunchbreak closure to ‘reset’ and allow staff to take proper safety breaks. Read the full story here.
12:00
- The General Pharmaceutical Council (GPhC) has said it is contacting pharmacy professionals who have left the register within the last three years to ask if they would be willing to temporarily join the pharmacy register during the COVID-19 pandemic. In a statement published on 19 March 2020, the GPhC said it had “powers to temporarily register fit, proper and suitably experienced people to act as pharmacists and pharmacy technicians to help protect public health in the event of an emergency.”
- However, the regulator added that people should return to practice only if they are fit to do so in terms of their health, skills, and personal circumstances. Sandra Gidley, president of the Royal Pharmaceutical Society, said the GPhC’s response was “absolutely the right thing to be doing, and I’m glad it’s happening sooner rather than later as we need to ensure that people have any training they might need so that they can backfill.” Gidley added that she was “heartened to see that lot of pharmacists who have desk jobs, and so are not necessarily in a patient-facing environment, have been saying that they’re happy to help.”
09:00
- The list of key workers who are able to still send their children to school has been published. Click here for the full list. Although the list does not mention pharmacists directly, it specifies that frontline health and social care workers and “producers and distributers of medicines” are included. This description has not gone down well with some pharmacists on social media, but the RPS has published a letter that pharmacy teams can use when communicating with schools. Read the letter here.
Disappointing that the key worker list could not have explicitly stated pharmacy staff given the huge selfless contribution every pharmacy team member is making to the #COVID19 effort every day @NikkiKF
@NHSEngland
@10DowningStreet
https://t.co/NIcZ56KAy8
pic.twitter.com/X2RPdoMVMZ— Graham Stretch (@GrahamStretch) March 20, 2020
Thursday 19 March
17:00
- NHS England has announced that the implementation of structured medication reviews and the medicines optimisation service specification through Primary Care Networks (PCNs) will be postponed until October 2020. The services were expected to be rolled out from April 2020, with pharmacists working in PCNs taking the lead on their delivery. However, a letter from Nikita Kanani, NHS England’s medical director for primary care, and Ed Waller, NHS England’s director of primary care strategy, said the services would be postponed to free-up capacity in general practice to cope with demand from Covid-19 cases. NHS England also said it was delaying the deadline for PCNs to submit their workforce plans from the end of June to 31 August 2020.
15:30
- Pharmacies have been told that there are “currently no medicine shortages as a result of COVID-19”, by chief pharmaceutical officer for England, Keith Ridge. In a letter to community pharmacists, Ridge also said it was “essential” that GPs did not write prescriptions for longer periods than usual, and that pharmacies did not order larger than normal quantities of drugs to ensure shortages did not appear. Ridge also said that NHS 111 call handlers had been given additional instructions to avoid sending patients with COVID-19 symptoms, or those with symptoms in a family member, to the Community Pharmacist Consultation Service.

Source: Jeff Gilbert
On 19 March 2019, Keith Ridge, chief pharmaceutical officer for England, said there are “currently no medicine shortages as a result of COVID-19.”
- Scottish community pharmacists will be among frontline NHS staff to be tested for the coronavirus, Jeane Freeman, cabinet secretary for health and sport, has assured the profession, following a meeting with Community Pharmacy Scotland (CPS). Matt Barclay, CPS director of operations, told The Pharmaceutical Journal on 19 March 2020: “She gave us the assurance that community pharmacists — who are very much on the front line at the moment — will be tested. But we are still waiting for detail about what that looks like and when it will start.”
- In Wales, chief medical officer Frank Atherton has said that frontline NHS healthcare workers in patient-facing roles employed in acute medical assessment units; emergency departments; critical or intensive care units; primary care and frontline ambulance staff, would be tested for the virus. They are in the third priority group behind patients in hospital intensive care with pneumonia, acute respiratory stress syndrome or an influenza-like illness or those with the same conditions who need to be admitted to hospital.
- In England, the Pharmaceutical Services Negotiating Committee was pressing the UK government for coronavirus testing for pharmacists. The Royal Pharmaceutical Society was backing the call, and its president Sandra Gidley told The Pharmaceutical Journal on Thursday March 19: “It’s exercising a lot of people’s minds as community pharmacists are effectively the NHS front line as it’s been pushed back because GPs are closing their doors.”
12:30
- The government has commissioned two new services from community pharmacy to help the NHS cope with demand during the Covid-19 pandemic, the Pharmaceutical Services Negotiating Committee (PSNC) has said. Pharmacists will now be able to supply medicines to patients whose GP practice has closed “allowing them to continue receiving their medicines without a prescription”, the PSNC explained in a statement published on 19 March 2020. It added that the PSNC is also in talks over funding for a medicine delivery service to “support vulnerable patients self-isolating at home”, with further details to follow.
- The announcement also detailed the postponement of several contractual services, including hepatitis C testing, blood pressure testing, stop smoking support, and point-of-care testing. The national roll out of GP referrals to community pharmacy through the Community Pharmacist Consultation Service has also been delayed, although the existing pilot will remain in place. The PSNC added that it is also still in talks with the government to secure “a significant cash injection to ensure the resilience of the sector, supply chain and prevent closures..
BREAKING NEWS – COVID-19 Update: PQS, pharmacy services and payments https://t.co/0SnK9g86f8
pic.twitter.com/GJsTIiY6XI— PSNC (@PSNCNews) March 19, 2020
- A free secure instant messaging and video conferencing app is being made available to all NHS employees with an NHSmail account. Called Microsoft Teams, the digital tool also allows direct audio calls and has a virtual meeting function, according to NHS Digital, and it is being provided to help NHS staff work remotely. Sibby Buckle, chair of the Royal Pharmaceutical Society digital forum, said: “It’s going to improve communication. At the moment we are still a bit siloed so this will definitely improve things across the health care team especially if I can instantly message my local GP rather than having one of my dispensers hanging on the phone for ten minutes waiting to get through to the practice.”
Wednesday 18 March
15:00
- Public Health England (PHE) has issued guidance to advise against the use of products for diagnosing COVID-19 infection in community settings, such as pharmacies. PHE said that there was “little information” on the accuracy of the tests, or on how a patient’s antibody response develops or changes during COVID-19 infection, and that is was “not known” whether either a positive or negative result was reliable. “Currently there is no published evidence about the suitability of these tests for diagnosing COVID-19 infection in a community setting,” the guidance said.
- The cost of delivering medicines to vulnerable patients who are self-isolating or have COVID-19 will be reimbursed, Simon Stevens, the chief executive of NHS England, has said. A joint letter from Stevens and Amanda Pritchard, chief operating officer at NHS England and NHS Improvement, said that “where required” CCGs would be funded for the service, as well as for an NHS urgent medicines supply service for patients whose GP surgery was closed. Reimbursement would also be made for “payments to contractors who are required to close due to COVID-19 related reasons”. Read the full letter here.

Source: Shutterstock.com
In a statement issued on 16 March 2020, the Pharmaceutical Services Negotiating Committee said it was in “complex” and “ongoing” discussions with the Department of Health and Social Care and NHS England and NHS Improvement “to address the potential impact on community pharmacy” of COVID-19
- Community Pharmacy Wales has announced that the community pharmacy sore throat test and treat service has been suspended.
- Most pharmacies in England have now received limited supplies of protective equipment (PPE), but local pharmaceutical committees have warned that it is not enough. Tania Farrow, chief officer of Suffolk LPC, said guidance was to use the PPE in cases where there has been a direct risk of transmission of COVID-19, or for decontamination of rooms where people with symptoms have been isolated awaiting further instruction. But she said the PPE supplies were not adequate and there were no stocks to order from the wholesalers. “We now need to go much further in protecting hard working community frontline staff as they struggle to maintain patient services,” she said.
10:00
- Andrew Evans, chief pharmacist for Wales, has announced a series of changes to community pharmacy services in Wales, including suspension of the medicine use review service until April 2021.
Following last week’s announcement by @vaughangething that primary care contracts would be relaxed, I have today written to #communitypharmacy contractors in Wales setting out the detailed changes we are making to support them during the current #coronavirus situation pic.twitter.com/emYzhSwDEZ
— Andrew Evans (@AndrewEvansCPhO) March 17, 2020
- Among a package of measures designed to tackle the financial effects of the COVID-19 pandemic, the government has said it has postponed reforms to the off-payroll working rules IR35, until April 2021. Pharmacy bodies had criticised the effect these changes could have on the pharmacist locum workforce and Gareth Jones, head of corporate affairs at the National Pharmacy Association, said: “This is a sensible response to the ongoing spread of COVID-19.”
References
Rose Marie Parr, Scotland’s chief pharmaceutical officer, has sent a message of thanks to Scotland’s pharmacy teams. In the video, Parr said she wanted to add her thanks to those of the First Minister, Nicola Sturgeon, and the cabinet secretary for health, Jeane Freeman. “We can see in these really difficult and challenging times how pharmacy has become the front line”, she said, adding that pharmacy “is now showing itself in the really professional and important light that it always has been, but is now much more obvious to the public”.


