Shutterstock.com
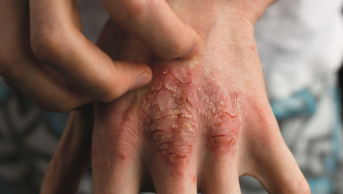
There is no evidence of the clinical benefit of including emollient bath additives in the standard management of eczema in children, researchers have found[1]
.
The study, published in The BMJ (2 May 2018), was a randomised open-label superiority trial comparing two parallel groups of children aged 1–11 years who had been diagnosed with atopic dermatitis.
In the first group, participants were prescribed emollient bath additives to be used regularly for 12 months and, in the second group, participants were asked to use no bath additives for 12 months. Both groups were advised to continue with their usual eczema care during the trial.
Eczema control was measured by the patient-oriented eczema measure (POEM) and was assessed weekly for 16 weeks. The mean POEM score over the 16-week period was 7.5 in the bath additives group, compared to a based line score of 9.5, and 8.4 in the no bath additives group, compared to a baseline score of 10.1.
No statistically significant difference was found in weekly POEM scores between groups over the 16 weeks.
The groups also did not differ in terms of the secondary outcomes, which included number of eczema exacerbations resulting in primary healthcare consultation and quality of life after one year, economic outcomes or adverse effects.
The authors said that their findings were timely for clinicians and prescribing advisers, as prescribing guidelines vary widely in their advice on the use of bath additives and pressure on budgets has led to formularies becoming increasingly restrictive. They added that reviews have estimated that bath additives might contribute to up to one-third of the costs of eczema in the UK.
Further research needs to be carried out on optimal regimens for leave-on treatments, soap substitutes, and frequency of bathing in eczema treatment, they concluded.
References
[1] Santer M, Ridd M, Francis N et al. Emollient bath additives for the treatment of childhood eczema (BATHE): multi-centre pragmatic parallel group randomised controlled trial of clinical and cost-effectiveness. BMJ 2018;361:k1332. Available at: http://eprints.nottingham.ac.uk/50464/ (accessed 3 May 2018)