Dignitana
A cap that cools the scalp to reduce hair loss (alopecia) in women undergoing chemotherapy for breast cancer has been cleared for marketing by the US Food and Drug Administration (FDA).
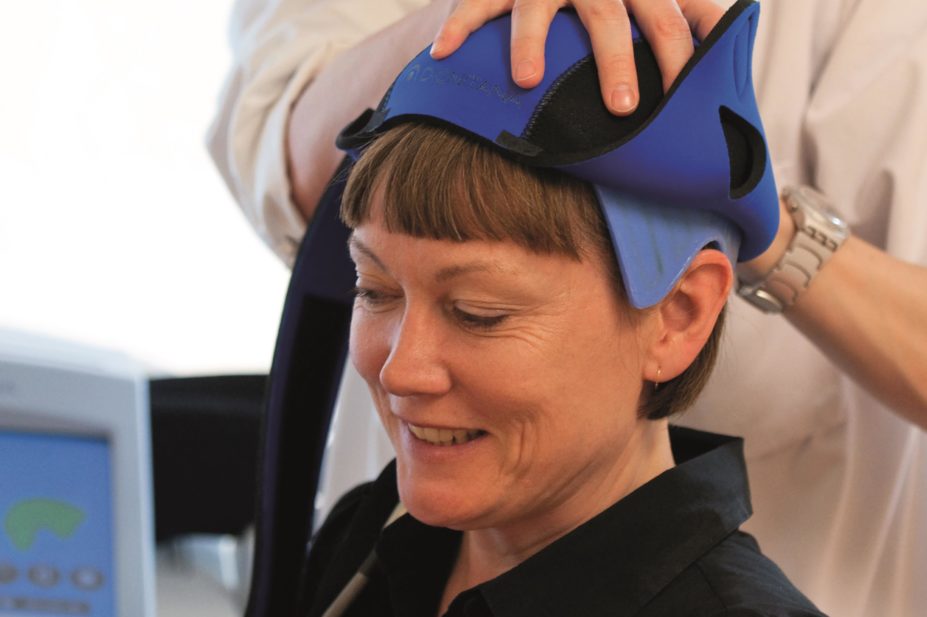
The DigniCap Cooling System is a computer-controlled setup that circulates cooled liquid to a cap worn on the patient’s head during chemotherapy, covered by a second layer that insulates and holds the cap in place. The cooling is intended to constrict blood vessels in the scalp to reduce the amount of chemotherapy reaching hair follicle cells and its effect on them, with the aim of reducing hair loss.
“We are pleased to see a product for breast cancer patients that can minimize chemotherapy-induced hair loss and contribute to the quality of life of these individuals,” says William Maisel, acting director of the Office of Device Evaluation in the FDA’s Center for Devices and Radiological Health. “Managing the side effects of chemotherapy is a critical component to overall health and recovery.”
The cooling cap, manufactured by Swedish medical technology company Dignitana, has been indicated to reduce the frequency and severity of alopecia in breast cancer patients undergoing chemotherapy in which alopecia-inducing chemotherapeutic agents and doses are used.
The FDA considered evidence from a study of 122 women with stage I and II breast cancer undergoing treatment with chemotherapy regimens linked to hair loss. More than 66% of women using the cap reported losing less than half their hair.
The risk of chemotherapy missing an isolated group of breast cancer cells in the scalp because of the cooling cap was described as extremely rare. Reported side effects included cold-induced headaches, neck and shoulder discomfort, chills and pain associated with wearing the cooling cap for an extended period of time.