Shutterstock
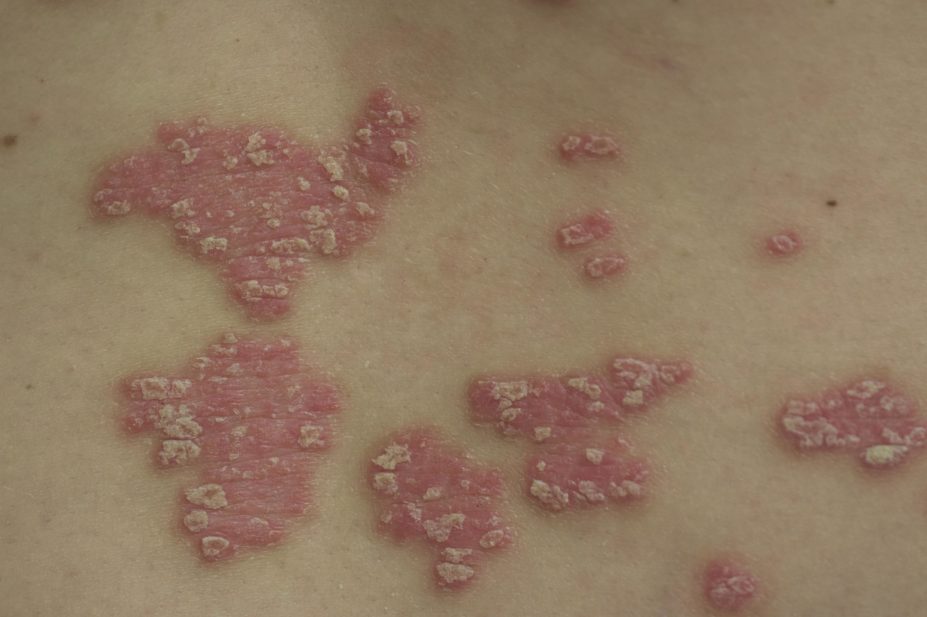
The US Food and Drug Administration (FDA) has approved an injectable treatment, ixekizumab (Taltz; Eli Lilly), for adults with moderate to severe plaque psoriasis.
The drug is a monoclonal antibody that inhibits interleukin-17A, which contributes to skin inflammation and the formation of plaques. The FDA advises that it is intended for patients who are candidates for systemic therapy, phototherapy or a combination of both.
“[This] approval provides patients suffering from plaque psoriasis with another important treatment option to help relieve the skin irritation and discomfort from the condition,” explains Julie Beitz, director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research.
According to the FDA, ixekizumab’s safety and efficacy was established in three randomised, placebo-controlled clinical trials involving data from 3,866 participants with plaque psoriasis who were candidates for systemic or phototherapy therapy. The most common side effects include upper respiratory tract infections and injection site reactions.
In a review of the three trials, an improvement of 75% or greater was achieved in almost 90% of patients treated with ixekizumab, compared with rates of around 3–4% in those treated with placebo. In those receiving etanercept, a biologic used to treat psoriasis as well as conditions such as rheumatoid arthritis, 50% of patients improved.
At the end of February 2016, the European Medicines Agency’s Committee for Medicinal Products for Human Use also recommended that a marketing authorisation be granted for ixekizumab. The recommendation will now go to the European Commission for approval.