Shutterstock.com
Open access article
The Royal Pharmaceutical Society has made this article free to access in order to help healthcare professionals stay informed about an issue of national importance.
To learn more about coronavirus, please visit: https://www.rpharms.com/resources/pharmacy-guides/wuhan-novel-coronavirus
Source: Shutterstock.com
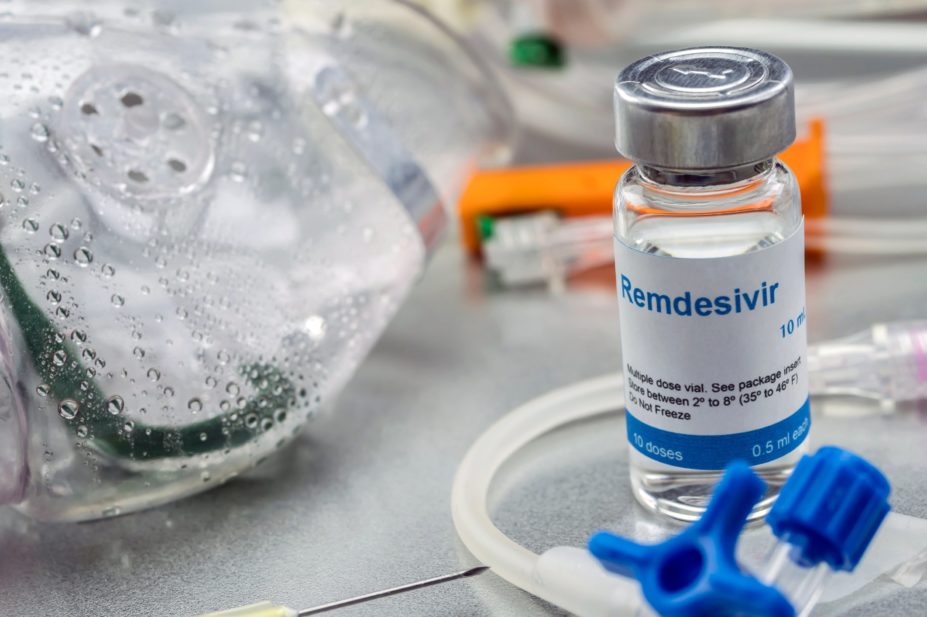
Remdesivir will soon be available as a treatment for some patients with confirmed COVID-19
Remdesivir will become the first COVID-19 treatment in the UK to be made available for use outside a clinical trial.
The Department of Health and Social Care (DHSC) announced on 26 May 2020, that selected COVID-19 patients would soon be able to access the investigational antiviral medicine following evidence that it could shorten recovery time.
The move to make remdesivir —which inhibits RNA-dependent RNA polymerase and, therefore, interferes with RNA replication — available to selected NHS patients has been supported by the Early Access to Medicines Scheme (EAMS) and scientific opinion of the Medicines and Healthcare products Regulatory Agency (MHRA).
The DHSC said that, “for the time being, and due to limited supplies,” the treatment would be prioritised for patients who have the “greatest likelihood of deriving the most benefit”.
June Raine, chief executive of the MHRA, said that the organisation was “committed” to ensuring that patients had fast access to promising new treatments for COVID-19.
“[There are several] trials testing the efficacy of remdesivir against [SARs-CoV-2] — some of which have been unsuccessful — but others, such as the double-blind adaptive COVID-19 treatment trial, have shown that hospitalised COVID-19 patients recovered faster (median 4 days) than patients not receiving remdesivir,” said Gino Martini, chief scientist at the Royal Pharmaceutical Society.
“Therefore, the MHRA has allowed remdesivir to be used in the UK for patients with high unmet medical need (determined by a doctor) through the EAMS, where it will be provided to the NHS free of charge by Gilead Sciences throughout the EAMS period. It will also continue to be used in clinical trials.”
He added that the EAMS allowed doctors and Gilead Science to collate real-world evidence about the effectiveness of remdesivir and its eventual authorisation if it is found help patients with COVID-19.
Meanwhile, all clinical trials assessing the use of hydroxychloroquine or chloroquine to treat or prevent COVID-19 are being reviewed by the MHRA Commission on Human Medicines following the results of a large observational study in The Lancet
which examined the effects of the two drugs on hospitalised COVID-19 patients.
The study was unable to confirm a benefit of either drug, used either alone or in combination with macrolide antibiotics, for patients with COVID-19, and found that their use was linked to an increased risk of serious heart rhythm complications and a decrease in in-hospital survival.
A spokesperson for the MHRA said that clinical trial sponsors were being approached “to urgently provide more information” and that, following assessment, further regulatory action would be taken “if necessary”.
Richard Hobbs, head of the Nuffield Department of Primary Care Health Sciences and co-principle investigation on the ‘Platform randomised trial of interventions against COVID-19 in older people’ (PRINCIPLE), told The Pharmaceutical Journal he would be “surprised” if the MHRA did not permit them to carry on with the trial.
“There was not sufficient detail in the paper to check whether they adequately adjusted for potential confounders — the authors themselves said you cannot judge a drug’s safety on the data that they were using, which you can’t — you need randomised control trials, which is why trials like PRINCIPLE exist,” he explained.
You may also be interested in

Antivirals have little effect on mortality in patients hospitalised with COVID-19, suggest WHO trial interim results
Government tightens availability of remdesivir treatment for COVID-19 patients as supplies shrink