BSIP SA / Alamy Stock Photo
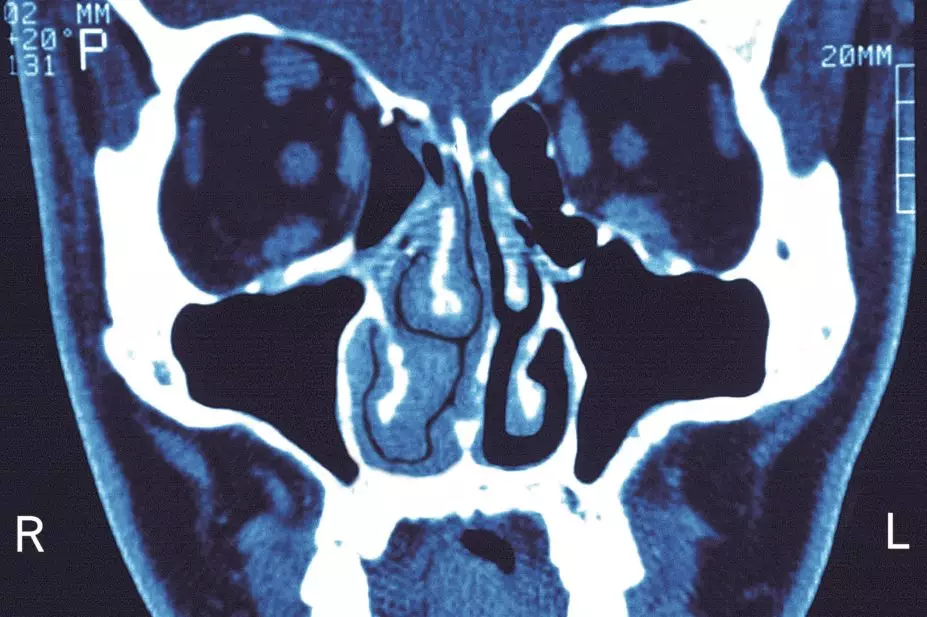
Topical intranasal steroids (INS) are underused among patients with chronic rhinosinusitis (CRS), a Canadian study has found.
Using data involving 19,057 adult patients who received a diagnosis of CRS from 2011–2014 in the province of Alberta, researchers found that only 3,821 (20.1%) of patients were using INS treatment in the 2014–2015 financial year.
Among the patients who did receive INS, the average quantity used was 3.1 bottles, indicating that patients only used INS for around two to three months.
Lead author Luke Rudmik, a clinical associate professor at the University of Calgary, says the findings show that guidelines alone are not enough to improve quality of care.
“There may be multiple factors involved, some of which involve a lack of understanding of chronic sinusitis, which leads to a lack of knowledge on recommended treatments,” he says.
“There may also be a disconnect between provider beliefs about the clinical effectiveness of topical steroids and practice guideline recommendations.”
The team also found significant geographic variation in both the rate and quantity of INS use: across 64 geographic regions, rates of INS use ranged from 14.7% to 41.7% and average quantity used ranged from one to five bottles.
“This is important because it shows some providers are doing a good job at prescribing topical steroid sprays while others are not; therefore, if we can identify why there are differences in behaviour, we can begin developing strategies to improve the overall prescribing practices,” Rudmik adds.
Reporting their findings in JAMA Otolaryngology – Head & Neck Surgery
[1]
(online, 25 August 2016), the team point out that their findings show little difference from when INS prescribing rates were last assessed in North America, when a study[2]
published in 2011 involving data from patients across the United States with CRS showed that only 10% of all outpatient visits resulted in a prescription of a topical INS spray.
Claire Hopkins, an ear, nose and throat (ENT) consultant at Guy’s Hospital in London, says that while it is disappointing that INS use has remained low, it is not surprising. She suggests that there are multiple factors at play, including that many patients self medicate and that adherence can be impeded by costs.
She adds that some primary care physicians may avoid prescribing INS because historically they were associated with systemic side effects.
“New formulations have much lower levels of systemic absorption (for example, mometasone) and so are safe for long-term use, with histological samples showing no evidence of thinning of the nasal mucosa and cortisol assays showing no suppression of the hypothalamus-pituitary axis,” she says.
“This information, and the recommendations of the guidelines, takes a long time to reach primary care physicians, who provide the majority of care to these patients.”
Hopkins also adds that INS may not be sufficient alone to control CRS symptoms and, rather than adding on other therapies, patients may end up discontinuing treatment altogether.
Amena Warner, head of clinical services at charity Allergy UK, says that the results of the study are worrying given that INS are one of only a few effective therapies for CRS, but adds that Alberta’s population may not be comparable with the entire UK population.
“This was a Canadian study in one province only of Canada and cannot therefore be assumed that the UK population would be the same.
“Further UK research into this topic area is required, perhaps as a comparative study or to investigate a UK population to see if topical nasal steroid sprays are being prescribed as and when required,” she adds.
References
[1] Rudmik L, Xu Y, Liu M et al. Utilization patterns of topical intranasal steroid therapy for chronic rhinosinusitis. JAMA Otolaryngology – Head & Neck Surgery 2016. doi: 10.1001/jamaoto.2016.1110
[2] Lee LN & Bhattacharyya N. Regional and specialty variations in the treatment of chronic rhinosinusitis. Laryngoscope 2011;121(5):1092–1097. doi: 10.1002/lary.21550
You may also be interested in

NHS England says PCNs should work with community pharmacy ‘in the best interests of patient care’
People harmed by valproate could receive £100,000 payout, patient safety commissioner says
