Shutterstock.com
Open access article
The Royal Pharmaceutical Society has made this article free to access in order to help healthcare professionals stay informed about an issue of national importance.
To learn more about coronavirus, please visit: https://www.rpharms.com/resources/pharmacy-guides/wuhan-novel-coronavirus
Source: Shutterstock.com
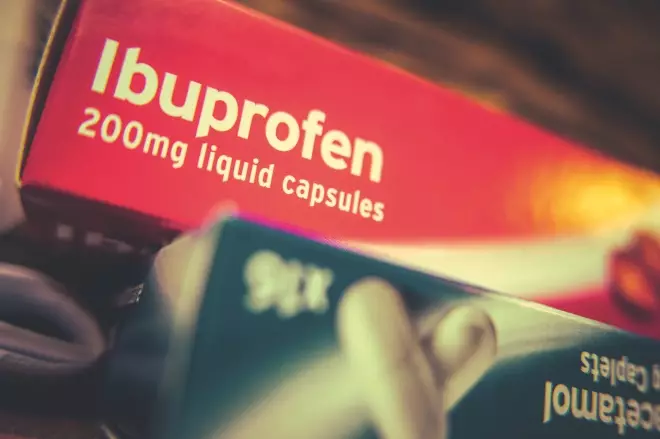
Reports that taking ibuprofen increases the risk and severity of COVID-19 have been circulating on social media
The Committee of Human Medicines — an advisory body of the Medicines and Healthcare products Regulatory Agency — and the National Institute for Health and Care Excellence (NICE) are to review the evidence on the impact of non-steroidal anti-inflammatory drugs (NSAIDs) on the severity of COVID-19 infection.
This is following claims, backed by France’s health minister, Oliver Veran, that ibuprofen may exacerbate symptoms of the virus.
In a message sent out to all clinical staff on 17 March 2020, Stephen Powis, medical director at NHS England, said there was currently “no literature on the impact of NSAID use in COVID-19”, although there was “some literature” suggesting NSAIDs may increase complications from simple acute respiratory infections or slow recovery. But he said that the evidence was “not conclusive overall”.
“In view of the current lack of clarity the Committee of Human Medicines … and NICE have been asked to review the evidence,” he added. “It is therefore suggested that, in the interim, for patients, who have confirmed COVID-19 or believe they have COVID-19, that they use paracetamol in preference to NSAIDs.
“Those currently [taking] NSAIDs for other medical reasons (e.g. arthritis) should not stop them.”
The NHS also provided updated guidance for patients to advise individuals with COVID-19 to take paracetamol to treat their symptoms, and provide reassurance that there was “currently no strong evidence” that ibuprofen could make [COVID-19] worse.
“But until we have more information, take paracetamol to treat the symptoms of coronavirus, unless your doctor has told you paracetamol is not suitable for you,” the guidance says.
Speaking at a House of Commons health and social care committee meeting on 17 March 2020, Patrick Vallance, the government’s chief scientific adviser, told MPs: “It may or may not be right, I don’t know. The sensible thing to do for the moment would be not to take it — to take paracetamol or something else.”
Gino Martini, chief scientist at the Royal Pharmaceutical Society said the evidence was “inconclusive”.
“While there is some evidence to suggest that analgesics and NSAIDS interact with the immune response, the mechanism and clinical significance of this interaction are not clear and do not provide justification to stop taking these medicines.”
Martini added that, aligned with current clinical practice, paracetamol was the preferred first line treatment to control temperature in patients with underlying health conditions.
Esmita Charani, a senior lead research pharmacist in the department of infectious diseases at Imperial College London said that she did not believe that there was enough evidence to support the claims that ibuprofen could worsen COVID-19 symptoms.
“Furthermore, some of the information shared on social media is false,” she said.
“It’s disheartening to hear so much confusion amongst my pharmacist colleagues who have been in touch with me about what is fact and what is fiction,” she added.
A correspondence article published in The Lancet on 11 March 2020 highlighted that human pathogenic coronaviruses, such as SARS-CoV-2, bind to their target cells through angiotensin-converting enzyme 2 (ACE2), which is expressed by epithelial cells of the lung, intestine, kidney and blood vessels.
It explained that the expression of ACE2 was “substantially increased” in patients with type 1 or type 2 diabetes mellitus, who are treated with angiotensin-converting enzyme inhibitors and angiotensin II type-I receptor blockers and “can also be increased by thiazolidinediones and ibuprofen”.