UrbanImages / Alamy Stock Photo
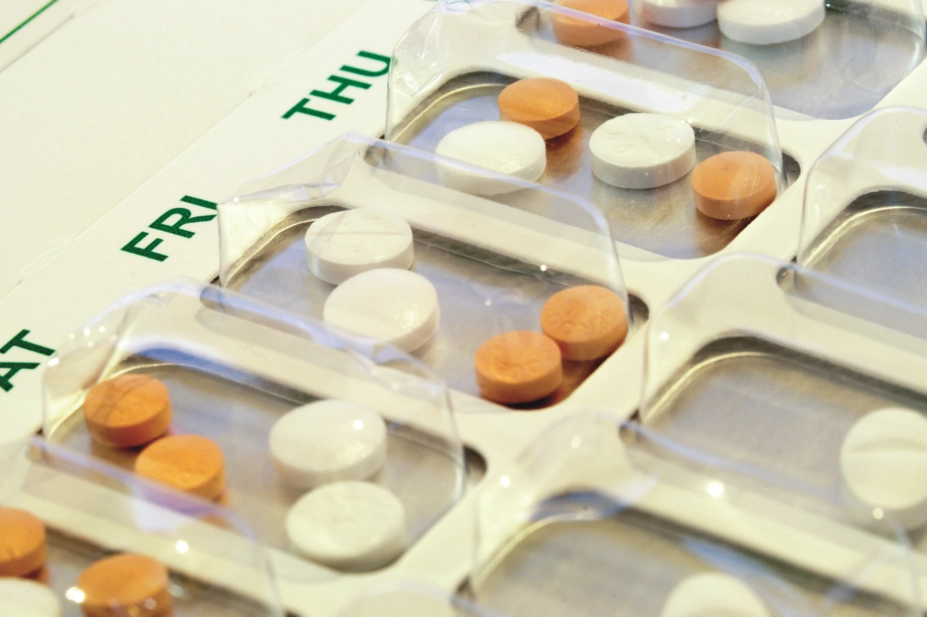
Exclusive: An estimated 64 million multicompartment compliance aids (MCAs) are given out by community pharmacies each year in England, a new report has shown, as data show rising reported incidences of associated harm.
The report, commissioned by the Academic Health Science Network North East and North Cumbria and seen by The Pharmaceutical Journal, is part of an investigation into the use of MCAs, and includes the results of a telephone survey of 50 randomly-selected community pharmacists in England.
The survey results suggest that MCAs are supplied to around 1.2 million patients each year. Some 94% of pharmacies dispensed medicines using MCAs over a two-year period, but only 28% completed a needs assessment for patients before commencing an MCA. Only 11% of the pharmacies surveyed reassessed the needs of those patients who receive MCAs.
The report comes as an investigation by The Pharmaceutical Journal revealed more than 500 patient safety incidents involving the terms ‘monitored dosage’ or ‘dosette box’ were reported to the National Reporting and Learning System in the first half of 2018.
Of these incidents, the data show that 5 reports were linked to ‘moderate’ patient harm and 51 were linked to ‘low’ patient harm. The data also revealed a steady increase in incident reports in the past few years, from 603 reports in 2016 to 972 reports in 2017. In 2016 and 2017, there were 103 and 96 cases of low or moderate harm, respectively.
Experts have said the figures of harm are likely to be an underestimate and that the widespread use of MCAs represent a “gargantuan patient safety issue”.
This new report from the Academic Health Science Network North East and North Cumbria recommends that the General Pharmaceutical Council (GPhC) take responsibility for ensuring that all patients receive an appropriate needs assessment before a decision is made to use a MCA, and that an annual reassessment is undertaken for all patients who use one.
The report adds that it is likely that most damage caused by inappropriate MCA use results from the fact that patients either: cannot refuse a medicine because it is in an MCA and go on to suffer adverse effects as a result; don’t have the root cause of their non-adherence addressed; or, have a progressive illness and don’t get reassessed once they are on a tray, which could mean the MCA ceases to become useful and could become a risk.
Andre Yeung, a specialist pharmacy advisor and local professional network chair for Northumberland, Tyne and Wear, who led the report, said: “This is a gargantuan patient safety issue that has ‘hidden’ in plain sight for many years now. For the most part, despite all the risks, pharmacists don’t know if people need an MCA before supplying one, and once on an MCA, patients usually stay on it.”
The report also identified other negative implications of repackaging medicines into a MCA, including patients not supplying their informed consent; reduced medicine stability; increased medicines wastage; medicines identification becoming more difficult; and increased error frequency and severity.
Commenting on the report, Lelly Oboh, a consultant pharmacist for older people at Guy’s and St Thomas’ NHS Foundation Trust, believes that many errors involving MCAs are not reported because they involve patients who do not have contact with a pharmacist or anyone else who would notice an error.
“A lot of the errors are never found,” she said.
Also responding to the report, a spokesperson from the GPhC said that, as part of its inspections, the regulator routinely looks at how medicines are delivered to patients — including MCAs — to seek assurances that they are being supplied to the patient as safely as possible. However, the spokesperson added that it is not within its remit to be “prescriptive about the exact ways medicines are supplied”.
“We expect pharmacy professionals to have considered relevant guidance on the safe use of MCAs issued by professional and other organisations.”
The Pharmaceutical Journal revealed on 28 January 2019 that Boots is changing the way it dispenses medication for care home residents from monitored dosage systems (MDS) to patient pack dispensing (PPD) from 1 March 2019.
Usage of multicompartmental compliance aids
- 94% of pharmacies dispense medication in multi-compartment compliance aids (MCAs);
- 113 is the average number of patients per pharmacy supplied with MCAs;
- 28% of pharmacies complete a needs assessment for patients before commencing an MCA;
- 11% of pharmacies re-assess the needs of those patients who receive MCAs once a year.
You may also be interested in
GPhC writes to pharmacy teams after methotrexate dispensed with instruction to take once daily

Medicines commission calls for greater clarity on risk of suicidal behaviour from antidepressants
