Shutterstock.com
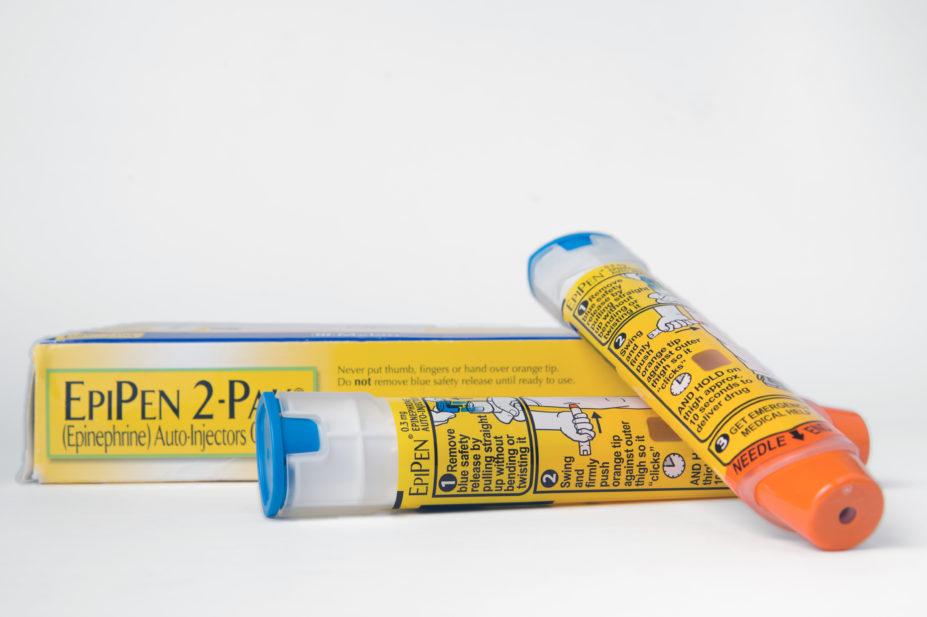
US drug manufacturer Mylan has agreed a US $465m settlement with the US Department of Justice in response to allegations that it overcharged the healthcare programme Medicaid for its anaphylaxis treatment EpiPen (epinephrine).
Although the pen is marketed as a branded product, Mylan was accused of classifying the product as a generic sale which meant it reduced the rebates that it had to pay to Medicaid under the terms of the drug rebate programme.
In a statement, the US Department of Justice said that the settlement was made to resolve claims that the company “violated the False Claims Act by knowingly misclassifying EpiPen as a generic drug to avoid paying rebates owed primarily to Medicaid”.
Acting US attorney William D Weinreb said: “Taxpayers rightly expect companies like Mylan that receive payments from taxpayer-funded programs to scrupulously follow the rules. We will continue to protect the integrity of Medicaid and ensure a level playing field for pharmaceutical companies.”
In a statement, Mylan stressed that the settlement “does not contain an admission or finding of wrongdoing”.
Mylan’s chief executive officer Heather Bresch said: “Bringing closure to this matter is the right course of action for Mylan and our stakeholders to allow us to move forward.”
As part of the deal, Mylan has signed a Corporate Integrity Agreement with the Office of Inspector General of the Department of Health and Human Services and agreed to reclassify EpiPen auto-Injector for purposes of the Medicaid drug rebate program ‘and pay the rebate applicable to innovator products effective as of April 1, 2017’.
The settlement was signed by two of Mylan NV subsidiaries — Mylan Inc. and Mylan Specialty LP.
You may also be interested in

What are the views of the RPS and Pharmaceutical Press on AI training using copyrighted materials?

Government should consider ways to prevent ‘inappropriate overseas prescribing’ of hormone drugs, review recommends
