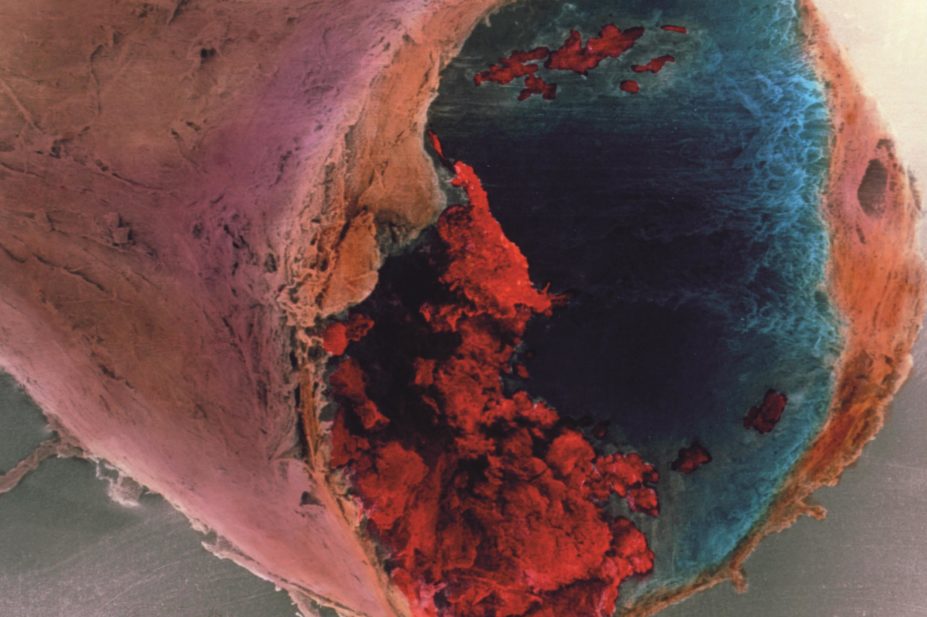
PROFESSOR P.M. MOTTA, G. MACCHIARELLI, S.A NOTTOLA / SCIENCE PHOTO LIBRARY
Anti-platelet therapy is an important strategy for patients with coronary artery disease, who are at risk of coronary thrombosis. However, currently available therapies – such as clopidogrel – have a slow onset and are irreversible.
Researchers report in Arteriosclerosis, Thrombosis and Vascular Biology (online, 17 December 2015[1]
) on a first-in-man study of a new anti-platelet drug called PZ-128. It has a novel mode of action, targeting the protease-activated receptor-1.
PZ-128 was administered to participants with coronary artery disease or associated risk factors via intravenous infusion for 1-2 hours. The infusion resulted in a dose-dependent and rapid inhibition of platelet aggregation. However, this was reversible, with platelet function recovered within 24 hours of the last 0.5mg/kg dose.
The drug will now be tested in a phase II study of patients with acute coronary syndromes, including those undergoing percutaneous coronary intervention.
References
[1] Gurbel PA, Bliden KP, Turner SE, et al. Cell-Penetrating Pepducin Therapy Targeting PAR1 in Subjects With Coronary Artery Disease. Arteriosclerosis Thrombosis & Vascular Biology 2016; 36: 189-197. doi: 10.1161/atvbaha.115.306777