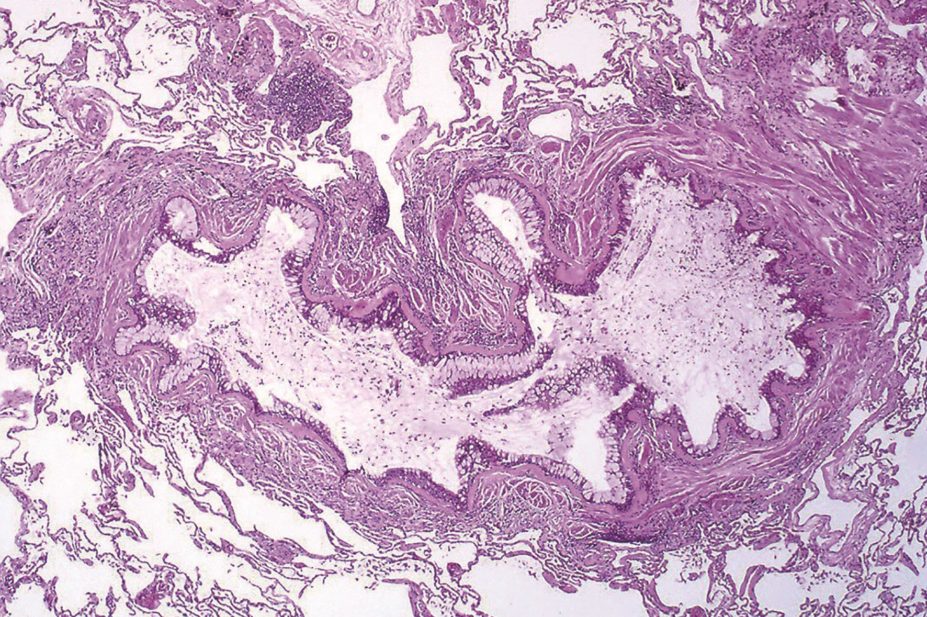
Yale Rosen / Wikimedia Commons
An early trial of the experimental oral asthma therapy fevipiprant has shown it is effective in reducing airway inflammation among patients with persistent eosinophilic asthma.
Patients with this subtype of asthma, involving the infiltration of inflammatory cells into the airways, often have poorly controlled disease in spite of steroid treatment.
The researchers say fevipiprant, a drug that targets this inflammation, could become the first new oral therapy for asthma in 20 years.
“We already know that using treatments to target eosinophilic airway inflammation can substantially reduce asthma attacks,” says Christopher Brightling, clinical professor in respiratory medicine at the University of Leicester, who led the study, published in The Lancet Respiratory Medicine
[1]
(online, 5 August 2016).
“This new treatment, fevipiprant, could likewise help to stop preventable asthma attacks, reduce hospital admissions and improve day-to-day symptoms making it a ‘game changer’ for future treatment.” However, the researchers note that longer term studies will be needed to assess the impact of fevipiprant on asthma exacerbations.
The study involved 61 people with persistent, moderate-to-severe asthma with evidence of eosinophilic airway inflammation. Patients were randomly assigned to receive fevipiprant twice daily or placebo for 12 weeks, in addition to conventional treatment including inhaled corticosteroids.
From baseline to 12 weeks, fevipiprant significantly reduced eosinophilic inflammation compared with placebo; sputum eosinophil percentage reduced from an average of 5.4 to 1.1 in the fevipiprant group (a 78% reduction) and from 4.6 to 3.9 (a 22% reduction) in the placebo group.
Bronchial biopsy samples from 26 patients at week 12 also showed a greater reduction in bronchial submucosal eosinophil numbers from baseline in fevipiprant-treated patients compared with placebo-treated patients.
The researchers also report some evidence that the study drug improved clinical symptoms, with significantly greater improvement from baseline in asthma-related quality of life and post-bronchodilator forced expiratory volume in one second among the fevipiprant group. Asthma control was also improved in a subgroup of patients with uncontrolled asthma who received fevipiprant compared with those who received placebo. The rate of adverse events was also comparable between the two groups.
Fevipirant is an antagonist of prostaglandin D2 receptor 2, which is thought to be involved in mediating eosinophilic inflammation of the airways. Evidence from other treatments, such as the monoclonal antibody mepolizumab, have indicated that targeting eosinophilic inflammation could be an effective strategy for reducing asthma exacerbations in patients with elevated eosinophil levels.
Ian Adcock, professor of respiratory cell and molecular biology at the National Heart and Lung Institute, Imperial College London, who was not involved in the research, says that if the results from the trial are repeated in a larger multi-centre study, fevipiprant will become an important treatment for patients with this type of asthma, particularly as it is a tablet and much cheaper than some newer antibody therapies developed for these patients.
“Future areas of development are required, however, including the need to fully understand the mechanism of drug action and the need to develop smarter biomarkers for the subjects who may respond best to these treatments,” he says. He adds that it will also be important to develop new therapies for the 50% of patients with severe asthma who do not have eosinophilic asthma.
Samantha Walker, director of research and policy at the charity Asthma UK, says that while the study findings show promise for fevipiprant, they should be treated with “cautious optimism”.
“The possibility of taking a pill instead of using an inhaler will be a very welcome one among the 5.4 million people in the UK with asthma, particularly as this study focused on people who develop the condition in later life, some of whom we know can struggle with the dexterity required to use an inhaler,” she says.
“More research is needed and we’re a long way off seeing a pill for asthma being made available over the pharmacy counter,” adds Walker. “But it’s an exciting development and one which, in the long term, could offer a real alternative to current treatments.”
References
[1] Gonem S, Berair R, Singapuri A et al. Fevipiprant, a prostaglandin D2 receptor 2 antagonist, in patients with persistent eosinophilic asthma: a single-centre, randomised, double-blind, parallel-group, placebo-controlled trial. The Lancet Respiratory Medicine 2016. doi: 10.1016/S2213-2600(16)30179-5