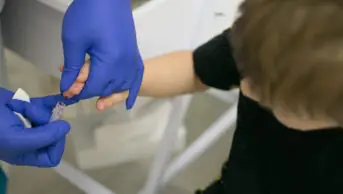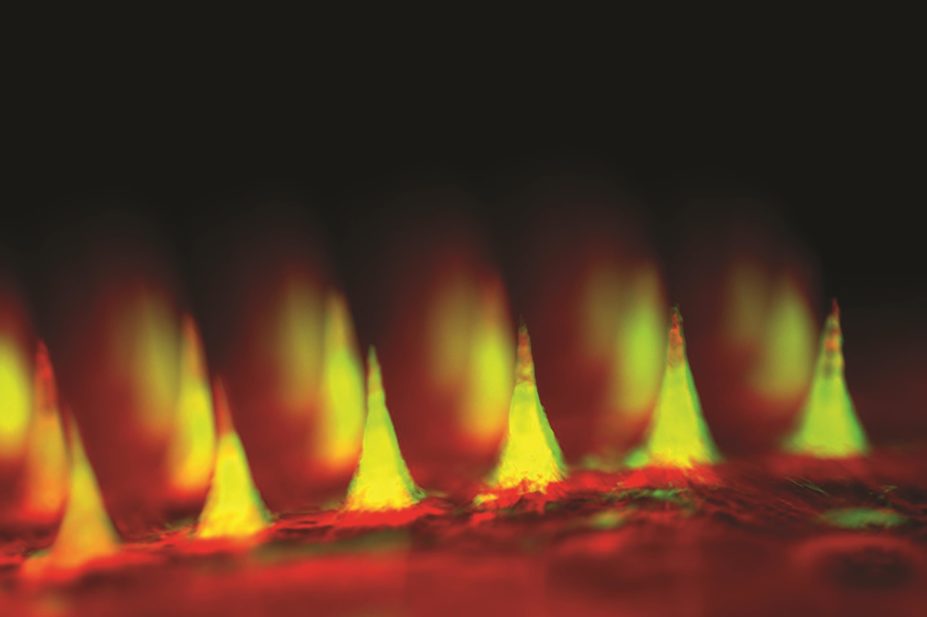
Courtesy of Dr Zhen Gu
A ‘smart insulin patch’ that detects increases in blood sugar and responds by releasing necessary doses of the hormone could mean an end to painful injections, say US researchers.
The small patch, designed to emulate pancreatic beta cells, comprises more than a hundred microneedles containing insulin and glucose-sensing hormones.
Trials of the patch in mice with type 1 diabetes show it can keep blood sugar levels low for up to nine hours by mimicking the vesicles that store insulin within beta cells, researchers from the University of North Carolina (UNC) outline in the Proceedings of the National Academy of Sciences
[1]
on 22 June 2015.
The microneedles, made from hyaluronic acid and held on a silicon strip, are loaded with microscopic balls of insulin. These artificial vesicles also hold glucose oxidase enzyme, which when processing high levels of glucose creates a hypoxic environment causing the vesicles to split, releasing the insulin inside.
In tests of the new technology, one group of mice were given a standard insulin injection while the other was treated with the microneedle patch. As expected in the mice receiving standard insulin treatment, blood glucose dropped to normal after the injection before quickly climbing back up into the hyperglycaemic range.
By contrast, in mice treated with a patch, blood glucose fell to normal levels within 30 minutes and remained that way for several hours. The researchers also found they could alter the response by varying the dose of enzyme contained in the microneedles, suggesting that if similar success is seen in human trials, the patch could be tailored specifically for the patient.
By carrying out a glucose tolerance test, the team also showed the patch was responsive to glucose challenge but also avoided the potential for hypoglycaemia attendant with frequent insulin injections. It is believed to be the first insulin delivery system based on sensitivity to hypoxia rather than changes in pH, which seems able to produce a faster response, the researchers report. It produces a “convenient, painless and continuous administration of insulin”, they conclude. With further work the researchers hope to develop a patch in humans that will last several days.
Co-author Zhen Gu, from the UNC joint biomedical engineering department, says the patch is fast, easy to use and is made from non-toxic, biocompatible materials.
“The whole system can be personalised to account for a diabetic’s weight and sensitivity to insulin so we could make the smart patch even smarter.”
Victoria Ruszala, specialist pharmacist in diabetes and endocrinology at North Bristol NHS Trust, says that if proven in humans the patch would be a beneficial technological advance for those taking insulin.
“It’s not actually the injection that’s the problem for patients, it’s the continual decision-making which is wearing,” Ruszala adds. “Because the patch releases insulin in response to blood glucose you wouldn’t have to be thinking about it in the same way.”
Richard Elliott, research communications manager at Diabetes UK, says the charity will continue to watch the progress made with the technology with interest.
“Further studies and clinical trials in humans will be needed to find out if this promising new approach might help to simplify existing techniques for managing blood glucose – which we know can be a painful daily chore for millions of people with diabetes.”
References
[1] Yu J, Zhang Y, Ye Y et al. Microneedle-array patches loaded with hypoxia-sensitive vesicles provide fast glucose-responsive insulin delivery. PNAS 2015. doi:10.1073/pnas.1505405112.