Fei Li
Translocator protein (TSPO) is ubiquitous throughout our bodies. It transports cholesterol and other molecules across the membranes of mitochondria — the power stations inside our cells — and has also been implicated in Alzheimer’s disease, various cancers and cardiovascular disease. But scientists are still in the dark about how TSPO works, and its precise role in disease.
Now, two research groups have independently solved the structures of similar TSPOs from bacteria, offering clues about how TSPO functions that could spur the development of targeted drugs and better brain-imaging agents.
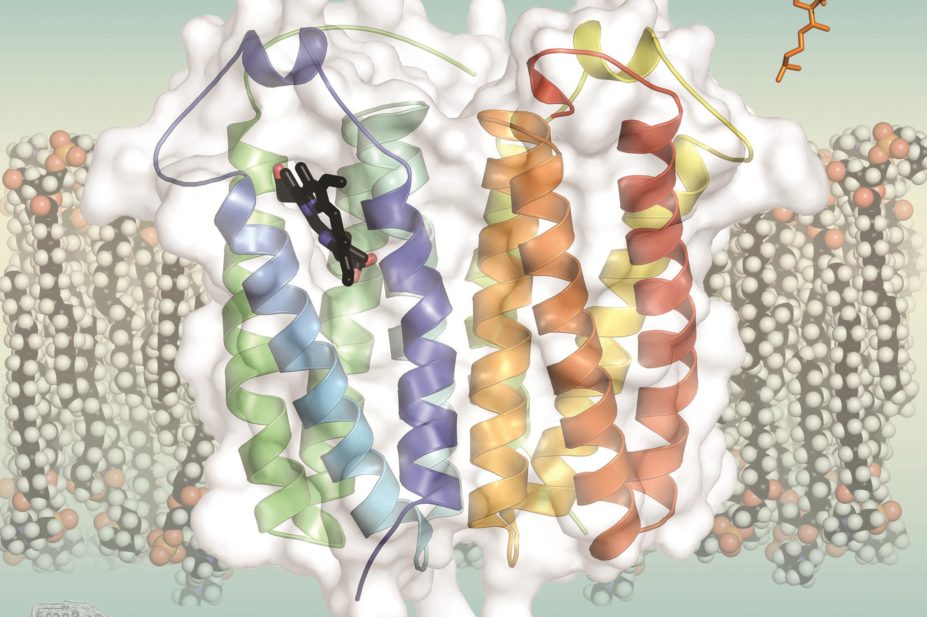
The team led by Wayne Hendrickson, a biochemist at Columbia University in New York, studied TSPO from Bacillus cereus, and a second group, led by Shelagh Ferguson-Miller at Michigan State University in East Lansing, used TSPO from Rhodobacter sphaeroides. The X-ray crystal structures, published this week in Science, are almost identical, and are very similar to those found in humans[1]
[2]
. “Knowing the structure in detail gives us an angle on the structure-based design of therapeutic compounds in future,” says Hendrickson.
“When we have two individual groups coming up with the same structure like this, it’s very gratifying,” says Chris Tate, a membrane-protein biochemist at the MRC Laboratory of Molecular Biology in Cambridge, UK, who was not involved in the work. “They’re important in determining the function of the protein.”
The structures show that TSPO contains five helical sections that span the membranes of mitochondria or cells, and reveal how ring-shaped molecules called porphyrins bind to the protein. Hendrickson’s team also found that TSPO could break down porphyrin into a blue compound they call bilindigin, which is similar to the biliverdin and bilirubin that colour bruises green and yellow.
Porphyrins generate reactive oxygen species, which can damage cells in many different ways. But biliverdin can block these reactive oxygen species, and Hendrickson speculates that bilindigin might do the same. “It’s our idea that the TSPO process might contribute to the control of oxidative stress,” he says. “Maybe that accounts for why it has been implicated in such a wide range of diseases.”
Furthermore, TSPO is also known to bind strongly to benzodiazepine drugs, such as diazepam (Valium). “It could be that some of the side effects of these compounds work through TSPO,” says Hendrickson. Diazepam’s primary target is the GABAA receptor in the brain, whose structure was solved[3]
in 2014. “Now we have the possibility of designing compounds that go to one or the other, but not both,” says Hendrickson.
The structures could also help scientists develop better ways to diagnose disease in the brain. Neurodegenerative conditions such as Alzheimer’s disease can trigger the formation of excess TSPO in certain cells, and researchers are now beginning to use the protein as a biomarker for inflammation in the brain. By using a radioactive version of a compound called PK11195, which binds strongly to TSPO, researchers have used positron emission tomography (PET) scans to highlight areas rich in TSPO. But the PK11195 ligand is not ideal because it can bind to other proteins. So understanding the structure of TSPO means that “we could design better PET ligands”, says Hendrickson.
References
[1] Guo Y, Kalathur RC, Liu Q et al. Structure and activity of tryptophan-rich TSPO proteins. Science 347, 551–555 (2015). doi: 10.1126/science.aaa1534.
[2] Li F, Liu J, Zheng Y et al. Crystal structures of translocator protein (TSPO) and mutant mimic of a human polymorphism. Science 347, 555–558 (2015). doi: 10.1126/science.1260590.
[3] Miller PS and Aricescu AR. Crystal structure of a human GABAA receptor. Nature 512, 270–275 (2014). doi: 10.1038/nature13293.