Barbara Johnston
A paper-based tool that can be used to detect counterfeit or degraded medicines and costs around US$1 is effective in detecting poor quality samples of the injectable antibiotic ceftriaxone.
The antibiotic, which is on the World Health Organization’s list of essential medicines, breaks down if stored at too high a temperature. In an experiment mimicking this degradation, the researchers subjected various samples of ceftriaxone to high temperatures and tested the products using both the card and liquid chromatography/mass spectroscopy. They showed that the degradation products were associated with specific colour changes on the card.
“We may be able to use the test card in the field to detect poor quality ceftriaxone before it is injected into a patient,” says Marya Lieberman, a specialist in analytical chemistry at the University of Notre Dame in Indiana, whose lab developed the test, known as the paper analytical device (PAD).
Lieberman, who presented the team’s results at the American Chemical Society annual meeting on 21 August 2016 in Philadelphia, Pennsylvania[1]
, says PADs can detect more than 60 types of medications, including the main classes of antibiotics, the four main antituberculosis drugs and antimalarials.
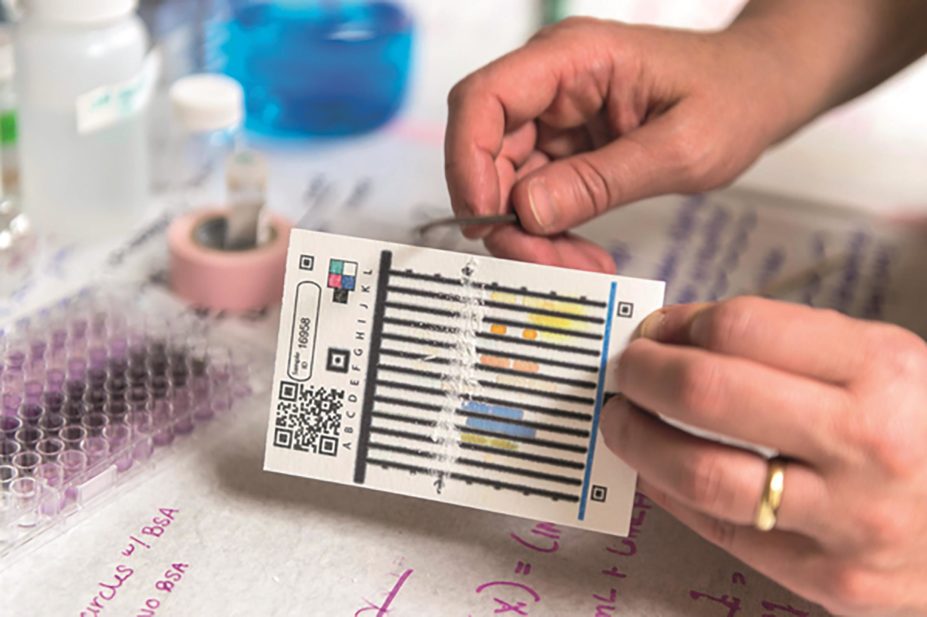
The PADs, which are the size of a playing card, contain 12 lanes loaded with reagents, which are divided by wax barriers. To carry out a test, a person spreads a small amount of a crushed tablet across the lanes before dipping the bottom of the card in water, which is then drawn up the lanes, bringing the reagent into contact with the sample.
Chemical reactions form a “colour barcode” that can be read by a trained operator or captured by a mobile phone camera and analysed by software. The result can be compared with known “barcodes” for high-quality samples.
The makers of the test say it could provide cheap screening for substandard drugs away from the lab in low-income countries, which have the greatest burden of false medications and the least ability to deal with them.
“Unscrupulous pharmaceutical vendors target countries that don’t have the technological infrastructure and regulatory resources to catch bad products in the marketplace,” says Lieberman. “We have developed a paper test card that a person can use outside a lab setting to quickly check a pill and see if it is fake.”
Falsified and substandard medications are a major public health problem in low-income countries. A recent study in 39 sub-Saharan countries estimated that 122,350 children under the age of five years died from malaria resulting from poor quality antimalarials in 2013[2]
. But the use of lab-based methods for detecting substandard medicines, such as infrared spectroscopy, nuclear magnetic resonance spectroscopy and high-performance liquid chromatography, is impeded by logistical and financial barriers in low-income countries.
“The PAD test cards are designed for field usability,” says Lieberman. She says that they are cheap, people can learn to use them in under half an hour and “the card results can be collected with a cell phone and read by an image analysis programme”.
“This enables us to archive test results and monitor drug quality over large regions, so regulators can get the data they need to keep bad quality products out of their markets,” she adds.
Paul Newton, director of the Lao-Oxford-Mahosot Hospital-Wellcome Trust Research Unit in Vientiane, Laos, says that the techniques developed by Lieberman’s lab are innovative and simple to use.
“They hold significant promise and it will be very interesting and useful to see how they perform in the field in practice,” he says. “They may be a useful screening device allowing pharmacists and medicine regulators to rapidly and simply screen for falsified medicines containing no active ingredients or wrong ingredients.”
Newton says that many methods are emerging to tackle the issue of poor-quality medications in low-income countries, and further investigation is needed to establish how best to put them to use.
“There are an increasing number of portable techniques and devices for detecting falsified medicines but there needs to be more research to identity their strengths and weaknesses, their cost-effectiveness and where in the medicine distribution chain they are best used.”
References
[1] Bliese S, Bendelsmith C, Hoehn J et al. Determining quality of antibiotics using paper analytical devices, high performance liquid chromatography, and portable X-ray fluorescence. Presented at the 252nd American Chemical Society National Meeting & Exposition; 21–25 August 2016; Philadelphia, Pennsylvania.
[2] Renschler JP, Walters KM & Newton PN. Estimated under-five deaths associated with poor quality antimalarials in Sub-Saharan Africa. The American Journal of Tropical Medicine and Hygiene 2015;92:119–126. doi: 10.4269/ajtmh.14-0725

