Shutterstock.com
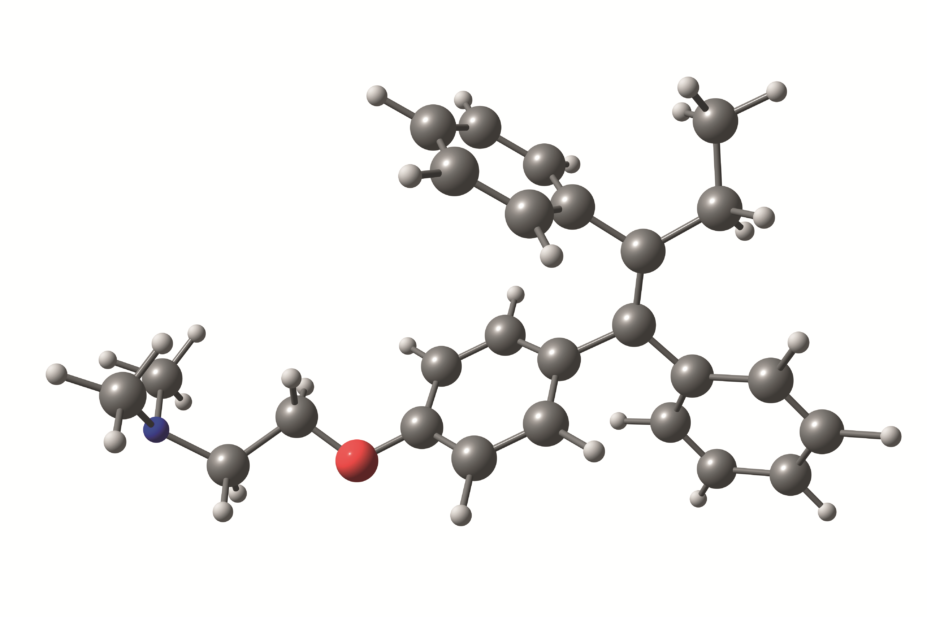
The International Breast Cancer Intervention Study found that tamoxifen reduces the risk of breast cancer by over 30% in high-risk women, but a third of participants discontinued treatment before the recommended five years were up.
To explore the reasons for this, researchers analysed the adherence rates among 4,279 UK women who were randomly assigned to placebo or tamoxifen in the trial.
They found that women who experienced nausea or vomiting in the first six months of treatment were around 45% less likely to be adherent for at least 4.5 years, whether in the tamoxifen or placebo arm. Headaches were also associated with non-adherence in the placebo arm, as were gynaecological symptoms in the tamoxifen arm.
Presenting their findings at the San Antonio Breast Cancer Symposium held from 6–10 December 2016, the researchers suggest that women may attribute menopause symptoms to tamoxifen and that better support and management of menopause symptoms could help improve treatment adherence[1]
.
References
[1] Smith SG, Sestak I, Forbes J et al. Menopausal symptoms as predictors of long-term adherence in the International breast cancer intervention study (IBIS-1). Presented at: San Antonio Breast Cancer Symposium; 6—10 December 2016; San Antonio, Texas. Available at: http://www.abstracts2view.com/sabcs/view.php?nu=SABCS16L_81 (accessed January 2017)