This content was published in 2010. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
Pharmacists are experts in medicines and, since chemistry is the defining science of pharmacy, they should also be experts in chemistry.
A drug’s journey from administration through to excretion requires it to endure a “lifetime” of existence in a seemingly chaotic chemical world.
Imagine for a moment we have a small molecule (our drug; molecular weight a few hundred g/mol) trying to survive in a world bursting with other much larger molecules (molecular weights in the order of a few thousand g/mol) such as enzymes, carbohydrates, lipids and nucleic acids, through which we expect it to pass unscathed (or at least maintain its pharmacological activity) and still be capable of fulfilling its role in restoring the balance towards good health.
Having an appreciation of the chemical structure of a drug, its properties and potential chemical vulnerability (due to the presence of its constituent functional groups) will therefore help pharmacists become an expert, not only in medicines, but also in the underlying scientific principles behind their actions and use.
It is the intention of this article to re-emphasise the importance of chemistry to pharmacists by using the selective serotonin reuptake inhibitor citalopram as an example.
Citalopram
Citalopram (and other SSRIs) selectively block the transport protein that is responsible for the uptake of serotonin from nerve synapses without affecting the levels of the other amine-containing neurotransmitters, noradrenaline and dopamine.
Citalopram’s three-dimensional shape (Figure 1) enables it to bind to its target protein and inhibit the protein’s natural function. A lack of serotonin at certain sites in the brain is thought to be related to depression and thus, by blocking its reuptake, the serotonin that is released has a longer duration of action to counteract the reduced levels in the brain.
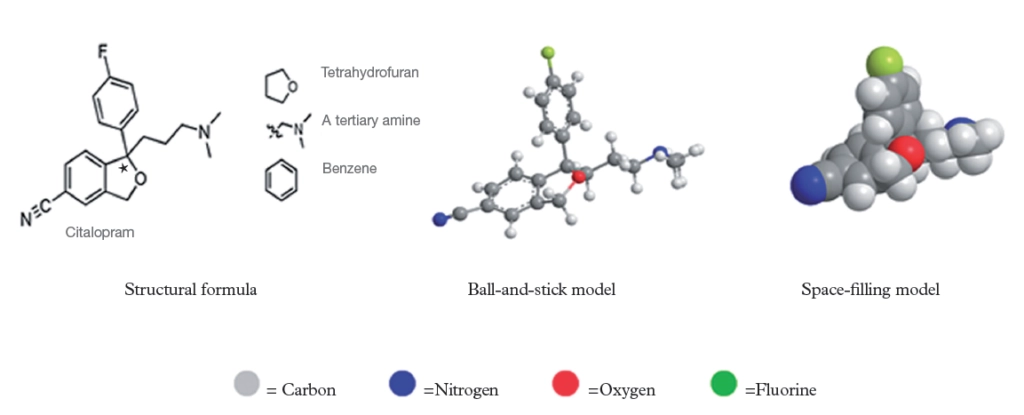
Citalopram was originally created in 1989 by the pharmaceutical company Lundbeck and it was initially marketed as a combination containing equal quantities of two enantiomers — a (so-called) racemic mixture. As can be seen from Figure 1, citalopram contains a carbon atom (denoted *) to which four different groups are attached: an oxygen-linked group, an amine-containing alkyl chain and two differently substituted benzene rings.
More specifically, citalopram contains a tertiary amine along with a tetrahydrofuran ring fused to a cyano-substituted benzene ring. The remainder of the molecule consists of a 4-fluorobenzene ring connected so as to create a chiral centre (denoted * in Figure 1).
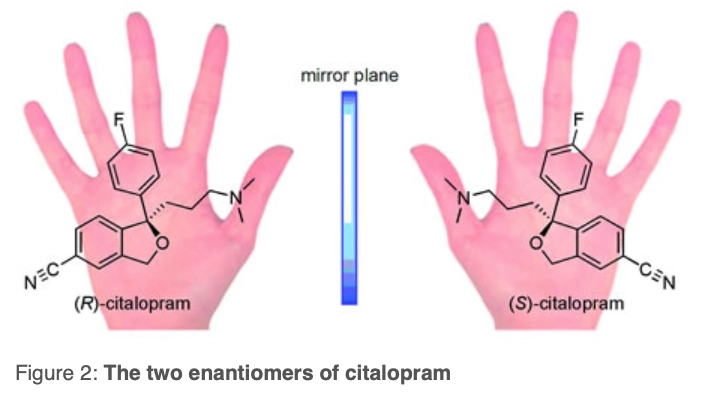
Such a condition allows us to denote that the molecule is chiral and, as such, we can refer to the chiral centre as either R or S — the two enantiomers of citalopram (Figure 2). Reflection of the molecule reverses the sense of chirality and changes the descriptor R→S or S→R.
Understanding this nomenclature helps to identify the individual enantiomers and enables us to discuss each in turn with other members of the scientific community without the risk of confusion as to the enantiomer being discussed — a critical distinction if one considers the sometimes tragic consequences of the differing pharmacological activities of enantiomers. Thalidomide is a classic example, where early tests showed the S-enantiomer was teratogenic while the R-enantiomer was effective as a sedative. Unfortunately, under physiological conditions, the drug racemises, giving equal amounts of both enantiomers.
Such a disparity in activity can be explained by the differing 3D shape of the two enantiomers. For example, the two mirror images (enantiomers) of citalopram in Figure 2, although having identical connectivity between the bonds, cannot be superimposed onto one another. As a consequence, they interact differently with other molecules that are chiral (see Panel below).
Panel: Chiral molecules
Our bodies are full of chiral molecules and, indeed, our hands are chiral (Figure 2), which explains why we cannot fit a left (chiral) glove on our right hand comfortably.
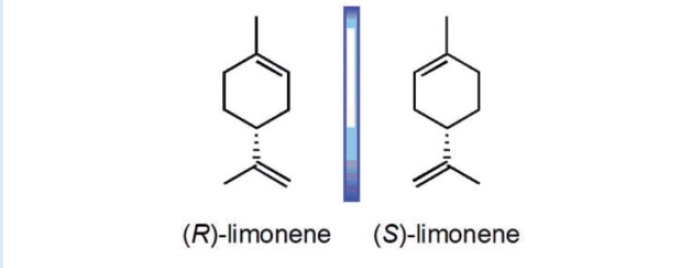
Similarly, the chiral olfactory receptors in our noses interact with enantiomers differently too.
For example, one enantiomer of limonene smells of lemons while the other smells of oranges, despite having the same bond connectivity.
They are non-superimposable mirror images.
Enzymes and receptors are chiral. They are proteins that are composed of many amino acids (often up to several thousand) and there are 20 different amino acids, 19 of which are chiral, which make up human protein sequences and thus impart chirality to the overall protein.
As such, when the enantiomers of a chiral drug, such as citalopram, come into contact with its target protein, one enantiomer will experience superior binding to the protein’s chiral active site because the shape of the two molecules will be complementary to each other.
The other enantiomer, on the other hand, which does not have the same 3D shape, cannot bind in the same manner and thus cannot elicit the same response from the protein because the molecular shapes are no longer as complementary.
Enantiomers and patents
Lundbeck’s patent for citalopram expired in 2003. This allowed other companies to produce generic versions. The general trend in pharmaceutical research in recent years has seen a move towards the preparation and marketing of single enantiomers of chiral drugs since it has been demonstrated that most enantiomers have different pharmacological activity, and pharmacokinetic and pharmacodynamic properties to each other. Accordingly, it is better to market a single enantiomer than a racemic mixture.
For racemic drugs that have been on the market for several years and are approaching the end of their patent life, switching to a single, more effective enantiomer can be financially rewarding. Arguably, a single enantiomer can be considered as a new invention and thus a new patent can be filed.
Nevertheless, pharmaceutical companies do have to prove that the new single enantiomer formulation is a better alternative to the original racemic mixture and that they did not know this at the time of the original patent application.
Consequently, Lundbeck was able to capitalise on the practice of chiral switching and released an updated formulation, escitalopram. It is called escitalopram because it is the S-enantiomer of citalopram and thus Lundbeck acquired a new patent for it.
Other drugs that have been the subject of chiral switching are the proton-pump inhibitor omeprazole (racemic) to esomeprazole (S-enantiomer), the non-steroidal anti-inflammatory drug ibuprofen (racemic) to dexibuprofen (S-enantiomer) and the antibiotic ofloxacin (racemic) to levofloxacin (S-enantiomer) among others.
However, it is no longer possible to patent a new drug in its racemic form today, since the issue relating enantiomers and their potentially differing biological activity is now an established fact.
The dose of citalopram required for the treatment of depression is 20mg once daily and, for escitalopram, is 10mg once daily. As can be seen, the daily recommended dose of escitalopram is half that of citalopram, a direct consequence of using a single enantiomer in the case of escitalopram as compared with a racemic mixture with citalopram whereby half of the mixture (ie, one enantiomer) is pharmacologically inactive.
Citalopram and MAOIs
Citalopram (and the other SSRIs, which include escitalopram) is contraindicated with monoamine-oxidase inhibitors (MAOIs). For an explanation of this adverse interaction, we need to understand a little more about the chemical role of monoamine oxidases.
Monoamine oxidases (MAOs) are natural enzymes that catalyse the oxidation of amines and contain flavin adenine dinucleotide as an essential “helper molecule”.
There are two forms of MAO in humans: MAO-A and MAO-B. The natural function of these monoamine oxidases is to catalyse the removal of an amine (deamination) from a molecule as part of the metabolism of these compounds, the product of the reaction being the corresponding aldehyde and amine, which can be subsequently acted on by other enzymes.
The significance of MAO-catalysed serotonin deamination is that its levels in the body are reduced. Consequently, if the MAO enzyme is inhibited (eg, by MAOIs), the levels of serotonin will subsequently increase. These increased levels, in combination with the increased levels due to the inhibition of serotonin uptake proteins, lead to the potentially life-threatening condition known as serotonin toxicity.
As a result, SSRIs and MAOIs should not be given together. Moreover, it is recommended that an SSRI should not be started until two weeks after stopping an MAOI, while an MAOI should not be started until at least one week after an SSRI has been stopped to enable to body to rid itself of the contraindicated drug.
Conclusion
It has been the intention of this article to demonstrate the close link between the science of chemistry, which underpins pharmacy, and pharmacy practice.
With regard to the example used here, a prior understanding of the chemical structure of citalopram, alongside knowledge of its mechanism of action, should have enabled the prediction of such contraindications as seen between SSRIs and MAOIs, a feature that further highlights the link between chemistry and pharmacy practice.
Glossary
Enantiomer One pair of molecular species that are mirror images of each other and are not superimposable (Figure 2)
Chiral centre A carbon atom to which four different groups are attached
Racemic mixture A mixture containing equal amounts of two enantiomeric species
Chiral switching Replacing a racemic drug with a more effective single enantiomer
Deamination The removal of nitrogen from a molecule