Introduction
Benzodiazepines, when used short-term, can be rapidly effective and relatively safe in a number of therapeutic areas. However, the long-term prescribing of these drugs is widespread. The British National Formulary recommends that benzodiazepines should not be used for more than a matter of a few weeks.[1]
Major clinical problems can occur with long-term use, eg, physical and psychological dependence, and they are recognised as being drugs of potential abuse. In addition, people may develop tolerance to their effects and gain little therapeutic benefit from chronic consumption. Withdrawal of benzodiazepines should always be gradual since abrupt withdrawal may induce a variety of adverse effects.[2]
Despite numbers of prescriptions steadily falling, there were still approximately 1.6 million benzodiazepine prescriptions issued in Scotland in 2004 (data on file, ISD).
There is clinical agreement that patients taking benzodiazepines for more than a short period should be regularly reviewed. Several published methodologies aimed at reducing benzodiazepine prescribing with simple interventions such as a letter to patients advising them to cut down or stop benzodiazepines have been shown to be successful.[3],[4],[5]
the past, pharmacists have provided successful supporting roles in assisting GPs to reduce benzodiazepine prescribing with similar projects, involving sending letters to patients encouraging them to reduce consumption themselves.[6],[7] Another involved the pharmacist inviting patients into a clinic to offer support to those being weaned off benzodiazepines.[8]
More recently, the role of the pharmacy technician has been highlighted in assisting with the reduction of benzodiazepine prescribing.[9]
Following discussions with the pharmacist and drug liaison worker, the GPs in a Paisley practice recognised there was a need to address their high benzodiazepine prescribing and a strategy was developed to assist with this. The model drew on knowledge and skills from medical, pharmaceutical and drug misuse services, and enabled practitioners to address different features of the benzodiazepine problem appropriately. The purpose of this project was to assess the impact of a novel, pharmacist-led strategy to reduce benzodiazepine prescribing for patients under 70 years of age. (The review of patients over 70 years old is currently ongoing.) The setting was a two-partner general practice of 3,400 patients in Paisley with a high level of deprivation and high prescribing rate of benzodiazepines. Ethical approval for the project was sought from NHS Argyll and Clyde Local Research Ethics Committee but was deemed not to be required.
Method
A new prescribing policy for benzodiazepines was agreed with the GPs. It involved:
- Initiation of a voluntary ban on the prescribing of benzodiazepines on repeat prescription (unless there were exceptional circumstances)
- A maximum 28-day prescribing interval
- Issuing all new diazepam prescriptions in the 2mg formulation
- Agreeing on a withdrawal (stepdown) protocol
The policy was promoted via posters displayed in the practice, and all practice staff were educated about effective repeat prescribing systems to minimise inappropriate issuing of prescriptions. The Renfrewshire Drug Service, local prescribing advisers and community pharmacists and Paisley Local Health Care Co-operative were consulted about the new prescribing policy.
The inclusion criteria were patients aged 70 years and under who were on a repeat benzodiazepine prescription. Patients with a specific psychiatric indication for benzodiazepines were excluded and any benzodiazepine not requested in the previous three months was inactivated from repeat prescription, since this was considered a reasonable period for a patient not to require a stepdown regimen. Patient records were reviewed to ascertain the indication for prescribing a benzodiazepine and unclear diagnoses or past medical histories were referred to the GP for clarification.
Patients considered appropriate for stepdown had their benzodiazepine inactivated from repeat prescription to ensure they would have the opportunity to be reviewed and not be able to continue ordering their repeat prescription.
The BNF issues guidance about withdrawing benzodiazepines, together with approximate equivalent doses of benzodiazepines, and this information was incorporated into the practice protocol.[1]
Each appropriate patient’s benzodiazepine was converted to an equivalent diazepam dose (in 2mg tablets) and then reduced at a rate considered appropriate (usually by 2mg every 14 days), but this depended on past medical history or on previous unsuccessful stepdown attempts. All prescriptions were issued on acute prescription, and annotated for dispensing on a daily or weekly frequency because of the large quantity of 2mg tablets on each prescription. Instalment dispensing stepdown regimens were generally kept to a maximum of eight weeks per prescription, but occasionally, prescriptions for 14 weeks were issued. More complex regimens were spread over two or more prescriptions, which were prepared in advance and retained in the case notes until required. The Panel (below) illustrates a typical stepdown example.
Panel: Case study example
- Mr A was prescribed diazepam 5mg three times daily for anxiety and nitrazepam 5mg at night for insomnia in relation to disturbances with neighbours nine months previously. He was now considered appropriate for stepdown.
- His repeat prescriptions were inactivated and total daily benzodiazepine dose converted to an equivalent diazepam dose, in this case diazepam 20mg daily (taken as diazepam 2mg tablets) in divided doses.
- Two acute prescriptions were generated. The first (given to Mr A at GP consultation) was for diazepam 2mg — nine daily for 14 days reduced by 2mg every 14 days to six daily for 14 days (first stepdown done). The stepdown time period was eight weeks, and was issued for 420 tablets (dispense weekly). The second prescription (retained in the case notes and available to Mr A after eight weeks) was for diazepam 2mg — five daily for 14 days and reduced by 2mg every 14 days to zero. The stepdown time period was 10 weeks, and was issued for 210 tablets (dispense weekly). Total stepdown period was 14 weeks.
- An accompanying stepdown chart was givent o Mr A, which detailed the administration times for diazepam.
A stepdown chart and prescription were prepared by the pharmacist and placed in the patient’s medical notes and a letter sent to the patient informing him of the stepdown policy and that he or she should visit the GP before starting the stepdown programme. The pharmacist also provided information on adjuvant medication and advised on any other pharmaceutical care issue.
At review, the GP conducted a structured interview outlining the new policy and introduced the patient into the stepdown regimen. This included general support and encouragement and focused on measures to reduce anxiety and information about non-pharmacological ways of coping with stress or insomnia. Patients requiring psychological support were referred accordingly. Patients were not forced to stepdown off their benzodiazepine, but were motivated by the advice given about long-term hazards of benzodiazepines together with reassurance and support. A copy of the proposed stepdown regimen was given to the patient for his or her own information. Any patient invited by letter and who did not attend for benzodiazepine review within two months of their last prescription was presumed not to require a stepdown regimen and had their stepdown prescription cancelled.
Data collected included patient age and sex, drug, frequency of previous requests, whether a stepdown prescription was collected and whether a stepdown regimen was tolerated. The primary outcome measure obtained from GPASS (General Practice Administration System for Scotland), the general practice computer system, was the number of patients aged 70 or under on a repeat prescription for a benzodiazepine between baseline (April to June 2002) and the end of the study period (April to June 2005). Secondary outcome measures, obtained from Prescribing Information System for Scotland (PRISMS), were the total number of benzodiazepine tablets prescribed, together with the practice prescribing rates (prescriptions per 100 patients per quarter) compared with the Scottish average.
Results
We identified 369 patients aged 70 years or under as receiving a benzodiazepine on repeat prescription. Fifty-five per cent were female and average age was 49 years (range 16–70). Two hundred and six patients (56 per cent) were sent a letter inviting them to attend for review and, two months after sending each letter, the stepdown uptake was evaluated. One hundred and fifty-one patients (73 per cent) had collected their stepdown regimen, 53 (26 per cent) did not and for two patients (1 per cent) it was decided by the GP not to initiate a stepdown regimen. Twenty-four patients who received a stepdown regimen had to be restarted on their original (or a lower) benzodiazepine dose. One hundred and sixty-three patients were not sent a stepdown letter (see Table 1). Of these, the GPs or pharmacist considered 59 patients with psychiatric recommendation or concurrent social/family problems at the time of the study inappropriate for stepdown. Four patients prescribed ad hoc chlordiazepoxide for alcohol problems had their repeat inactivated and were invited into the surgery for review by the GP, who reduced the prescription.
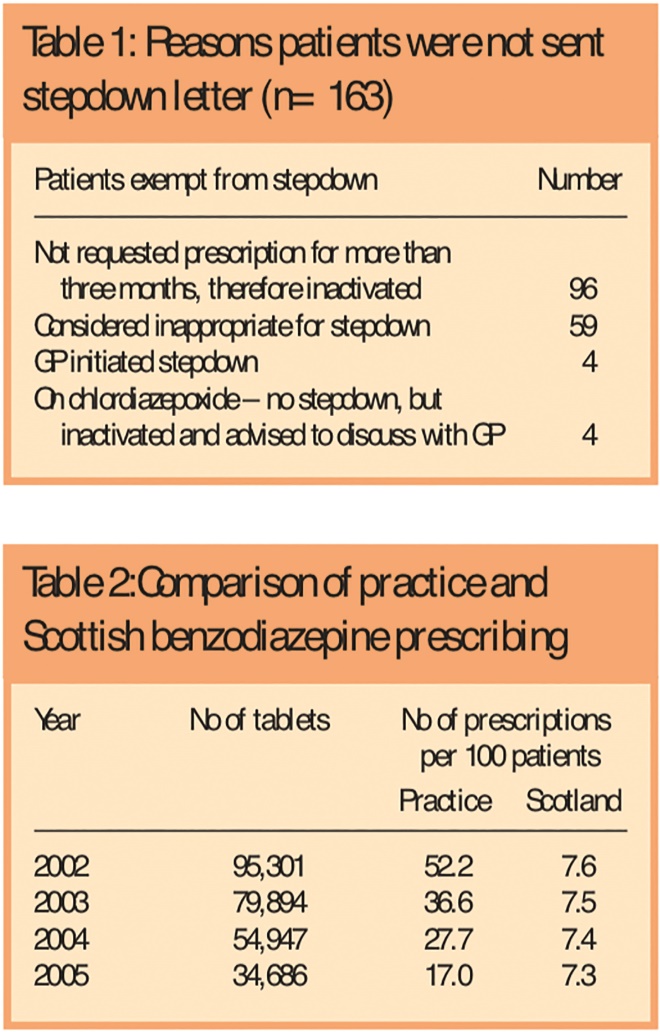
Tables 1 and 2: Reasons patients were not sent stepdown letter (n=163), and Comparison of practice and Scottish benzodiazepine prescribing
Of the initial 369 patients on repeat benzodiazepine prescription, 85 (23 per cent) remained on their repeat benzodiazepine prescription at the end of the study period. Table 2 shows the benzodiazepine prescribing rate, which had decreased by 67 per cent from 52.2 prescriptions/100 patients (Scotland 7.6/100) to 17/100 (Scotland 7.3/100). It also shows the total number of benzodiazepine tablets prescribed was reduced by 64 per cent from 95,301 tablets to 34,686 tablets per quarter.
Discussion
In April–June 2002, this GP practice had a benzodiazepine prescribing rate approximately seven times higher than the Scottish average. The reasons for this high prescribing rate are unclear, but it may have been due to a number of possibilities. First, the GPs could have had difficulty in changing their prescribing habits especially in a deprived population with the high workload and demands on their time. Secondly, there may have been a lack of awareness or even a fear of how to reduce benzodiazepine prescribing. However, initial GP concerns of being inundated with problematic patients turned out to be unfounded and the overall process was considered much easier than first thought. Despite potential barriers, the practice was able to achieve a major reduction in its rate and volume of benzodiazepines prescribed.
The fact that the practice recognised it had a high benzodiazepine prescribing rate was an important first step in being able to reduce it. The pharmacist provided support with regular feedback and meetings, allowing an opportunity to address prescribing at variance with the protocol, eg, patients having benzodiazepines added to repeat prescriptions inadvertently.
The pharmacist’s management of the stepdown process was key to ensuring a successful project, since his role was to develop the agreed protocol (in conjunction with the drug liaison worker), review the case notes, prepare the stepdown regimens, calculate quantities on prescriptions and send letters to patients. This ensured the GP had the maximum time to discuss benzodiazepine issues during a normal consultation. The pharmacist’s understanding of other disease states and concomitant drug therapy were also important in determining alternative benzodiazepine stepdown strategies, eg, if a patient was considered appropriate for stepdown but suffering from alcohol dependence or epilepsy, he or she was stepped down using smaller dose increments or over a longer period. In the future, it is envisaged that the ongoing work in the project could be more efficiently managed with support from a pharmacy technician. For example, once the pharmacist had decided upon a suitable stepdown regimen for a patient, the technician could prepare the stepdown regimens, calculate quantities on prescriptions and send letters to patients. This could free pharmacist time to focus on other clinical issues.
Ninety-six patients (26 per cent) had not requested their prescription for three months or more and this number of patients would suggest poor control over the repeat prescribing system with a tendency for GPs to put drugs directly onto repeat prescription instead of issuing an acute prescription. The change to new prescriptions being issued on acute prescription allowed more control over prescribing. There was also a lack of a repeat medication review within this patient group and Zermanski has highlighted this as a major clinical weakness in any repeat prescribing system.[10]
The secondary outcome data assessed the impact of the project as a whole and not just the patients reviewed. These data included prescribing changes induced by the protocol, prescribing for stepdown regimens in progress, ongoing patients who were excluded or who failed to tolerate the stepdown and patients outside the age criteria.A 64 per cent decrease in the number of benzodiazepine tablets prescribed was mainly caused by a reduction in prescribing of diazepam, nitrazepam and loprazolam and was achieved despite an increase in the number of diazepam 2mg tablets (as recommended in protocol). This large change was not only attributable to the stepdown regimens, but by a change in the prescribing habits of the GPs.
Patients were followed up to assess compliance with the stepdown regimens and patients who did not attend for benzodiazepine review within two months of their most recent prescription were presumed to have dropped out and the stepdown regimen was withdrawn. Interestingly, 53 patients (26 per cent) who were sent a stepdown letter did not make an appointment to discuss their prescription with the GP and therefore did not receive another prescription. The exact reasons for this are unknown, but it may have been that patients were not taking the drugs in the first place, eg, they were ordering the prescription and stockpiling the medicine, or passing the prescription to a third party — a well documented source of benzodiazepines diverted to the illicit market.2 It has also been reported that patients may sell part or all of their prescription.[11]
Subsequently, the policy of not prescribing blue diazepam 10mg was introduced because of its high street value compared with other diazepam strengths. The Renfrewshire Drug Service and Area Drug and Therapeutics Committee both supported this “no 10mg” policy and it was subsequently rolled out across NHS Argyll and Clyde.
Limitations
There were a number of recognised limitations to this project. Patients were not randomised into the project and compared against a control group, ie, there was no comparison between those who received a letter only compared with those who saw the GP. In 2004, NICE concluded that there was no compelling evidence of a clinically useful difference between the “Z” drugs (zopiclone, zolpidem, zapelon) and shorter-acting benzodiazepine hypnotics from the point of view of their effectiveness, adverse effects or potential for dependence or abuse.[12]
The extent of “Z” drug prescribing was not investigated in the project, but it is acknowledged that some patients may have been switched from a benzodiazepine to a “Z” drug. On comparison between the study periods, there had only been a 3 per cent increase in “Z” drug prescribing rates, compared with a 17 per cent increase across Scotland but, since the practice rate was still more than double the Scottish average, it is the subject of further work within the practice. Furthermore, the tertiary outcome of GP workload analysis, referrals to counselling or other support network and a patient perspective on the stepdown experience may also have provided useful data on the effectiveness of the project. However, again, these outcomes did not form part of this project, but could be investigated in a further study.
Conclusions
Despite well-documented problems associated with long-term benzodiazepine prescribing, poor control of repeat prescriptions together with multiple other factors can compound the problem of over-prescribing of benzodiazepines. This methodology developed from simple guidance from the BNF, provided a quick and effective method to assist GPs in reducing their benzodiazepine prescribing and was perceived by them to be a successful and worthwhile investment in time and resources.
The pharmacist provided a pivotal role in this project in the development and implementation of a practice policy to promote rational drug therapy. GP motivation and good communication between members of the health care and practice administration team were also important contributing factors. The stepdown programme has subsequently been tried in other localities within NHS Argyll and Clyde and has been met with varying degrees of success, largely dependent on GP motivation.
Pharmacists are suitably skilled to support rational benzodiazepine prescribing, with services ranging from pharmacist-led clinics to offering simple advice and support at the point of dispensing. Within the developing technician’s role, there is an ideal opportunity to facilitate approved stepdown methodologies. In addition, amendments to legislation removing restrictions on benzodiazepine prescribing for supplementary prescribers, further initiatives to reduce over prescribing of benzodiazepines can be achieved in the future.
Acknowledgements
The authors thank Ian Hamilton, Ian Mason and the practice staff within St James Medical Centre, Paisley, for all their support and co-operation in this project. The advice of ISD Scotland, Anne Cockroft, mathematics and statistics department, University of Paisley, and Heather Ashton, psychopharmacology department, University of Newcastle. is also gratefully acknowledged.
This paper was accepted for publication on 6 January 2006.
About the authors
Ian Towle, MSc, MRPharmS, is Paisley locality pharmacist and Joan Adams, BSc, is drug liaison worker at Renfrewshire Drug Service.
Correspondence to: Ian Towle, Primary Care Services, Unit 64, Second Floor, Sir James Clark Building, Abbey Mill Business Centre, Seedhill, Paisley PA1 1TJ (e-mail: Ian.towle@nhs.net)
References
[1] British National Formulary. Edition 49. London: Royal Pharmaceutical Society of Great Britain and British Medical Association; 2005. pp173–4.
[2] Ashton CH. Toxicity and adverse consequences of benzodiazepine use. Psychiatric Annals 1995;25:158–65.
[3] Cormack MA, Sweeney KG, Hughes-Jones H, Foot GA. Evaluation of an easy, cost effective strategy for cutting benzodiazepine use in general practice. British Journal of General Practice 1994;44:5–8.
[4] Wylie RM. Reducing benzodiazepine usage. British Journal of General Practice 1995;45:327.
[5] Heather N, Bowie A, Ashton H, McAvoy B, Spencer I, Brodie J et al. Randomised controlled trial of two brief interventions against long term benzodiazepine use: outcome of intervention. Addiction Research and Theory 2004;12:141–54.
[6] Morgan JD, Wright DJ, Chrystyn H. Pharmacoeconomic evaluation of a patient education letter aimed at reducing long term prescribing of benzodiazepines. Pharmacy World and Science 2002;24:231–5.
[7] Berry S. Ways to reduce the use of anxiolytic and hypnotic drugs in care. Prescribing and Medicines Management 2002;(March/April):11–12.
[8] Andalo D. Pharmacists and GPs work together to support benzodiazepine withdrawal. Prescribing and Medicines Management 2004;(March):1.
[9] Orwin M, Williams H. Benzodiazepine withdrawal at a pharmacy technician-led clinic. Prescribing and Medicines Management 2005;(March):3.
[10] Zermanski AG. Who controls repeats? British Journal of General Practice 1996;46:643–7.
[11] Lader M.H. Limitations on the use of benzodiazepines in anxiety and insomnia: are they justified? European Neuropsychopharmacology 1999;(9 December[Suppl ]): S399–405.
[12] National Prescribing Centre. Benzodiazepines and newer hypnotics. MeReC Bulletin 2005;15:17–20.