Key points:
- Treatment for initial acute myeloid leukaemia (AML) diagnosis is relatively standardised.
- Pre-clinical studies have identified several potential therapeutic targets such as CREB, Triad1, BCL-2 family members, JAK1/2 and STAT3.
- Completed clinical trials with agents targeting these specific proteins are limited, with results being more promising in the agents against BCL-2, JAK and STAT3.
- Relapsed/refractory disease remains the greatest challenge in AML therapy.
Introduction
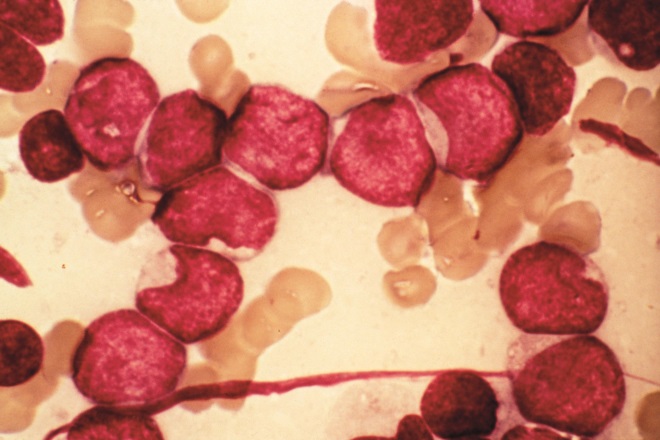
Source: Pr J Bernard / CNRI / Science Photo Library
Acute myeloid leukaemia (AML), pictured, is the most common type of adult acute leukaemia and the second most common type of paediatric leukaemia. AML results from aberrations in haematopoietic stem cells and, in particular, myeloid progenitor cells
Acute myeloid leukaemia (AML) is the most common type of adult acute leukaemia and the second most common type of paediatric leukaemia. AML results from aberrations in haematopoietic stem cells and, in particular, myeloid progenitor cells[1],
[2]
. The five-year survival rates for AML patients are inversely proportional to age at diagnosis; adults have five-year overall survival rates of approximately 30%[3]
, while children have a significantly better prognosis with five-year survival rates of approximately 60%[4]
. An additional subgroup consisting of younger adult AML patients (aged <60 years) exists, of whom approximately 65–85% achieve complete remission (CR)[3]
. An exception to this is the M3 subtype of AML, also known as acute promyelocytic leukaemia (APL), which is characterised by the t(15;17)(q22;q12) translocation that exhibits high sensitivity to all-trans-retinoic acid therapy (ATRA), arsenic trioxide, and chemotherapy, with more than 90% of patients achieving CR with current therapeutic approaches[5],
[6]
.
Prognosis in AML can be stratified based on the genetic characteristics of the disease[7],
[8]
. While significant progress has been made in the identification of disease pathogenesis[8]
, the most challenging aspect of AML treatment is the high occurrence of refractory disease[9]
; in patients newly diagnosed with AML, the rate of refractory disease ranges from 10% to 40%[9]
. Moreover, elderly, transplant-ineligible and chemotherapy-insensitive patients would benefit from decreased toxicity, which can be potentially obtained with more targeted therapies. This article discusses the current treatments for AML and the efforts to overcome relapsed disease, focusing on specific targets and their exploitation for future therapeutic development.
Sources
The sources referenced in this literature review include peer-reviewed primary clinical and pre-clinical studies, as well as relevant reviews examining the implications of targeting CREB, Triad1, BCL-2 family members, JAK and STAT3 in AML and any inhibitors developed against these targets. Manuscripts published in the past five to seven years were preferred. After initial identification of primary and review articles through searches in the PubMed interface for association of AML with the targets described here, the search was further focused on potential targeting agents. Upon identification of such agents, subsequent searches were conducted on their use in clinical trials, using the clinicaltrials.gov webpage as a source of ongoing and completed clinical trial information. Published results of completed clinical trials were then retrieved through PubMed and/or through published conference abstracts.
Discussion
Current therapeutic regimen for initial AML diagnosis
While progress has been made in the understanding of AML pathogenesis, the management of the disease, albeit standardised, exhibits some diversity in its execution[8]
. The standardised therapeutic regimen for AML comprises two phases: a remission induction phase and a consolidation therapy phase. The induction phase consists of chemotherapy administration combining two drugs, cytarabine (Ara-C) and an anthracycline, such as daunorubicin or idarubicin, aiming to reduce the leukaemic burden[9]
. The regimen is seven days of cytarabine and three days of the anthracycline[8],
[10]
. Cytarabine is a pyrimidine analogue, thereby inhibiting cellular proliferation by interfering with DNA synthesis[11]
. Anthracyclines, such as daunorubicin or idarubicin, are antibiotics that exert their anticancer properties through DNA intercalation and inhibition of topoisomerase II[12]
. Induction strategies may vary based on the specifics of each patient’s disease according to current clinical practice guidelines[13]
.
Following the induction phase, if CR is achieved, post-remission therapy is utilised, consisting of consolidation chemotherapy and/or haematopoietic cell transplantation (HCT). The treatment approach is based on the disease and patient characteristics, as well as availability of a matched sibling or alternative donor for HCT[13]
. However, as with other AML therapies, relapses occur even after post-remission therapy, underscoring the need for therapeutic targeting of refractory or relapsed AML.
Therapeutic regimen for refractory or relapsed AML
Approximately 10–40% of patients will not achieve CR, even after intense induction therapy. Treatment of refractory or relapsed AML remains one of the greatest challenges in the management of the disease[9]
. The main determinants of the likelihood of the leukaemia being refractory to induction therapy are the specific cytogenetics and molecular features of the disease of each patient, which can be further exploited in the development of targeted therapies[9],
[14]
. Salvage chemotherapy regimens are standard approaches for refractory or recurrent disease. However, there is major interest in the use of targeted approaches alone or in combination with chemotherapy.
Targeted therapies
Given the challenges in treatment of both primary and refractory/relapsed AML, studies have focused on a deeper examination of the development of myeloid malignancies, as a means to identify specific targets that could be exploited as alternatives to the current therapeutic regime. Targeted FLT3 and isocitrate dehydrogenase (IDH) inhibitors are examples of successful efforts of using targeted therapies in AML. Clinical trials with targeted FLT3 inhibitors report around a 50% response rate in AML patients harbouring the FLT3-ITD mutation[14],
[15],
[16]
. A phase I/II trial of ASP2215, a potent FLT3/AXL inhibitor, was recently completed in patients with relapsed/refractory AML, demonstrating significant clinical activity in patients with FLT3-mutated AML, with the overall response rate reaching 57% across all doses administered (clinical trial registration NCT02014558)[17]
. Additionally, a phase I/II trial of the multikinase inhibitor midostaurin, in combination with azacitidine, concluded that the greatest benefit was observed in AML and high-risk myelodysplastic syndrome (MDS) patients harbouring FLT3 mutations[18]
. More recently, a controlled, double-blind phase III trial focusing on younger adults with activating FLT3 mutations (internal tandem duplication or tyrosine kinase domain mutations) concluded that the addition of midostaurin to standard chemotherapy, for one year of maintenance therapy, significantly improved event-free survival and overall survival in this patient population[19]
. Additionally, FLT3-ITD-mutant AML patients, who had relapsed or were refractory to chemotherapy or had relapsed after haematopoietic stem cell transplantation, demonstrated a high response rate (composite CR [CRc]=43%) upon treatment with quizartinib (AC220), an oral FLT3 receptor tyrosine kinase inhibitor[20]
.
In addition to FLT3 inhibitors, agents selectively targeting the mutant forms of IDH1 and IDH2 have also been under development, as these mutations are detected in 15–20% of newly diagnosed AML patients[21],
[22]
. A phase I study of the IDH2 inhibitor AG-221 resulted in durable responses in refractory/relapsed AML patients with IDH2 mutations, with 18% of patients having CR, 15% having partial remission (PR) and 45% having stable disease (SD)[23]
. Furthermore, the mutant IDH1-selective inhibitor AG-120 has shown durable single agent responses in a phase I study, with 4/14 patients achieving CR, 2/14 bone marrow CR and 1/14 PR[24]
. Another IDH inhibitor, the pan IDH inhibitor AG-881, is also currently being evaluated in a phase I study (NCT02492737). The aforementioned examples of targeted therapy pave the way for additional targeted approaches for the treatment of AML. Below, CREB, Triad-1, BCL-2 family and JAK/STAT as subjects of targeted therapies and their utilisation in pipeline development and clinical trials in the context of AML are reviewed.
CREB
Pipeline development
Cyclic adenosine monophosphate response element binding protein (CREB) is a transcription factor regulating gene expression through both cyclic AMP (cAMP)-dependent and cAMP-independent pathways[25]
. The role of CREB in normal and malignant haematopoiesis has been investigated for the past decade, with a seminal study concluding that CREB functions as a proto-oncogene in haematopoiesis[26]
. Shankar et al. demonstrated that CREB transgenic mice developed a myeloproliferative disorder with aberrant bone marrow and spleen myelopoiesis[26]
. An earlier study examining the levels of CREB expression in bone marrow biopsies of AML patients compared with non-leukaemic patients established the association of CREB expression with malignant disease on account of the significant expression of CREB in leukaemic patients compared with non-leukaemic patients[27]
. Studies in AML preclinical models further demonstrated that CREB regulates a number of genes involved in signalling, oncogenesis, autophagy and DNA repair, with emphasis on the modulation of histone expression, particularly HIST1H3B
[28],
[29]
. Additionally, the inducible cAMP early repressor (ICER) was found to inhibit leukaemic phenotypes in vitro and in vivo through inhibition of specific CREB target genes, such as CDKN1A (p21) as well as the CREB gene itself[30]
, further supporting the potential use of CREB as a targeted therapy in a leukaemic environment.
Although CREB is a transcription factor, which presents a challenging class of proteins for the development of specific targeted inhibitors, several small-molecule inhibitors of CREB have been identified, such as Ro 31-122, NSC 12155, NSC 45576, naphthol AS-E phosphate, naphthol AS-E[31]
and XX-650-23[32]
. Of these compounds, naphthol AS-E phosphate has been predominantly examined for its anti-tumourigenic effects on a lung carcinoma cell line[33]
and on a metastatic breast-cancer line; however, it has not yet been tested in AML cell lines[33]
. XX-650-23, a small molecule inhibitor of CREB, disrupts the CBP-CREB interaction, thus impairing full activation of CREB-mediated gene transcription. Treatment of primary AML patient-derived bone marrow samples with XX-650-23 induced apoptosis and cell death, with the degree of apoptosis varying depending on the levels of CREB expression in the primary cells[32]
.
Triad1
Triad1 is an E3 ubiquitin ligase, whose expression is increased during myelopoiesis, particularly during granulocytic/monocytic differentiation of in vitro ATRA-treated AML cells, as well as in normal haematopoiesis[34]
. Triad1 has been specifically linked to AML upon discovery that it is a target gene of HoxA10[35]
. While studies on the role of Triad1 in AML or the development of specific agents targeting Triad1 are limited, the role of HoxA10 in myeloid lineage haematopoiesis is more established[36]
. Moreover, extensive research has been conducted on the pro-leukaemogenic effects of HoxA10 with its overexpression serving as a transforming event in mouse models of AML[37]
, and also through its cooperation with other proteins, such as the phosphatase SHP2[38]
and the transcription factor Cdx4[39]
, to induce myeloid leukaemia. Despite strong molecular evidence, HoxA10 inhibitors have yet to be developed.
BCL-2 family
Pipeline development
Members of the BCL-2 family of proteins are critical regulators of the apoptotic pathway, with members such as BCL-2 and BCL-XÂÂL functioning as anti-apoptotic, while members such as BAD and BAX as pro-apoptotic[40]
. With evasion of apoptosis being one of the hallmarks of cancer[41],
[42]
, a number of studies focused on understanding the role of the BCL-2 family of proteins in normal and malignant disease. Analysis of the expression of BCL-2-related genes in patients newly diagnosed with AML showed that levels of BCL-2 are higher in the CD34+ leukaemic cells than in their normal counterparts, with BCL-2 levels decreasing upon differentiation caused by induction therapy using ATRA in combination with Ara-C[43]
. High expression of BCL-2 was also detected in CD34+ cells of patients with minimal residual disease (MRD), further strengthening the association of BCL-2 with disease[43]
. BCL-2 also regulates the survival of AML cells with the leukaemogenic fusion MLL-AF9 through mediation of RUNX1[44]
. More recently, BCL-2 dependence in AML has been associated with mutations in IDH1 and IDH2 genes[45]
, providing additional evidence for the potential anti-leukaemic effects of BCL-2 inhibition.
Inhibition of the anti-apoptotic BCL-2 family members, which may contain the protein domains BH1-4, is caused by the binding of the BH3-only pro-apoptotic proteins to the hydrophobic groove of the pro-survival proteins[40]
. BH3-binding to specific pro-survival BCL-2 family members was exploited in the development of BH3-profiling, a methodology used to evaluate dependence of cancer cells to specific members of the BCL-2 family of proteins, thereby allowing for the administration of inhibitors to those specific proteins[46]
. As a result, BH3-profiling is readily used in studies focused on identifying small molecule BH3-mimetics to pharmacologically inhibit the pro-survival BCL-2 family members[47]
. One such putative BH3 mimetic, obatoclax mesylate, potentiated the pro-apoptotic effects of the multi-kinase inhibitor sorafenib in both in vitro and in vivo models of AML and, more importantly, replicated such combinatorial effects in primary AML cells, without affecting normal CD34+ cells[48]
.
ABT-737 is another BCL-2, BCL-XL and BCL-W inhibitor that synergised with FLT3 inhibitors in various AML cell lines and FLT3-mutant patient-derived AML blasts[49]
. ABT-737 also induced anti-leukaemic effects of bone marrow and peripheral blood-derived AML blasts, without a marked effect on the in vitro proliferation of CD34+/CD38– populations derived from two AML patients[50]
. Navitoclax (ABT-263) was also developed to inhibit BCL-2, BCL-XL and BCL-W; however, on account of dose-limiting thrombocytopaenia[51]
, it has not been further developed for leukaemia treatments, as leukaemia patients present with pre-existing thrombocytopaenia. To overcome the thrombocytopaenia caused by the more general BCL-2 family inhibitors navitoclax and ABT-737, a specific BCL-2 inhibitor, ABT-199, was developed[52]
.
Clinical trials
A phase I trial of obatoclax in haematologic malignancies, including patients with enrolling AML, chronic lymphocytic leukaemia (CLL), acute lymphocytic leukaemia (ALL) and myelodysplasia patients, found that obatoclax mesylate was well-tolerated. Furthermore, a patient with AML M4 with t(9;11)(p22;q23) maintained a CR for six months on trial (NCT00438178)[53]
. In a subsequent AML-restricted phase I/II study enrolling patients aged >70 years with previously untreated AML, obatoclax mesylate also demonstrated similar safety results; however, in this latter trial (NCT00684918), no CR was observed[54]
. The researchers speculate that the remarkable CR observed in the previous study was potentially caused by a mixed-lineage leukaemia (MLL) translocation of the patient. As a result, a study focusing on the effects of obatoclax mesylate in cell samples from young patients (aged <2 years) is currently under way, with specific interest in its activity on MLL-rearranged AML samples (NCT01150656). This study also aims to identify the mechanism of obatoclax-mediated cell death in MLL-rearranged AML through comprehensive gene and protein expression profiling and determine in vitro and in vivo combinations of obatoclax in the MLL-AML background.
A study of the BCL-2 specific inhibitor ABT-199 in patients with relapsed/refractory AML or AML patients who are unfit for intensive therapy has been completed, with the findings anticipated (NCT01994837). Two additional clinical trials of ABT-199 in combination with chemotherapy in AML patients are currently enrolling patients. One is a phase Ib study focused on patients aged 65 years or over who are not eligible for standard induction therapy because of comorbidity or other factors. It aims to evaluate the safety and preliminary efficacy of ABT-199 in combination with either azacitidine or decitabine in treatment-naïve patients who meet the study eligibility criteria (NCT02203773). The second study involves a combination of low-dose cytarabine with escalating doses of ABT-199, also in treatment-naïve subjects aged 65 years or over (NCT02287233).
JAK/STAT
Pipeline development
The JAK signalling pathway is activated upon stimulation by a variety of agents, including cytokines and growth factors stimulating receptor tyrosine kinases, leading to the activation of STATs. In haematologic malignancies, activation of the JAK/STAT pathway is common in myeloproliferative neoplasms (MPNs), particularly because of the prevalence of the JAK2 V617F mutant in diseases such as polycythaemia vera (PV), primary myelofibrosis (PMF) and essential thrombocythaemia (ET), with the majority of PV patients and around 50% of PMF and ET patients harbouring the mutation[55]
. All three myeloproliferative disorders could progress into AML, and so targeting the JAK pathway is of particular interest in AML[55]
.
STATs are downstream effectors of the JAK kinases, with cancer cells more dependent on STAT signalling compared with their normal counterparts, thus generating interest on developing STAT inhibitors for cancer therapy[56]
. It is noteworthy that while STAT1 and STAT5 are the major downstream effectors of the JAK2 V617F mutation in the induction of ET[57]
and PV[58]
respectively, phosphorylation of STAT3 is more prevalent in AML[59]
, especially in cases of AML resulting from the t(6;9) translocation[60]
. In de novo AML, presence of the JAK2 V617F mutation is rare, while constitutive activation of STAT3 is widely detected in all AML subtypes, independent of JAK-activating mutations[59],
[61]
. Additionally, constitutive activation of STAT3 in AML is associated with shorter disease-free survival of AML patients[61]
, providing additional supporting evidence that effective targeting of STAT3 in an AML background should also be explored, independent of JAK inhibition.
Currently, the FDA-approved JAK1/2 inhibitor ruxolitinib is being evaluated for antileukaemic effects in clinical trials, as discussed below. AZD1480 is another JAK2-specific inhibitor currently under development[62]
, exhibiting inhibition of tumour growth in preclinical models of solid tumours with high STAT3 activation[63]
. AZD1480 also shows antiproliferative effects in Hodgkin’s lymphoma[64]
and leads to inhibition of STAT3 activation and downstream effectors in paediatric tumour models[65]
and multiple myeloma[66]
.
A recent study using a murine AML model found regression of AML and reduction of the leukaemia-initiating potential of AML cells from mice upon the systemic administration of CpG-Stat3 siRNA[67]
, a small-interfering RNA-targeting STAT3 mRNA, which reduces STAT3 levels[68]
. Furthermore, the authors concluded that the STAT3-targeting was immunomodulatory, resulting in the increased expression of the immunostimulatory molecules MHC class II, CD40, CD80, and CD86, and an increase in plasma levels of pro-inflammatory cytokines such as IFN-γ, IL-12, and CXCL9/MIG[67]
, which indicates an increase in the immunogenicity of AML cells in vivo and generates a potent adaptive immune response.
Preliminary preclinical results using the small molecule inhibitor of STAT3, C188-9[69]
, in both AML cell lines and ex vivo paediatric AML patient samples suggest that this agent may have potential antileukaemic properties[70]
. However, more extensive studies with this molecule are needed to further confirm the observations made. A novel high-affinity STAT3 small molecule inhibitor, OPB-31121[71]
, inhibits phosphorylation of both STAT3 and STAT5 and demonstrated tumour-specific inhibition in a wide-range of haematopoietic malignancies, including AML, without suppression of normal haematopoietic cells[72]
. A phase I/II clinical study with OPB-31121 has been completed in patients with hepatocellular carcinoma (HCC) and the authors concluded that the inhibitor was well-tolerated[73]
.
AZD9150 (ISIS-STAT3Rx or ISIS 481464) is a next generation anti-sense oligonucleotide inhibitor of STAT3. A phase Ib clinical trial was conducted with this agent in patients with solid tumours or lymphomas with at least one prior systemic therapy (NCT01563302)[74]
. While there was no response in solid tumour patients, partial responses were observed in two of three diffuse large B-cell lymphoma (DLBCL) patients (>50% tumour reduction), leading to an ongoing phase II clinical study in patients with refractory lymphomas[74]
.
Clinical trials
Based on the documented progression of myeloproliferative disorders to AML, and with the former exhibiting high prevalence of JAK2 mutations, as well as the low incidence of JAK1 mutations in de novo AML[55],
[75]
, the effects of the JAK1/2 inhibitor ruxolitinib were examined in patients with refractory leukaemias[76]
. The aim of the study was to determine the activity of ruxolitinib in refractory/relapsed leukaemias and establish its safety profile within this patient group. This agent was well-tolerated and, interestingly, all 3/38 patients that achieved either CR or CR with insufficient recovery of blood counts (CRi) were patients with post-MPN AML, suggesting potential antileukaemic effects of ruxolitinib, especially in the aforementioned patient population[76]
.
As a follow-up to the results of the ruxolitinib study in patients with refractory/relapsed leukaemias[76]
, a phase I/II clinical study of ruxolitinib in combination with decitabine in AML patients is currently ongoing. The phase I cohort allows patients with refractory/relapsed AML to enrol, while the phase II cohort is restricted to post-MPN AML patients or MDS/MPN patients with more than 20% blasts (NCT02257138). Another study is aiming to investigate whether STAT3 activation could be used as a potential prognostic and therapeutic target in paediatric AML (NCT01057290).
Conclusion
While progress has been made in the molecular understanding of AML pathogenesis, a number of specific inhibitors for pathways discussed in this review have yet to be developed for assessment in clinical trials. Small molecule inhibitors of CREB have been recently developed, despite the low “druggability” of the transcription factor protein category; however, the studies suggesting potential anticancer properties of such agents were performed with treatments at high micromolar concentrations, presenting a potential limiting factor for further pharmaceutical development of these agents and their application in in vivo clinical studies.
Pertaining to clinical trials, BCL-2 inhibitors are of particular focus in AML as they could potentially be coupled with BH3 profiling[52]
, if the latter is approved for use as a screening tool of clinical trials. The BCL-2 inhibitor ABT-199 exhibits the highest specificity of all BCL-2 inhibitors discussed, and is currently evaluated in three clinical trials, alone and in combination with chemotherapy. Based on the strong preclinical evidence for antileukaemic effects, the results of these trials are anxiously awaited.
The JAK1/2 kinase inhibitor ruxolitinib demonstrated promising antileukaemic effects, especially in patients with post-MPN AML, and suggests a possibility that other JAK1/2 inhibitors currently under development may also yield such results. Downstream of JAK kinases, the prevalence of STAT3 activation in AML and the mode of action of the JAK2 inhibitor AZD1480 support examination of potential efficacy of this agent in AML, particularly in post-MPN AML[76]
. Additionally, as constitutive activation of STAT3 has been found to be present in the majority of AML, independent of JAK2 activating mutations, development of STAT3 inhibitors are of strong interest in AML treatment. One such anti-STAT3 therapy, AZD9150[74]
, is of potential interest. While it has yet to be tested in leukaemia patients, the promising efficacy observed in DLBCL[74]
, along with the strong evidence implicating STAT3 in AML leukaemogenesis, suggest it warrants further examination in AML models.
Notably, in addition to the pathways discussed in this review, efforts in developing targeted therapies for AML are also focused on various other targets such as bromodomains[77]
, histone deacetylases[78]
, MEK[79]
, DOT1L[80]
and CRM1[81]
.
In conclusion, advances have been made in the molecular characterisation of AML subtypes; however, the greatest challenge in the treatment of AML remains the effective targeting of relapsed disease. The standard therapy for newly diagnosed AML patients remains intensive chemotherapy with consideration of stem-cell transplantation, both of which harbour significant toxicities. Despite the toxicities, the disease is still at high risk of recurrence. In addition to patients with relapsed disease, development of new and targeted treatments is particularly required for elderly or transplant-ineligible patients, as all groups would benefit from decreased toxicity, which can be potentially obtained with more targeted therapies. Therefore, there is great interest in combining targeted agents with chemotherapy for some newly diagnosed patients with AML in the context of clinical trials. For refractory/relapsed disease, efforts have been directed in the development of targeted therapy, of which targeting of JAK/STAT, BCL-2 family members, CREB and Triad1 have been discussed in this article.
Eliza Vakana is a scientist, PhD graduate of Northwestern University, Robert H Lurie Comprehensive Cancer Center and division of hematology/oncology, Northwestern University Medical School Chicago, IL 60611. Jessica K Altman is associate professor in medicine — hematology/oncology at Robert H Lurie Comprehensive Cancer Center and division of hematology/oncology, Northwestern University Medical School, Chicago, IL 60611. Leonidas C Platanias is director of the Robert H Lurie Comprehensive Cancer Center and professor of medicine in the division of hematology/oncology, Northwestern University Medical School, Chicago, IL 60611 and staff physician at the department of medicine, Jesse Brown VA Medical Center, Chicago, IL 60612. Correspondence to: elizavakana@gmail.com
Acknowledgements:
The research of Leonidas C Platanias is supported by grants CA77816 and CA121192 from the National Institutes of Health.
Financial and competing interests disclosure:
Jessica K Altman has been on advisory boards for BMS, Novartis, Spectrum, Ariad, Seattle Genetics and Astellas within the past 36 months. Eliza Vakana and Leonidas C Platanias have no relevant affiliations or financial involvement with any organisation or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. No writing assistance was utilised in the production of this manuscript.
Reading this article counts towards your CPD
You can use the following forms to record your learning and action points from this article from Pharmaceutical Journal Publications.
Your CPD module results are stored against your account here at The Pharmaceutical Journal. You must be registered and logged into the site to do this. To review your module results, go to the ‘My Account’ tab and then ‘My CPD’.
Any training, learning or development activities that you undertake for CPD can also be recorded as evidence as part of your RPS Faculty practice-based portfolio when preparing for Faculty membership. To start your RPS Faculty journey today, access the portfolio and tools at www.rpharms.com/Faculty
If your learning was planned in advance, please click:
If your learning was spontaneous, please click:
References
[1] Gojo I & Karp JE. New strategies in acute myelogenous leukemia: leukemogenesis and personalized medicine. Clin Cancer Res. 2014;20:6233–6241. doi: 10.1158/1078-0432.CCR-14-0900
[2] Estey EH. Acute myeloid leukemia: 2014 update on risk-stratification and management. Am J Hematol 2014;89:1063–1081 doi: 10.1002/ajh.23834
[3] Sekeres MA. Treatment of older adults with acute myeloid leukemia: state of the art and current perspectives. Haematologica 2008;93:1769–1772. doi: 10.3324/haematol.2008.000497
[4] Rubnitz JE. How I treat pediatric acute myeloid leukemia. Blood 2012;119:5980–5988. doi: 10.1182/blood-2012-02-392506
[5] Lo-Coco F, Avvisati G, Vignetti M et al. Retinoic acid and arsenic trioxide for acute promyelocytic leukemia. N Engl J Med 2013;369:111–121. doi: 10.1056/NEJMoa1300874
[6] Coombs CC, Tavakkoli M & Tallman MS. Acute promyelocytic leukemia: where did we start, where are we now, and the future. Blood Cancer J 2015;5:e304. doi: 10.1038/bcj.2015.25
[7] Yohe S. Molecular Genetic Markers in Acute Myeloid Leukemia. J Clin Med 2015;4:460–478. doi: 10.3390/jcm4030460
[8] Rowe JM & Tallman MS. How I treat acute myeloid leukemia. Blood 2010;116:3147–3156. doi: 10.1182/blood-2010-05-260117
[9] Thol F, Schlenk RF, Heuser M et al. How I treat refractory and early relapsed acute myeloid leukemia. Blood 2015;126:319–327. doi: 10.1182/blood-2014-10-551911
[10] Dohner H, Estey EH, Amadori S et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood 2010;115:453–474. doi: 10.1182/blood-2009-07-235358
[11] Ramos NR, Mo CC, Karp JE et al. Current approaches in the treatment of relapsed and refractory acute myeloid leukemia. J Clin Med 2015;4:665–695. doi: 10.3390/jcm4040665
[12] Minotti G, Menna P, Salvatorelli E et al. Anthracyclines: molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol Rev. 2004;56:185–229. doi: 10.1124/pr.56.2.6
[13] National Comprehensive Cancer Network. Acute Myeloid Leukemia (Version 1.2015). Available at: http://www.nccn.org/professionals/physician_gls/PDF/aml.pdf (accessed 8 November 2015).
[14] Altman JK & Platanias LC. Acute myeloid leukemia: potential for new therapeutic approaches targeting mRNA translation pathways. Int J Hematol Oncol. 2013;2:243–250. doi: 10.2217/ijh.13.23
[15] Takahashi K, Kantarjian H, Pemmaraju N et al. Salvage therapy using FLT3 inhibitors may improve long-term outcome of relapsed or refractory AML in patients with FLT3-ITD. Br J Haematol. 2013;161:659–666. doi: 10.1111/bjh.12299
[16] Wander SA, Levis MJ & Fathi AT. The evolving role of FLT3 inhibitors in acute myeloid leukemia: quizartinib and beyond, Ther Adv Hematol. 2014;5:65–77. doi: 10.1177/2040620714532123
[17] Levis MJ, Perl AE, Altman JK et al. Results of a first-in-human, phase I/II trial of ASP2215, a selective, potent inhibitor of FLT3/Axl in patients with relapsed or refractory (R/R) acute myeloid leukemia (AML). J Clin Oncol 2015;33(abstract 7003).
[18] Strati P, Kantarjian H, Ravandi F et al. Phase I/II trial of the combination of midostaurin (PKC412) and 5-azacytidine for patients with acute myeloid leukemia and myelodysplastic syndrome. Am J Hematol. 2015;90:276–281. doi: 10.1002/ajh.23924
[19] Stone RM, Mandrekar S, Sanford BL et al. The Multi-Kinase Inhibitor Midostaurin (M) Prolongs Survival Compared with Placebo (P) in Combination with Daunorubicin (D)/Cytarabine (C) Induction (ind), High-Dose C Consolidation (consol), and As Maintenance (maint) Therapy in Newly Diagnosed Acute Myeloid Leukemia (AML) Patients (pts) Age 18-60 with FLT3 Mutations (muts): An International Prospective Randomized (rand) P-Controlled Double-Blind Trial (CALGB 10603/RATIFY [Alliance]), Proceedings of Conference The Multi-Kinase Inhibitor Midostaurin (M) Prolongs Survival Compared with Placebo (P) in Combination with Daunorubicin (D)/Cytarabine (C) Induction (ind), High-Dose C Consolidation (consol), and As Maintenance (maint) Therapy in Newly Diagnosed Acute Myeloid Leukemia (AML) Patients (pts) Age 18-60 with FLT3 Mutations (muts): An International Prospective Randomized (rand) P-Controlled Double-Blind Trial (CALGB 10603/RATIFY [Alliance]), Orlando, FL. 2015;57.
[20] Hills RK, Gammon G, Trone D et al. Quizartinib Significantly Improves Overall Survival in FLT3-ITD Positive AML Patients Relapsed after Stem Cell Transplantation or after Failure of Salvage Chemotherapy: A Comparison with Historical AML Database (UK NCRI data), Proceedings of Conference Quizartinib Significantly Improves Overall Survival in FLT3-ITD Positive AML Patients Relapsed after Stem Cell Transplantation or after Failure of Salvage Chemotherapy: A Comparison with Historical AML Database (UK NCRI data), Orlando, FL. 2015.
[21] Levis M. Targeting IDH: the next big thing in AML. Blood 2013;122:2770–2771. doi: 10.1182/blood-2013-09-522441
[22] Abbas S, Lugthart S, Kavelaars FG et al. Acquired mutations in the genes encoding IDH1 and IDH2 both are recurrent aberrations in acute myeloid leukemia: prevalence and prognostic value. Blood 2010;116:2122–2126. doi: 10.1182/blood-2009-11-250878
[23] Stein EM, DiNardo C, Altman JK et al. Safety and Efficacy of AG-221, a Potent Inhibitor of Mutant IDH2 That Promotes Differentiation of Myeloid Cells in Patients with Advanced Hematologic Malignancies: Results of a Phase 1/2 Trial, Proccedings of Conference Safety and Efficacy of AG-221, a Potent Inhibitor of Mutant IDH2 That Promotes Differentiation of Myeloid Cells in Patients with Advanced Hematologic Malignancies: Results of a Phase 1/2 Trial, Orlando, FL. 2015;57(abstract 323).
[24] de Botton S, Pollyea DA, Stein EM et al. Clinical Safety And Activity Of Ag-120, A First-In-Class, Potent Inhibitor Of The Idh1 Mutant Protein, In A Phase 1 Study Of Patients With Advanced Idh1-Mutant Hematologic Malignancies, EHA Learning Center 2015;100704.
[25] Delghandi MP, Johannessen M & Moens U. The cAMP signalling pathway activates CREB through PKA, p38 and MSK1 in NIH 3T3 cells, Cell Signal 2005;17:1343–1351. doi: 10.1016/j.cellsig.2005.02.003
[26] Shankar DB, Cheng JC, Kinjo K et al. The role of CREB as a proto-oncogene in hematopoiesis and in acute myeloid leukemia. Cancer Cell 2005;7:351–362. doi: 10.1016/j.ccr.2005.02.018
[27] Crans-Vargas HN, Landaw EM, Bhatia S et al. Expression of cyclic adenosine monophosphate response-element binding protein in acute leukemia. Blood 2002;99:2617–2619. doi: 10.1182/blood.V99.7.2617
[28] Pellegrini M, Cheng JC, Voutila J et al. Expression profile of CREB knockdown in myeloid leukemia cells. BMC Cancer 2008;8:264. doi: 10.1186/1471-2407-8-264
[29] Mitton B, Dutta R, Hsu Y et al. The role of pp90rsk-mediated CREB phosphorylation in acute myelogenous leukemia. Blood 2014;124:229.
[30] Pigazzi M, Manara E, Baron E et al. ICER expression inhibits leukemia phenotype and controls tumor progression. Leukemia 2008;22:2217–2225. doi: 10.1038/leu.2008.244
[31] Xiao X, Li BX, Mitton B et al. Targeting CREB for cancer therapy: friend or foe. Current Cancer Drug Targets 2010;10:384–391. doi: 10.2174/156800910791208535
[32] Mitton B, Dutta R, Hsu YC et al. The pp90rsk-CREB Signaling Pathway Regulates Apoptosis In Acute Myelogenous Leukemia. Proceedings of conference, New Orleans, LA, 2013.
[33] Li BX, Yamanaka K & Xiao X. Structure-activity relationship studies of naphthol AS-E and its derivatives as anticancer agents by inhibiting CREB-mediated gene transcription. Bioorg Med Chem. 2012;20:6811–6820. doi: 10.1016/j.bmc.2012.09.056
[34] Marteijn JA, van Emst L, Erpelinck-Verschueren CA et al. The E3 ubiquitin-protein ligase Triad1 inhibits clonogenic growth of primary myeloid progenitor cells. Blood 2005;106:4114–4123. doi: 10.1182/blood-2005-04-1450
[35] Shah CA, Wang H, Bei L et al. HoxA10 regulates transcription of the gene encoding transforming growth factor beta2 (TGFbeta2) in myeloid cells. J Biol Chem. 2010;286:3161–3176. doi: 10.1074/jbc.M110.183251
[36] Magnusson M, Brun AC, Miyake N et al. HOXA10 is a critical regulator for hematopoietic stem cells and erythroid/megakaryocyte development. Blood 2007;109:3687–3696. doi: 10.1182/blood-2006-10-054676
[37] Quere R, Andradottir S, Brun AC et al. High levels of the adhesion molecule CD44 on leukemic cells generate acute myeloid leukemia relapse after withdrawal of the initial transforming event. Leukemia 2011;25:515–526. doi: 10.1038/leu.2010.281
[38] Wang H, Lindsey S, Konieczna I et al. Constitutively active SHP2 cooperates with HoxA10 overexpression to induce acute myeloid leukemia. J Biol Chem. 2009;284:2549–2567. doi: 10.1074/jbc.M804704200
[39] Bei L, Huang W, Wang H et al. HoxA10 activates CDX4 transcription and Cdx4 activates HOXA10 transcription in myeloid cells. J Biol Chem. 2011;286:19047–19064. doi: 10.1074/jbc.M110.213983
[40] Besbes S, Mirshahi M, Pocard M et al. New dimension in therapeutic targeting of BCL-2 family proteins. Oncotarget 2015;6:12862–12871. doi: 10.18632/oncotarget.3868
[41] Hanahan D & Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013
[42] Hanahan D & Weinberg RA. The hallmarks of cancer. Cell 2000;100:57–70. doi: 10.1016/S0092-8674(00)81683-9
[43] Andreeff M, Jiang S, Zhang X et al. Expression of Bcl-2-related genes in normal and AML progenitors: changes induced by chemotherapy and retinoic acid. Leukemia 1999;13:1881–1892. doi: 10.1038/sj.leu.2401573
[44] Goyama S, Schibler J, Cunningham L et al. Transcription factor RUNX1 promotes survival of acute myeloid leukemia cells. J Clin Invest. 2013;123:3876–3888. doi: 10.1172/JCI68557
[45] Chan SM, Thomas D, Corces-Zimmerman MR et al. Isocitrate dehydrogenase 1 and 2 mutations induce BCL-2 dependence in acute myeloid leukemia. Nat Med. 2015;21:178–184. doi: 10.1038/nm.3788
[46] Del Gaizo Moore V & Letai A. BH3 profiling–measuring integrated function of the mitochondrial apoptotic pathway to predict cell fate decisions. Cancer Lett. 2013;332:202–205. doi: 10.1016/j.canlet.2011.12.021
[47] Lessene G, Czabotar PE & Colman PM. BCL-2 family antagonists for cancer therapy. Nat Rev Drug Discov. 2008;7:989–1000. doi: 10.1038/nrd2658
[48] Rahmani M, Aust MM, Attkisson E et al. Inhibition of Bcl-2 antiapoptotic members by obatoclax potently enhances sorafenib-induced apoptosis in human myeloid leukemia cells through a Bim-dependent process. Blood 2012;119:6089–6098. doi: 10.1182/blood-2011-09-378141
[49] Kohl TM, Hellinger C, Ahmed F et al. BH3 mimetic ABT-737 neutralizes resistance to FLT3 inhibitor treatment mediated by FLT3-independent expression of BCL2 in primary AML blasts. Leukemia 2007;21:1763–1772. doi: 10.1038/sj.leu.2404776
[50] Baev DV, Krawczyk J, O’Dwyer M et al. The BH3-mimetic ABT-737 effectively kills acute myeloid leukemia initiating cells. Leuk Res Rep 2014;3:79–82. doi: 10.1016/j.lrr.2014.06.001
[51] Rudin CM, Hann CL, Garon EB et al. Phase II study of single-agent navitoclax (ABT-263) and biomarker correlates in patients with relapsed small cell lung cancer. Clin Cancer Res. 2012;18:3163–3169. doi: 10.1158/1078-0432.CCR-11-3090
[52] Pan R, Hogdal LJ, Benito JM et al. Selective BCL-2 inhibition by ABT-199 causes on-target cell death in acute myeloid leukemia, Cancer Discov. 2014;4:362–375. doi: 10.1158/2159-8290.CD-13-0609
[53] Schimmer AD, O’Brien S, Kantarjian H et al. A phase I study of the pan bcl-2 family inhibitor obatoclax mesylate in patients with advanced hematologic malignancies. Clin Cancer Res. 2008;14:8295–8301. doi: 10.1158/1078-0432.CCR-08-0999
[54] Schimmer AD, Raza A, Carter TH et al. A multicenter phase I/II study of obatoclax mesylate administered as a 3- or 24-hour infusion in older patients with previously untreated acute myeloid leukemia. PLoS One 2014;9:e108694. doi: 10.1371/journal.pone.0108694
[55] Kralovics R, Passamonti F, Buser AS et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med. 2005;352:1779–1790. doi: 10.1056/NEJMoa051113
[56] Furqan M, Akinleye A, Mukhi N et al. STAT inhibitors for cancer therapy. J Hematol Oncol. 2013;6:90. doi: 10.1186/1756-8722-6-90
[57] Chen E, Beer PA, Godfrey AL et al. Distinct clinical phenotypes associated with JAK2V617F reflect differential STAT1 signaling. Cancer Cell 2010;18:524–535. doi: 10.1016/j.ccr.2010.10.013
[58] Yan D, Hutchison RE & Mohi G. Critical requirement for Stat5 in a mouse model of polycythemia vera. Blood 2012;119:3539–3549. doi: 10.1182/blood-2011-03-345215
[59] Steensma DP, McClure RF, Karp JE et al. JAK2 V617F is a rare finding in de novo acute myeloid leukemia, but STAT3 activation is common and remains unexplained. Leukemia 2006;20:971–978. doi: 10.1038/sj.leu.2404206
[60] Oancea C, Ruster B, Brill B et al. STAT activation status differentiates leukemogenic from non-leukemogenic stem cells in AML and is suppressed by arsenic in t(6;9)-positive AML. Genes Cancer 2014;5:378–392. PMID: 25568664
[61] Benekli M, Xia Z, Donohue KA et al. Constitutive activity of signal transducer and activator of transcription 3 protein in acute myeloid leukemia blasts is associated with short disease-free survival. Blood 2002;99:252–257. doi: 10.1182/blood.V99.1.252
[62] Ioannidis S, Lamb ML, Wang T et al. Discovery of 5-chloro-N2-[(1S)-1-(5-fluoropyrimidin-2-yl)ethyl]-N4-(5-methyl-1H-pyrazol-3-yl)p yrimidine-2,4-diamine (AZD1480) as a novel inhibitor of the Jak/Stat pathway. J Med Chem 2011;54:262–276. hdoi: 10.1021/jm1011319
[63] Hedvat M, Huszar D, Herrmann A et al. The JAK2 inhibitor AZD1480 potently blocks Stat3 signaling and oncogenesis in solid tumors. Cancer Cell 2009;16:487–497. doi: 10.1016/j.ccr.2009.10.015
[64] Derenzini E, Lemoine M, Buglio D et al. The JAK inhibitor AZD1480 regulates proliferation and immunity in Hodgkin lymphoma. Blood Cancer Journal. 2011;1:e46. doi: 10.1038/bcj.2011.46
[65] Yan S, Li Z & Thiele CJ. Inhibition of STAT3 with orally active JAK inhibitor, AZD1480, decreases tumor growth in Neuroblastoma and Pediatric Sarcomas In vitro and In vivo. Oncotarget. 2013;4:433–445. doi: 10.18632/oncotarget.930
[66] Scuto A, Krejci P, Popplewell L et al. The novel JAK inhibitor AZD1480 blocks STAT3 and FGFR3 signaling, resulting in suppression of human myeloma cell growth and survival. Leukemia 2011;25:538–550. doi: 10.1038/leu.2010.289
[67] Hossain DM, Dos Santos C, Zhang Q et al. Leukemia cell-targeted STAT3 silencing and TLR9 triggering generate systemic antitumor immunity. Blood 2014;123:15–25. doi: 10.1182/blood-2013-07-517987
[68] Zhang Q, Hossain DM, Nechaev S et al. TLR9-mediated siRNA delivery for targeting of normal and malignant human hematopoietic cells in vivo. Blood 2013;121:1304–1315. doi: 10.1182/blood-2012-07-442590
[69] Xu X, Kasembeli MM, Jiang X et al. Chemical probes that competitively and selectively inhibit Stat3 activation. PLoS One 2009;4:e4783. doi: 10.1371/journal.pone.0004783
[70] Redell MS, Ruiz MJ, Alonzo TA et al. Stat3 signaling in acute myeloid leukemia: ligand-dependent and -independent activation and induction of apoptosis by a novel small-molecule Stat3 inhibitor. Blood 2011;117:5701–5709. http://dx.doi.org/10.1182/blood-2010-04-280123
[71] Brambilla L, Genini D, Laurini E et al. Hitting the right spot: Mechanism of action of OPB-31121, a novel and potent inhibitor of the Signal Transducer and Activator of Transcription 3 (STAT3). Mol Oncol. 2015;9:1194–1206. doi: 10.1016/j.molonc.2015.02.012
[72] Hayakawa F, Sugimoto K, Harada Y et al. A novel STAT inhibitor, OPB-31121, has a significant antitumor effect on leukemia with STAT-addictive oncokinases. Blood Cancer Journal 2013;3:e166. doi: 10.1038/bcj.2013.63
[73] Ikeda M, Okusaka T, Ishii H et al. A phase I, open-label, nonrandomized trial of OPB-31121, a STAT3 inhibitor, in patients with advanced hepatocellular carcinoma (HCC). J Clin Oncol 2014;32:5s (abstract 2616).
[74] Hong DS, Younes A, Fayad L et al. A phase I study of ISIS 481464 (AZD9150), a first-in-human, first-in-class, antisense oligonucleotide inhibitor of STAT3, in patients with advanced cancers. J Clin Oncol 2013;31 (abstract 8523).
[75] Lee HJ, Daver N, Kantarjian HM et al. The role of JAK pathway dysregulation in the pathogenesis and treatment of acute myeloid leukemia. Clin Cancer Res. 2013;19:327–335. doi: 10.1158/1078-0432.CCR-12-2087
[76] Eghtedar A, Verstovsek S, Estrov Z et al. Phase 2 study of the JAK kinase inhibitor ruxolitinib in patients with refractory leukemias, including postmyeloproliferative neoplasm acute myeloid leukemia. Blood 2012;119:4614–4618. doi: 10.1182/blood-2011-12-400051
[77] Roe JS, Mercan F, Rivera K et al. BET Bromodomain Inhibition Suppresses the Function of Hematopoietic Transcription Factors in Acute Myeloid Leukemia. Mol Cell. 2015;58:1028–1039. doi: 10.1016/j.molcel.2015.04.011
[78] Quintas-Cardama A, Santos FP & Garcia-Manero G. Histone deacetylase inhibitors for the treatment of myelodysplastic syndrome and acute myeloid leukemia. Leukemia 2011;25:226–235. doi: 10.1038/leu.2010.276
[79] Jain N, Curran E, Iyengar NM et al. Phase II study of the oral MEK inhibitor selumetinib in advanced acute myelogenous leukemia: a University of Chicago phase II consortium trial. Clin Cancer Res. 2014;20:490–498. doi: 10.1158/1078-0432.CCR-13-1311
[80] Stein EM, Garcia-Manero G, Rizzieri DA et al. A Phase 1 Study of the DOT1L Inhibitor, Pinometostat (EPZ-5676), in Adults with Relapsed or Refractory Leukemia: Safety, Clinical Activity, Exposure and Target Inhibition, Proccedings of Conference A Phase 1 Study of the DOT1L Inhibitor, Pinometostat (EPZ-5676), in Adults with Relapsed or Refractory Leukemia: Safety, Clinical Activity, Exposure and Target Inhibition, Orlando, FL. 2015.
[81] Ranganathan P, Yu X, Na C et al. Preclinical activity of a novel CRM1 inhibitor in acute myeloid leukemia. Blood 2012;120:1765–1773. doi: 10.1182/blood-2012-04-423160