Abstract
Aim
To estimate prescribed antibiotic course length in 2004, and compare with 1991–92 figures and recommended prescribing guidelines, and to explore whether there is scope for cost savings from increased guideline compliance.
Design
Retrospective database analysis of the prescription analysis and cost (PACT) data by primary care trust.
Setting
One strategic health authority in England for December 2004.
Outcome measures
Course length, prescribing rate, cost per prescription by antibiotic and PCT.
Results
The proportion of five-day courses has fallen to 15.7% but seven-day courses have increased to 65.9%. The PCT with the lowest prescribing rate has a relatively high cost per prescription, and vice versa. A survey of antibiotics prescribing guidelines in primary care indicates that shorter courses are recommended, but our results suggest average course length has increased.
Conclusions
These findings have important implications for the development of antibiotic resistance in primary care and for unnecessary expenditure on antibiotics. GPs should ensure their antibiotic prescribing is consistent with local guidelines.
Concern has been voiced in recent years about the serious problem of antibiotic resistance, both in the UK and worldwide. Most concerns have focused on inappropriate and unnecessary prescribing. Antibiotic use has been correlated with risk of development of resistant strains.1 Course length is often determined by “a pragmatic choice of length of treatment based on clinical experience. This often represents overtreatment, which may give rise to adverse events, adds to selective pressures for antibiotic resistance in bacteria, and increases costs.”2
Patients are routinely advised to complete a course of antibiotics. However, there is evidence that shorter courses, or no antibiotics, are as effective as longer courses: for patients with acute uncomplicated lower respiratory tract infection, strategies have been trialled under which no antibiotic was prescribed, or the patient was given a prescription to be dispensed only after three days. Such strategies make little difference to symptom resolution compared with an immediate prescription,3 and are likely to reduce patients’ belief in the efficacy of antibiotics and the risk of breeding resistant strains through reduced population exposure. On the other hand, literature on appropriate course length is relatively sparse.
In acute otitis media in children, the benefits of antibiotics are doubtful4,5 or, at best, modest.4 An international survey indicated that there was no increase in the rate of complications in countries with a lower rate of prescribing.5 If antibiotics are given, there is doubt as to the correct course length. One study found both three- and 10-day courses were equally effective in otitis media.6
Antibiotics have been shown to be effective in acute sinusitis in adults7 and children.8 But, one study found that 10-day courses were no more effective than three-day courses.9
Acute, uncomplicated cystitis in healthy women can be treated effectively with trimethoprim.10,11 Studies have shown that three days of treatment are as effective as longer courses.12,13 One group of researchers, who examined the use of three-, five- and seven-day courses of trimethoprim in uncomplicated cystitis in general practice,14 found no significant increase in treatment failure in patients treated with the three-day regimen. A Cochrane review15 considered that, of the trials reviewed that examined course length, there was no significant difference between a short (three to six days) or long (seven to 14 days) course of antibiotics. The methodological quality of a number of the trials was, in general, regarded as poor.
Unnecessary prescribing has implications for spending, although the price of many antibiotics is minimal. However, given the correlation between antibiotic consumption and resistance,1 and given the evidence that shorter courses are as effective as longer ones, there have been many attempts to persuade prescribers in primary care to think carefully before prescribing. Antibiotic usage in England increased between 1980 and the early to mid 1990s,16,17 but has since decreased,18–20 possibly indicating that the message about appropriate antibiotics usage is being heeded.21
Since 1998, it has been recommended in the UK that no antibiotics be prescribed for simple coughs and sore throats, and viral infections. Treatment for uncomplicated cystitis should be limited to three days in otherwise fit women and antibiotics should only be prescribed by telephone in exceptional circumstances.11 Recent guidance from the National Institute for Health and Clinical Excellence (NICE) has reinforced this message, recommending not prescribing or writing delayed prescriptions for upper respiratory tract infections and acute otitis media.22
Despite this, relatively little attention has been paid to the course lengths that are prescribed. Harris and Lloyd at the prescribing research unit at the University of Leeds23 examined primary care antibiotic prescribing data for England for the year 1992–93 for amoxicillin, ampicillin, cephalexin, erythromycin, penicillin V, trimethoprim and co-trimoxazole. They found that, nationally, over all the antibiotics, 36.8 per cent of courses were for five days and 47.1 per cent for seven days. Only for penicillin V was there a preponderance of five-day courses over seven days, but for ampicillin there was an equal divide between five and seven days.
As the Harris and Lloyd study was carried out more than a decade ago, we believed it was time to see whether there had been changes in antibiotic course length since then. In this study, we set out to establish (i) whether there is a reduction in prescribed antibiotic course length in general practice compared with a decade earlier, (ii) whether it follows current guidelines, and whether there is scope for cost savings from increased compliance.
A longitudinal analysis examining the reasons for any changes in course lengths is beyond the scope of this study. We do however examine cross-sectional variation in course length and prescribing rates between PCTs.
Method
Data for this study were extracted from 17 English primary care trusts (PCTs) within one strategic health authority comprising a mixture of urban and rural areas.
Guidelines analysis
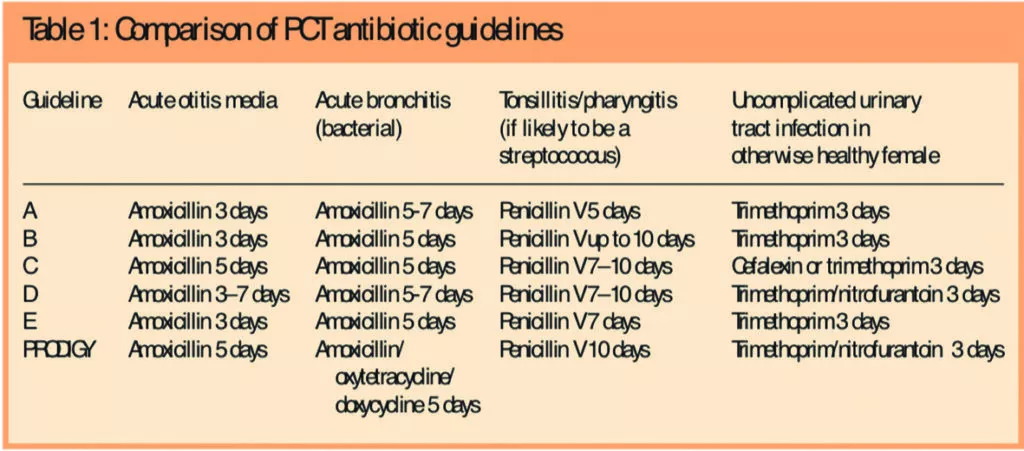
We requested copies of antibiotic prescribing guidelines from all subject PCTs, as well as PRODIGY guidelines. These were tabulated and compared for four common conditions (acute otitis media, acute bronchitis, tonsillitis/pharyngitis and uncomplicated urinary tract infection in women).
PACT data analysis
We obtained prescription analysis and cost (PACT) data from the NHS prescribing support unit for the PCTs for the month of December 2004. It was not possible to replicate Harris and Lloyd’s work exactly, because some of the antibiotics (ampicillin, co-trimoxazole) are no longer in routine use in primary care and others now in routine use were not then available. We therefore examined data for amoxicillin, flucloxacillin, cefalexin, cefaclor m/r 375mg, penicillin V, trimethoprim and co-amoxiclav. These were chosen because they represent the majority of antibiotic prescribing in the community. Each line of data was in the format: formulation; number of tablets or capsules; number of items.
We inferred course length from the number of tablets of a formulation prescribed divided by the number of tablets of that formulation routinely prescribed per day. The number of tablets routinely prescribed per day was based on the experience of one of the authors (JW). For example, all preparations of amoxicillin (whether 250mg or 500mg) are normally prescribed as one capsule three times a day, so that a prescription for 15 capsules corresponds with a five-day course, 21 with a seven-day course and so on (details available from corresponding author). The “number of items” is the frequency of prescriptions for a given formulation with a given number of tablets in the dataset. Thus for every line of data we had an (estimated) course length, and frequency (number of prescriptions of that preparation for that course length).
We restricted our analysis to acute courses only. Small numbers of tablets are often single doses used in dental or other prophylaxis and large numbers are for longer-term treatment (for example post-splenectomy prophylaxis). We therefore excluded course lengths of less than three days and more than 14 days.
Data were obtained on the registered population in each PCT for December 2004, and prescription costs were taken from list prices in the British National Formulary.25 From the above, we estimated: (i) course length by antibiotic; (ii) any potential savings from closer adherence to recommended course lengths; and (iii) prescribing rate and cost per prescription by PCT. Outlying PCTs were investigated in further detail.
In the experience of one of the authors (JW), the availability of standard patient packs may influence course length. Where a high proportion of prescriptions are for a number of units equal to standard pack sizes, there may be circumstantial evidence (and thus one could raise a hypothesis) to suggest some link between pack size and prescribed course length. We therefore calculated the proportion of prescriptions where the number of tablets prescribed is a multiple of the number of tablets in a pack.
Demography may be an explanatory factor for geographical variation in prescribing rates. Formal statistical analysis is not possible due to small sample size (n=17 PCTs). However, we compared specific therapeutic group age-sex related prescribing unit (STAR-PU) per patient; a measure of prescribing rate stratified by demography,24 across the PCTs to quantify the demographic variation across the SHA.
Results
Guidelines analysis
A request for copies of antibiotics prescribing guidelines resulted in five sets covering the SHA (with many PCTs sharing guidelines), and one from another English region. Of these, one had no recommended course lengths, and was essentially a list of antibiotics approved for use in primary care. We therefore excluded this from further analysis. Comparisons are shown for four typical clinical indications.
For uncomplicated urinary tract infection, there is a consistent recommendation for a three-day course of trimethoprim (with cefalexin or nitrofurantoin also recommended in two cases), and a five- or five to seven-day amoxicillin course for acute bronchitis. However, there is some divergence of opinion in recommended course length for acute otitis media and tonsillitis or pharyngitis, with amoxicillin recommended for between three and seven days in the former, and between five and seven to 10 days for the latter, despite prodigy guidance recommending a 10-day course (Table 1).
PACT data analysis
Course lengths for solid oral dosage forms
We obtained data on 81,152 prescriptions for antibiotics dispensed in the SHA (population 2.3 million) for December 2004. Of these, 3,294 were for course lengths of less than three days or more than 14 days were excluded from our analysis. Our sample size was therefore 77,858 prescriptions.
Overall, 15.7 per cent of courses were for five days (12,250 of 77,858) and 65.9 per cent were for seven days (51,277 of 77,858) (Table 2).
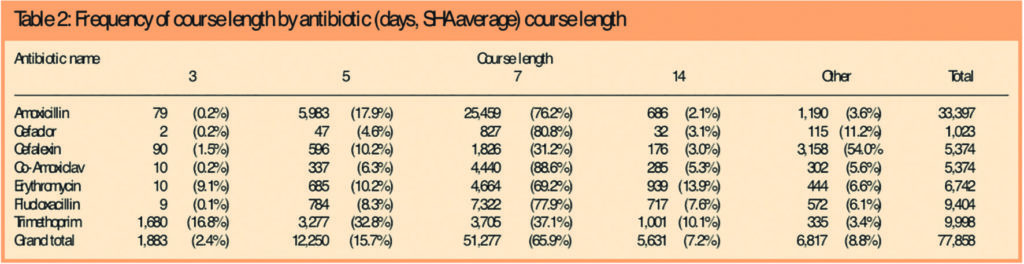
For trimethoprim, the prevalence of the recommended three-day course by PCT ranged from 4.4 per cent to 25.1 per cent of the total prescriptions for the drug (SHA average 16.8 per cent). For penicillin V, five-day courses ranged from a minimum of 4.4 per cent in one PCT to 27.5 per cent in another with an SHA average of 8.9 per cent. For seven-day courses the reverse was true, ranging from 37.1 per cent to 59.6 per cent, averaging 50 per cent. With amoxicillin, the percentage of five-day courses varied from 7.7 per cent to 42.1 per cent. The SHA average was 17.9 per cent.
Co-amoxiclav is a combination of amoxicillin and potassium clavulanate. The latter is not an antibiotic but is included to overcome microbial beta-lactamases, which can render amoxicillin ineffective. Percentages of seven-day courses ranged from 74.2 per cent to 88.2 per cent, while five-day courses ranged from 3.2 per cent to 13.9 per cent. For flucloxacillin there was a preponderance of seven-day courses for both 250mg and 500mg (approximately 78 per cent for each), but with a wide range in proportions of 14-day courses across the PCTs (2.5 per cent to 13 per cent).
For cefaclor m/r 375mg, a slow-release formulation used at a twice-daily dosage, the preponderance was for seven-day courses (mean 90.1 per cent, range 66.7 per cent to 100 per cent), with only two PCTs having 20 per cent or more of prescriptions for five days.
The results for cefalexin appear to show more than 50 per cent of prescriptions for a course length other than three, five, seven or 14 days. This is most likely due to lack of consistency in prescribing patterns with different doses being prescribed for different indications. We therefore exclude cefalexin from further analysis. Excluding cefalexin, the mean course length by PCT varies between 7.23 and 8.14 days.
Children’s antibiotic mixtures
For amoxicillin, we also received data on the number of bottles of the elixir, at two strengths — 125mg/5ml and 250mg/5ml. These are in 100ml bottles and at the usual dosage of 5ml three times a day provide a course of just under seven days. Occasionally, a prescription is written for, say, 10 days’ supply and this is dispensed from two 100ml bottles. This, subsequently, appears on the prescribing data as 200ml.
Prescriptions that specify five days’ supply of amoxicillin elixir are, in the experience of one of the authors (JW, a former locum community pharmacist), very rare. Paediatric patients, therefore, usually receive a fixed, six and two-thirds day course of amoxicillin. The vast majority of the prescriptions in our data, for both strengths of amoxicillin elixir, were for one 100ml bottle.
Pack size as a possible influence on prescribing
Observational analysis comparing pack sizes with number of units prescribed suggests around 55 per cent of prescriptions are for a number of units equal to a multiple of the pack size, although this varies by antibiotic between 80 per cent to 90 per cent for amoxicillin, cefaclor and co amoxiclav to 10 per cent to 20 per cent for erythromycin and flucloxacillin, and less than 1 per cent for trimethoprim (details available from corresponding author).
Potential savings from adherence to minimum course lengths
If all prescriptions for trimethoprim were for three days, penicillin V, seven days and amoxicillin, five days, then expenditure on these three antibiotics across the SHA for the month of December 2004 would have seen a modest fall of £19,900 (from £70,100 to £50,200). If replicated every month, this could total £239,000 over a year, although this is likely to be an overestimate given the preponderance of bacterial infections in the winter months. If the cheapest generic had been prescribed in each case, a nominal £600 could have been saved (£7,200 over 12 months).
PCT prescription rates and cost per prescription
The antibiotic prescribing rate in the PCTs ranged from 22.6 to 43.8 courses per 1,000 population per month — almost a two-fold difference (Figure 1).

STAR-PU per patient varies by only 2 per cent between the two PCTs (results not shown), suggesting demography is unlikely to be a major explanatory factor for the difference in prescribing. The mean number of courses per 1,000 population per month is 33.8, and the average cost per prescription is £2.82 (range £2.52 to £3.17). Interestingly, there seems to be an inverse relationship between prescribing rate and cost per prescription (single factor ANOVA F=8.847, p=0.009, Figure 1), but this is of borderline significance once the two outliers (labelled A and Q in Figure 1) are excluded (F=4.648, p=0.050).
Outliers
Our findings suggest an inverse relationship between number or prescriptions per head of population and the cost per prescription (Figure 1). There are two possible explanations for this. Either PCTs with fewer prescriptions per head are prescribing longer courses, or they are prescribing antibiotics with a more expensive unit cost.
We investigated the two PCTs with the highest and lowest prescribing rate in further detail (labelled A and Q on Figure 1). Overall, PCT A has a tendency to prescribe longer courses than PCT Q (P=0.0003). This is particularly the case with amoxicillin and flucloxacillin (Table 3). There is also a significant difference in the mix of antibiotics prescribed (chi-squared=275.33, P<0.00001), with a higher proportion of prescriptions for co-amoxiclav (5.8 per cent versus 2.6 per cent), flucloxacillin (17.6 per cent versus 11 per cent), and penicillin V (9.2 per cent versus 4.7 per cent) in PCT A.

Discussion
Overall, antibiotics usage varied almost twofold across this English region and this in itself is worthy of investigation. The course length data, however, are of concern. Our results indicate that, since the work carried out by Harris and Lloyd23 using 1992–93 data, the proportion of five-day courses has almost halved and the proportion of seven-day courses has risen substantially. For penicillin V, the only antibiotic found by Harris and Lloyd to have a preponderance of five-day courses, the majority are now for seven days. For trimethoprim, there is good evidence that a
three-day course is appropriate in uncomplicated cystitis in otherwise reasonably healthy women, so one would expect a majority of prescriptions for trimethoprim (used almost exclusively to treat urinary tract infections) to be for three days. This was not the case in the data that we examined, with almost five out of every six prescriptions issued for longer courses across the SHA.
By contrast, in the data for children’s amoxicillin elixir the vast majority were for a single bottle (six and two-thirds days). Thus, it seems that course length for children is fixed by the bottle size, but there is no such constraint with solid oral dosage forms. Indeed, there seems to be little logic or science behind the choice of course length in many cases.
Our request for antibiotics prescribing guidelines resulted in a comprehensive set covering the SHA (with many PCTs sharing the same guidelines). However, one of the six had no recommended course-length data, and the five sets we examined may not be representative of the country as a whole. Nevertheless, the differences in recommendations for course lengths for the same antibiotic in the same clinical situation is of concern. It is unlikely that local differences in antibiotic resistance patterns would explain such a difference in course length.
If all prescriptions had been for the recommended length there is the potential for the SHA to have saved up to £239,000 over the year. This is a modest sum compared with the entire prescribing budget for the SHA (around £375m). Nevertheless, this represents an opportunity cost in terms of benefits foregone from investing elsewhere (for example, this would fund a one-year course of herceptin for about 12 breast cancer patients).
In general, amoxicillin and penicillin V are recommended for most infections except for UTI (Table 1). Our data suggest these two antibiotics account for around 60 per cent of all prescriptions (excluding trimethoprim). This leaves around 40 per cent of prescriptions that are being prescribed as second line or possibly inappropriately prescribed first line. In the experience of a small sample of GPs (personal communication), the first-line failure rate is between 10 per cent and 25 per cent. Thus there may be substantial scope for increasing adherence to PCT guidelines.
We found an inverse relationship between cost per prescription and prescribing rate across the PCTs, although this is likely to be driven by three or four outliers. Closer investigation revealed that the PCT with the lowest prescribing rate was prescribing longer courses than the PCT with the highest prescribing rate, leading to a higher cost per prescription. It was also prescribing proportionately more co-amoxiclav, the most expensive of the antibiotics considered. However, care must be exercised in interpreting these figures in the absence of diagnosis or outcomes data. Other factors, such as patient pressure to prescribe may be driving the higher rate, and should be investigated. Strategies such as delayed dispensing of prescriptions may be particularly effective in addressing this.
Strengths and weaknesses
This analysis has a number of limitations. We inferred course lengths from the numbers of tablets prescribed. This is a somewhat imprecise activity, but should not have a great impact on the results, as prescription patterns for most antibiotics are well established; based on our assumed tablet/capsules per day, just under 90 per cent of courses were for either three, five, seven or 14 days, with the remainder appearing to be for irregular dosing. The exception is cefalexin, where assuming four tablets daily resulted in more than 50 per cent of prescriptions being for irregular time spans. This did not improve when assuming two, three or five tablets daily, suggesting that there is some heterogeneity in cefalexin dose.
A further limitation is that the prescribing data are not linked to diagnosis and so it is difficult to determine whether antibiotics are being used appropriately or for an appropriate course length for a given patient. However, where one antibiotic is used almost exclusively for one indication (for example, trimethoprim in UTI), it is possible to draw general conclusions. Further research with linked data (such as the general practice research database) would allow firmer conclusions to be drawn.
The data available to us were based on one winter month of prescriptions. Due to the preponderance of bacterial infections in the winter months, the prescribing rate may be higher than at other times in the year, but there is no reason to suppose that prescribed course lengths will be any different from any other time of year. Therefore while we may have overestimated the potential for reducing unnecessary expenditure, conclusions about course lengths should be valid, and our results do suggest general trends in prescribing habits. This analysis attempted to replicate Harris and Lloyd’s methods as closely as possible, therefore a broad comparison can be made between these results and theirs of 1992–93. This suggests the proportion of seven-day courses has increased and the proportion of five-day courses reduced; results that run counter to recommended prescribing guidelines.
Implications
With retrospective data, it is not possible to attribute causation, and care must be taken not to over-interpret such findings. What this study has shown is that there is an increase in course length compared with ten years previously (at least in the study SHA). However, these results suggest the following hypotheses:
- Many antibiotics are now in patient packs containing seven days’ supply of the drug at the normal dosage schedule. This may have influenced prescribers to increase the number of prescriptions for seven days’ supply, and indeed longer courses may be prescribed in multiples of pack size, but it would be difficult to determine any such causal influence. Our observational analysis finds the modal prescribed quantities are in many cases the standard pack size, with overall 55 per cent of prescriptions for multiples of these.
- Both geographical (between the subject PCTs), and intertemporal demographic change and cultural variation may offer an explanation, although it is not possible to detect such influences with the data available. The ethnicity and background of the prescribing physician has also been postulated as a possible explanatory variable.26,27 Other candidates could include the age structure, and relative deprivation of the population. Further work investigating this and other causative influences on geographical and temporal variation in prescribing habits would be useful.
Conclusion
There is substantial variation in prescribing habits for antibiotics within just one SHA area, and these habits do not appear to follow recommended guidelines. Due to the data available to us, we were unable to perform formal statistical tests of change in course length over time, but our results suggest there seems to be a trend towards longer courses of antibiotics compared with over a decade years ago.
In view of the relatively sparse data on appropriate course lengths, and the relatively poor quality of some studies, larger-scale analyses would help to determine appropriate course lengths for various antibiotics in common conditions. Future work should focus on investigating the determinants of course length variation and reasons for non-compliance with guidelines.
Acknowledgements
We thank David Lloyd and Sarah McDiarmid, prescribing support unit, Information Centre for Health and Social Care, Leeds, for supplying PACT data, the subject PCTs for supplying prescribing guidelines, Bob Fleetcroft, GP and honorary senior lecturer, University of East Anglia, and David Price, GP and GPIAG professor of primary care respiratory medicine, University of Aberdeen.
About the authors
Edward C. F. Wilson, MSc, is lecturer in health economics, Health Economics Group, School of Medicine, Health Policy and Practice, University of East Anglia, Norwich. John V. Wilson, MRPharmS, FRSPH, is pharmacist (retired) and former regional prescribing adviser, NHS Executive, Trent.
Correspondence to: Edward C. F. Wilson, Health Economics Group, School of Medicine, Health Policy and Practice, University of East Anglia, Norwich NR4 7TJ(tel 01603 591444, e-mail ed.wilson@uea.ac.uk)
References
- Bronzwaer SL, Cars O, Buchholz U, Molstad S, Goettsch W, Veldhuijzen IK, et al. A European study on the relationship between antimicrobial use and antimicrobial resistance. Emerging Infectious Diseases 2002; 8(3):278-82.
- Gruneberg R. Any Questions. BMJ 1989;298:378.
- Little P, Rumsby K, Kelly J, Watson L, Moore M, Warner G, et al. Information leaflet and antibioticprescribing strategies for acute lower respiratory tract infection: a randomized controlled trial.JAMA 2005;293:3029–35.
- Del Mar C, Glasziou P, Hayem M. Are antibiotics indicated as initial treatment for children withacute otitis media? A meta-analysis. BMJ 1997;314:1526–9.
- Froom J, Culpepper L, Jacobs M, DeMelker RA, Green LA, van Buchem L, et al. Antimicrobials foracute otitis media? A review from the International Primary Care Network. BMJ1997;315:98–102.
- Chaput de Saintonge DM, Levine DF, Savage IT, Burgess GW, Sharp J, Mayhew SR, et al. Trial ofthree-day and ten-day courses of amoxycillin in otitis media. BMJ 1982;284:1078–81.
- Lindbaek M, Hjortdahl P, Johnsen UL. Randomised, double blind, placebo controlled trial ofpenicillin V and amoxycillin in treatment of acute sinus infections in adults. BMJ1996;313:325–9.
- Wald ER, Chiponis D, Ledesma-Medina J. Comparative effectiveness of amoxicillin andamoxicillin-clavulanate potassium in acute paranasal sinus infections in children: a double-blind, placebo-controlled trial. Pediatrics 1986;77:795–800.
- Williams JW Jr, Holleman DR Jr, Samsa GP, Simel DL. Randomized controlled trial of 3 vs 10 daysof trimethoprim/sulfamethoxazole for acute maxillary sinusitis. JAMA 1995;273:1015–21.
- Managing urinary tract infection in women. Drug and Therapeutics Bulletin 1998;36:30–2.
- Standing Medical Advisory Committee Sub-group on Antimicrobial Resistance. The path of leastresistance. London: Department of Health; 1998.
- Charlton CA, Crowther A, Davies JG, Dynes J, Haward MW, Mann PG, et al. Three-day and ten-day chemotherapy for urinary tract infections in general practice. BMJ 1976;1:124–6.
- Norrby SR. Short-term treatment of uncomplicated lower urinary tract infections in women.Reviews of Infectious Diseases 1990;12:458–67.
- Lipman T, Price D. Decision making, evidence, audit, and education: case study of antibioticprescribing in general practice. BMJ 2000;320:1114–8.
- Lutters M, Vogt N. Antibiotic duration for treating uncomplicated, symptomatic lower urinary tract infections in elderly women. Cochrane Database of Systematic Reviews. 2002; (3):CD001535.
- DaveyPG,BaxRP,NeweyJ,ReevesD,RutherfordD,SlackR,etal.Growthintheuseof antibiotics in the community in England and Scotland in 1980–93. BMJ 1996;312:613.
- Gould IM, Jappy B. Trends in hospital antimicrobial prescribing after 9 years of stewardship. The Journal of Antimicrobial Chemotherapy 2000;45:913–7.
- Majeed A, Wrigley T. Antibiotic prescribing rates in England are falling. BMJ 2002;325:340.
- Ashworth M, Latinovic R, Charlton J, Cox K, Rowlands G, Gulliford M. Why has antibiotic prescribing for respiratory illness declined in primary care? A longitudinal study using the General Practice Research Database. Journal of Public Health 2004;26:268–74.
- Smith GE, Smith S, Heatlie H, Bashford JN, Hawker J, Ashcroft D, et al. What has happened to antimicrobial usage in primary care in the United Kingdom since the SMAC report? Description of trends in antimicrobial usage using the General Practice Research Database. Journal of Public Health 2004;26:359–64.
- Wrigley T, Tinto A, Majeed A. Age and sex specific antibiotic prescribing patterns in general practice in England and Wales, 1994 to 1998. Health Statistics Quarterly 2002;14:14–20.
- National Institute for Health and Clinical Excellence. NICE Clinical Guideline 69: Respiratory tract infections — antibiotic prescribing. Prescribing of antibiotics for self-limiting respiratory tract infections in adults and children in primary care. London: NICE; 2008.
- Harris CM, Lloyd DC. Consider short courses of antibiotics. BMJ 1994;308:919.
- Lloyd DC, Harris CM, Roberts DJ. Specific therapeutic group age-sex related prescribing units (STAR-PUs): weightings for analysing general practices’ prescribing in England. BMJ 1995;311:991–4.
- British Medical Association and Royal Pharmaceutical Society of Great Britain. British National Formulary 48. London; 2004.
- Gill P, Scrivener G, Lloyd D, Dowell T. The effect of patient ethnicity on prescribing rates. Health Trends 1995;27:111–4.
- Mangione-Smith R, Elliott MN, Stivers T, McDonald L, Heritage J, McGlynn EA. Racial/ethnic variation in parent expectations for antibiotics: implications for public health campaigns. Pediatrics 2004;113:e385–94.