Steve Gschmeissner / Science Photo Library
Heparin-induced thrombocytopenia (HIT) is an immune-mediated syndrome of thrombocytopenia and thrombosis as a result of heparin exposure. HIT is the most common type of drug-induced, immune-mediated thrombocytopenia and is associated with high morbidity and mortality if undiagnosed
[1]
.
The wide use of heparin in hospitalised patients demonstrates that HIT can develop in patients receiving the therapy for a number of indications, such as atrial fibrillation, venous thromboembolism (VTE), invasive procedures, surgery, via dialysis and even thromboprophylaxis. HIT is more common in surgical patients than in medical patients. Other groups that have shown to be at higher risk are women and older patients
[2]
.
Studies have documented a variable incidence of HIT, which ranges from <0.1% to 5% depending on the patient population and type of heparin received. In incidence, HIT is ten times higher with unfractionated heparin (UFH) compared with low molecular weight heparin (LMWH)
[3]
. Up to 50% of patients who develop HIT will present with a thrombus, with venous thrombus being more common than arterial thrombosis, occurring at a 4:1 ratio[4]
. The thrombotic
sequelae can be limb- and life-threatening, therefore, it is vital not to misdiagnose.
Pathophysiology, pathogenesis and risk factors
The pathophysiology of HIT is similar to a bacterial host defence mechanism where immunoglobulin G (IgG) antibodies bind to the heparin-platelet factor 4 (PF4) complexes. PF4 is a cytokine, a positively charged molecule of unknown function, released by alpha granules of platelets, that binds to the negatively charged heparin in the plasma. This complex undergoes a conformational change, exposing a neo -antigen to which the IgG antibodies bind. The formation of the anti-PF4–heparin complex binds to FcγIIa receptors on circulating platelets, which are removed by splenic macrophages, resulting in platelet consumption and ultimately causing thrombocytopenia. The anti-PF4–heparin complex also leads to platelet activation and aggregation, as well as production of platelet-derived microparticles, promoting excessive thrombin generation, resulting in a hypercoagulable state and leading to thrombosis. Warkentin and Kelton
described the duration of seropositivity as ranging from 50 to 85 days after stopping heparin, but this depended on the type of assay performed
[5]
.
Not all patients who develop the anti-PF4–heparin antibodies will clinically present with HIT, demonstrating that some of these complexes are not pathogenic. The high incidence of HIT associated with surgical patients is related to the concentration of circulating PF4; these molecules are released at the point of trauma to the endothelial cells
[6]
, increasing the chances of antigenic complexes forming. In addition, the wide spread use of UFH
intraoperatively increases the chances of HIT developing.
The occurrence of HIT is dependent on the duration and formulation of heparin administered because molecular weight, chain length and degree of sulphation affects the binding to PF4. UFH is derived from animal sources (porcine-origin is only available in the UK) and has a molecular weight ranging from 3,000 to 30,000 Daltons. It is a negatively charged sulphated glycosaminoglycan and has a high binding affinity to PF4. Conversely, LWMHs are depolymerised products of UFH, with smaller molecular weights ranging from 2,000 to 10,000 Daltons and have a lesser binding affinity to PF4. These structural differences explain why HIT is more likely with UFH (1–5%) than with LMWH (0.1–1%)
[2]
.
Diagnosis
The diagnosis of HIT is based on a combination of clinical manifestations and laboratory tests. Thrombocytopenia, a general term for a low blood platelet count, in hospitalised patients can be the result of several factors, such as sepsis, viral infection, drugs other than heparin, intravascular devices (e.g. cardiopulmonary bypass, intra-aortic balloon pumps, extracorporeal membrane oxygenation [ECMO]), fluid resuscitation (with dilution), disseminated intravascular clotting (DIC) and other autoimmune conditions; therefore, HIT may remain undiagnosed
[7]
. Conversely, to prevent the over-diagnosis of this rare syndrome, clinical features can be reviewed using the pre-test 4T scoring system (see Table 1) as a differential diagnosis tool to determine the likelihood of HIT. A low 4T score of ≤3 can exclude HIT, therefore, no further investigations would be required
[8]
. In 2012, a meta-analysis showed that a negative predictive value of a low probability 4T score is 99.8%
[9]
. Treatment of HIT, as described later, is indicated at a 4T score of >3, while performing laboratory tests for further investigations.
| Table 1: The 4T scoring system [8] | |||
|---|---|---|---|
| 4Ts | 2 points | 1 point | 0 point |
| Thrombocytopenia severity | Platelet count falls by >50% and platelet nadir 20–100 | Platelet count falls by 30–50% OR platelet nadir 10–19 | Platelet count falls by <30% OR platelet nadir <10 |
| Timing of platelet count fall | Clear onset between days 5–10 OR platelet fall ≤1 day (prior heparin exposure within 30 days) | Consistent with days 5–10 fall, but not clear (e.g. missing platelet counts); onset after day 10 OR fall ≤1 day (prior heparin exposure 30–100 days ago) | Platelet count fall <4 days without recent exposure |
| Thrombosis or other sequelae | New thrombosis (confirmed); skin necrosis, acute systemic reaction postintravenous | Progressive or recurrent thrombosis, non-necrotising skin lesions | None |
| Thrombocytopenia due to o ther reasons | None apparent | Possible | Definite |
Total score: 0–3: Low score 4–5: Intermediate score 6–8: High score | |||
Other HIT probability scores have been developed that factor in specific patient groups, such as patients who have had cardiopulmonary bypass (CPB), which has a good predictive value of 97%
[10]
. This particular score encompasses the platelet count pattern, time from CPB and CPB duration.
Clinical features
Thrombocytopenia
Although the definition of thrombocytopenia is a platelet count less than 150×10
9/L, a fall in platelets by 50% from baseline can be a marker of HIT. The onset of thrombocytopenia is usually between five and ten days of receiving heparin. A faster onset, within minutes to hours, can occur in patients who have had a recent exposure (typically within the past 100 days) to heparin and, therefore, developed anti-PF4–heparin antibodies
[2],[11]
.
The British Society of Haematology recommend that all patients have a baseline platelet count before heparin administration because this can assist in the diagnosis of HIT if fit were to develop[2]
. For patients who are considered at a greater risk of developing HIT, such as surgical patients receiving UFH, regular monitoring of platelets is recommended every two to three days.
Thromboembolic events
Despite thrombocytopenia, bleeding is an uncommon complication of HIT. The occurrence of thrombosis is associated with a mortality rate of 30% in HIT
[1]
. These events most commonly present as deep vein thrombosis or pulmonary embolism and, less so with arterial thrombus, presenting as a heart attack, stroke or peripheral arterial thrombosis leading to limb necrosis that requires amputation. Post-operative patients are more likely to develop a VTE, whereas arterial thrombosis is more prevalent in patients with a history of cardiovascular disease
[12]
Heparin-induced skin lesions
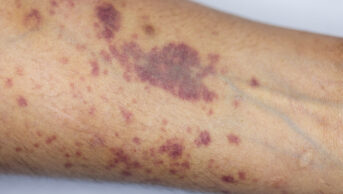
Skin lesions are rare and can occur at the site of injection, which can become necrotic. This inflammatory response is usually accompanied by fever[2]
.
Laboratory testing
Following the initial screening stages (i.e. the 4T test and clinical features), laboratory tests should be conducted to test for the presence of antibodies. For suspected HIT, the two main tests are immunoassays and functional tests.
The immunoassay, specifically enzyme-linked immunosorbent assay (ELISA), is used to detect the IgG -only or IgG/A/M antibodies that bind to the PF4–heparin complexes. If the ELISA test is positive, it is repeated using high-dose heparin (100units/ml) as a confirmatory step. These tests are highly sensitive but not specific because non-pathogenic antibodies are also detected, therefore, the chances of false positives are likely with the potential of over-diagnosis, which highlights the importance of the initial screening stage. The result of this test is measured in optical density; a reading above one would indicate a high risk of thrombosis
[2]
.
For patients who score between four and five on the 4Ts score, which is classified as intermediate, a negative particle gel immunoassay can be conducted. This can be used as rapid test to rule out diagnosis, however, it is less sensitive. A positive result would indicate further specific tests are required and an alternative anticoagulant should be initiated[2]
.
The heparin-induced platelet activation (HIPA) test and serotonin release assay (SRA) are examples of these functional tests. The HIPA test detects the heparin-dependent platelet aggregation. This is conducted by introducing heparin low dose (0.1–0.5units/ml) and high dose (100units/ml) to the patient’s plasma. This is then quality controlled with a known HIT patient’s plasma
[11]
. The SRA is a gold standard test, but it involves radiation techniques. Functional tests are the preferred methods and are only conducted in specialist laboratories. Washed platelets achieve greater sensitivity[2]
.
Treatment
The initial management of suspected HIT is to stop all heparin, including heparin locks and flushes. As a result, the platelet count starts to rise within two to three days and typically reaches baseline within four to ten days, whereas it can take up to two to three months for the antibodies to clear. Prophylactic platelet transfusion is not recommended as it may exacerbate the patient’s prothrombotic state, but it can be considered in patients with concurrent bleeding complications in view of proven efficacy
[2],[11]
.
An alternative non-heparin anticoagulant at a treatment dose should be offered even if the patient has no clinical presentation of a thrombosis. The available alternative agents should be selected based on the patient’s clinical status. These are argatroban, bivalirudin, danaparoid and fondaparinux, which are all administered parenterally. Table 2 shows the pharmacological differences and dosing of all agents.
For surgeries that require intraoperative heparin, it is recommended that the procedure be delayed until the antibodies have disappeared.
Vitamin K antagonists (VKAs), such as warfarin, should not be initiated until the platelet count has recovered because these agents are also prothrombotic since they inhibit the body’s natural anticoagulants; proteins C and S. If the patient is on a coumarin at the point of HIT diagnosis, it should be reversed. Once the patient is able to start taking warfarin, there should be an overlap period with the alternative non-heparin anticoagulant. Warfarin should be continued for one or three months
[2]
for HIT and thrombosis associated with HIT, respectively.
| Table 2: Non-heparin anticoagulant treatment options | ||||
|---|---|---|---|---|
| Fondaparinux | Danaparoid | Argatroban | Bivalirudin | |
| Target | Factor Xa | Factor Xa and factor IIa (thrombin) | Factor IIa (thrombin) | Factor IIa (thrombin) |
| Half-life | 17–21 hours | 24 hours | 40–50 minutes | 25 minutes |
| Clearance | Renal | Renal | | Enzymatic (80%) and renal (20%) |
| Route of administration | Subcutaneous injection | Intravenous infusion | Intravenous infusion | Intravenous infusion |
| Dosing regimen | <50kg:5mg daily 50–100kg: 7.5mg daily >100kg: 10mg daily (see addendum) | Intravenous bolus (weight dependent) followed by reducing dose intravenous infusion
Bolus dose: <60kg:1,500units 60–74kg:2,250units 75–90kg:3,000units >90kg:3,750units
Infusion rate: Then 300units/hour for 4 hours Then 150 or 200units/hour maintenance dose | (No bolus) Start at rate of: 2µg/kg/min
Start at a reduced rate of 0.5µg/kg/min in impaired liver function, critical illness or after cardiac surgery | (No bolus) Start at rate of: 0.15–2mg/kg/min |
Ongoing monitoring | None required | Adjust dose according to anti-Xa (target 0.5–0.8units/ml) | Adjust dose according to activated partial thromboplastin time ratio (APTTr) (target 1.5–3) | Adjust dose according to APTTr (target 1.5–2.5) |
Argatroban is a direct thrombin inhibitor and reversibly binds to thrombin. It is licensed for the treatment of HIT and is administered as a continuous infusion, requiring dose adjustments according to activated partial thromboplastin time ratio (APTTr) with a starting infusion rate of 2µg/kg/min, with a maximum recommended dose of 10µg/kg/min[13]
. Argatroban reaches steady state within 1 to 3 hours, therefore, the APTTr should be taken 2 hours after initiating the infusion to guide the dose adjustments, with subsequent APTTr to be taken less frequently when in range. Lower doses starting at 0.5µg/kg/min in hepatic impairment or critically ill patients are recommended. In view of argatroban being metabolised via the hepatic system, a dose reduction is required alongside more frequent monitoring of APTTr. Research in the critical care setting has demonstrated that patients are more sensitive to argatroban and require low doses to avoid excessive anticoagulation[14]
.
However, argatroban use increases prothrombin time, causing a false rise in international normalised ratio (INR), therefore, it can be challenging when switching a patient to longer-term management with a VKA. Consequently, concurrent argatroban and a coumarin should be administered for at least five days, with two consecutive days of INR≥4. An upper target INR for this period is not given, but to prevent over-anticoagulation the recommendation would be to stop argatroban with an INR of >5. In this situation, the argatroban should be stopped for four hours and then INR repeated
[2]
.
The benefits of argatroban are that it is rapid acting, which significantly reduces the risk of new thrombosis and, in turn, reduces mortality rates[15]
. This agent would be preferable in patients with end-stage renal disease who are on haemodialysis because no dose adjustments are needed[9]
. Argatroban has been demonstrated to be a successful anticoagulant in patients with multiple organ dysfunction syndrome and in patients undergoing percutaneous coronary intervention (PCI).
Bivalirudin is an oligopeptide analogue of hirudin, which inhibits thrombin. It is licensed for use in patients undergoing PCI, but its use in HIT is off-label. It is administered as a continuous infusion requiring dose adjustments according to APTTr, with a starting infusion rate of 0.15–2mg/kg/min.
The American College of Chest Physicians (ACCP)
[11]
recommend the use of bivalirudin for patients diagnosed with HIT who require urgent cardiac surgery or PCI. Cohort studies have demonstrated that the rate of death, Q wave myocardial infarction, repeat operation of cardiac revascularisation or stroke were similar for patients treated with bivalirudin with or without HIT.
The advantage of bivalirudin is that it has the shortest half-life (25 mins) of all the agents, and coagulation reaches baseline within 1 to 2 hours of stopping the infusion. It is a unique anticoagulant because its clearance is predominantly by proteolysis. However, the half-life extends up to four hours in patients on dialysis, therefore, they require a lower initial dose and frequent monitoring
[16]
.
Danaparoid is a heparinoid, containing a mixture of heparin sulphate, dermatan sulphate and chondroitin sulphate (which inhibits factor Xa, as well as factor II at a lesser degree). This is administered intravenously at a therapeutic dose, as outlined in table 2. Within the European Union, prophylactic doses are also being used, however, these are not recommended in the UK after reports of treatment failures. Monitoring anti-Xa assay and dose adjustments are required for patients with severe renal impairment or body weight above 90kg[11]
.
In vivo
studies have documented rare cross-reactivity of HIT with danaparoid. Furthermore, this agent has a long half-life, so rapid reversal after discontinuation will not occur. This agent is usually selected for patients with normal renal function. Prothrombin time and INR are not effected by danaparoid, so the transition to a VKA is not as complicated as with agents that influence these monitoring parameters. The majority of evidence for the treatment in pregnant patients who develop HIT is for danaparoid, therefore, it is suggested as a first-line therapy by the ACCP in this patient group
[11]
.
Fondaparinux sodium is a synthetic pentasaccharide that inhibits activated factor X. Dosage is based on weight and administered subcutaneously once a day, which is ideal for ease of use outside the critical care setting. It does not require routine monitoring, however, it is contraindicated in patients with a creatinine clearance of 20ml/min
[17]
.
Although this agent has a similar structure to heparin, fondaparinux has weak affinity for PF4 and the incidence of HIT with this agent is minimal. It is successfully used as a treatment for HIT, however, its use for this indication is off-label, as well as controversial in view of its immungenicity
[18]
.
Fondaparinux can also be considered in pregnant HIT patients if danaparoid is unavailable.
Challenges with treatment options
These treatments are more expensive than heparin, associated with a relatively increased risk of bleeding and no antidote is currently available for any of them. However, this is offset by the short half-life of selected anticoagulants. The challenge remains when transitioning to an oral anticoagulant for long-term management because of the potential of thrombosis if a VKA is prematurely initiated and/or the parenteral anticoagulant is prematurely discontinued.
Possible future therapies
Observational data show that the treatment of acute HIT can be simplified with possible oral options being investigated for efficacy in this context, such as direct oral anticoagulants (DOACs)
[19]
. Suggestions have been made that high-dose intravenous immunoglobulins (IVIG) may have an adjunctive effect alongside the alternative anticoagulant, however, data are limited.
Financial and conflicts of interest disclosure:
The authors have no relevant affiliations or financial involvement with any organisation or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. No writing assistance was used in the production of this manuscript.
- On 21 August 2017, this article was updated to clarify contraindication
s in fondaparinux dosing in renal function.
Addendum
Therapeutic doses of fondaparinux (5mg, 7.5mg, 10mg daily) are contraindicated in patients with a creatinine clearance of less than 30ml/min and should be used with caution in moderate renal impairment (30–50ml/min)[a]
. However, it is important to note that the thromboprophylactic or acute coronary syndrome dose of fondaparinux 2.5mg daily is contraindicated in patients with a creatinine clearance of less than 20ml/min[b]
.
References:
[a]
Summary of Product Characteristics. Arixtra fondaparinux sodium solution for injection 10mg/0.8ml. Aspen. August 2014
[b]
Summary of Product Characteristics. Arixtra fondaparinux sodium solution for injection 2.5 mg/0.5ml. Aspen. September 2016
Reading this article counts towards your CPD
You can use the following forms to record your learning and action points from this article from Pharmaceutical Journal Publications.
Your CPD module results are stored against your account here at The Pharmaceutical Journal. You must be registered and logged into the site to do this. To review your module results, go to the ‘My Account’ tab and then ‘My CPD’.
Any training, learning or development activities that you undertake for CPD can also be recorded as evidence as part of your RPS Faculty practice-based portfolio when preparing for Faculty membership. To start your RPS Faculty journey today, access the portfolio and tools at www.rpharms.com/Faculty
If your learning was planned in advance, please click:
If your learning was spontaneous, please click:
References
[1] Fran chini M. Heparin-induced thrombocytopenia: an update. Thromb J 2005;3:14. doi: 10.1186/1477-9560-3-14
[2] Watson H, Davidson S & Keeling D. Guidelines on the diagnosis and management of heparin-induced thrombocytopenia: second edition. Br J Haematol 2012;159(5):528–540. doi: 10.1111/bjh.12059
[3] Greinacher A, Alban S, Omer -Adam MA et al . Heparin-induced thrombocytopenia: a stoichiometry-based model to explain the differing immunogenicities of unfractionated heparin, low-molecular-weight heparin, and fondaparinux in different clinical settings. Thromb Res 2008;122(2):211–220. doi: 10.1016/j.thromres.2007.11.007
[4] Warkentin TE & Kelton JG. A 14-year study of heparin-induced thrombocytopenia. Am J Med 1996;101:502–507. doi: 10.1016/S0002 -9343(96)00258-6
[5] Warkentin TE & Kelton JG. Temporal aspects of heparin-induced thrombocytopenia. New Eng J Med 2001;344:1286–1292. doi: 10.1056/NEJM200104263441704
[6] Prechel MM & Walenga JM. Emphasis on the role of PF4 in the incidence, pathophysiology and treatment of heparin induced thrombocytopenia. Thromb J 2013;11:7. doi: 10.1186/1477-9560-11-7
[7] Krzych LJ, Nowacka E & Knapik P. Heparin-induced thrombocytopenia. Anaesthesiol Intensive Ther 2015;47(1):63–76. doi: 10.5603/AIT.2015.0006
[8] Lo GK, Juhl D, Warkentin TE et al . Evaluation of the pretest clinical score (4T’s) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost 2006;4:759–765. doi: 10.1111/j.1538-7836.2006.01787.x
[9] Cuker A, Gimotty PA, Crowther MA et al . Predictive value of the 4Ts scoring system for heparin-induced thrombocytopenia: a systematic review and meta-analysis. Blood 2012;120(20):4160–4167. doi: 10.1182/blood-2012-07-443051
[10] Lillo -le Louët A, Boutouyrie P, Alhenc -Gelas M et al . Diagnostic score for heparin-inducted thrombocytopenia after cardiopulmonary bypass. J Thromb Haemost 2004;2:1882–1888. doi: 10.1111/j.1538-7836.2004.00949.x
[11] Linkins L, Dans AL, Moores LK et al . Treatment and prevention of heparin-induced thrombocytopenia, antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012;141(2 Suppl):e495S -e530S. doi: 10.1378/chest.11-2303
[12] Boshkov LK, Warkentin TE, Hayward CP et al. Heparin-induced thrombocytopenia and thrombosis: clinical and laboratory studies. Br J Haematol 1993;84:322–328. doi: 10.1111/j.1365-2141.1993.tb03072.x
[13] Summary of Product Characteristics, Exembol Multidose 100mg/ml concentrate for solution for infusion, Mitsubishi Tanabe Pharma Europe Ltd, date of revision of the text: 23/10/2014.
[14] Alatri A, Armstrong AE, Greinacher A et al. Results of a consensus meeting on the use of argatroban in patients with heparin-induced thrombocytopenia requiring antithrombotic therapy – a European perspective. Thromb Res 2012;129:426–433. doi: 10.1016/j.thromres.2011.11.041
[15] Lewis B, Wallis D, Berkowitz S et al . ARG -911 Study Investigators. Argatroban anticoagulant therapy in patients with heparin-induced thrombocytopenia. Circulation 2001;103:1838–1843. doi: 10.1161/01.CIR.103.14.1838
[16] Warkentin TE, Greinacher A & Koster A. Bivalirudin. J T hromb Haemost 2008;99(5):830–839. doi: 10.1160/TH07 -10-0644
[17] Summary of Product Characteristics. Arixtra Fondaparinux sodium solution for injection 2.5mg/0.5ml, Aspen, date of revision of the text: 09/2016.
[18] Burch M & Cooper B. Fondaparinux -associated heparin-induced thrombocytopenia. Baylor University Medical Center 2012;25(1):13–15. PMID: 22275775
[19] Linkins LA, Warkentin TE, Pai M et al . Rivaroxaban for the treatment of suspected or confirmed heparin-induced thrombocytopenia study. J Thromb Haemost 2016;14:1206–1210. doi: 10.1111/jth.13330