Open access article
The Royal Pharmaceutical Society has made this article free to access in order to help healthcare professionals stay informed about an issue of national importance.
To learn more about coronavirus, please visit: https://www.rpharms.com/resources/pharmacy-guides/wuhan-novel-coronavirus
Vaccines have been the success story of the COVID-19 pandemic, with three already approved for use in the UK, and promising late-stage trial results for a further two vaccines backed by the UK government.
This feature illustrates how each type of vaccine works, and brings together key data for pharmacists on the vaccines currently available, as well as those in the pipeline. It will be updated regularly to include the latest information.
Although data on vaccine efficacy are included, direct comparisons between vaccines are not possible owing to the trials using different endpoints and taking place at different times — when there were differing case rates and circulating variants.
Click on the following links to take you to the vaccines you want to learn more about:
mRNA vaccines: Pfizer/BioNTech; Moderna; CureVac
Adenoviral vector vaccines: University of Oxford/AstraZeneca; Janssen
Protein-based vaccines: Novavax; GSK/Sanofi
mRNA vaccines
mRNA vaccines are made using a new technology, which — in the case of COVID-19 — uses a nanoparticle to deliver the genetic instructions for human cells to make the SARS-CoV-2 spike protein, priming the immune system to attack the SARS-CoV-2 virus if it later infects the body (see Figure).
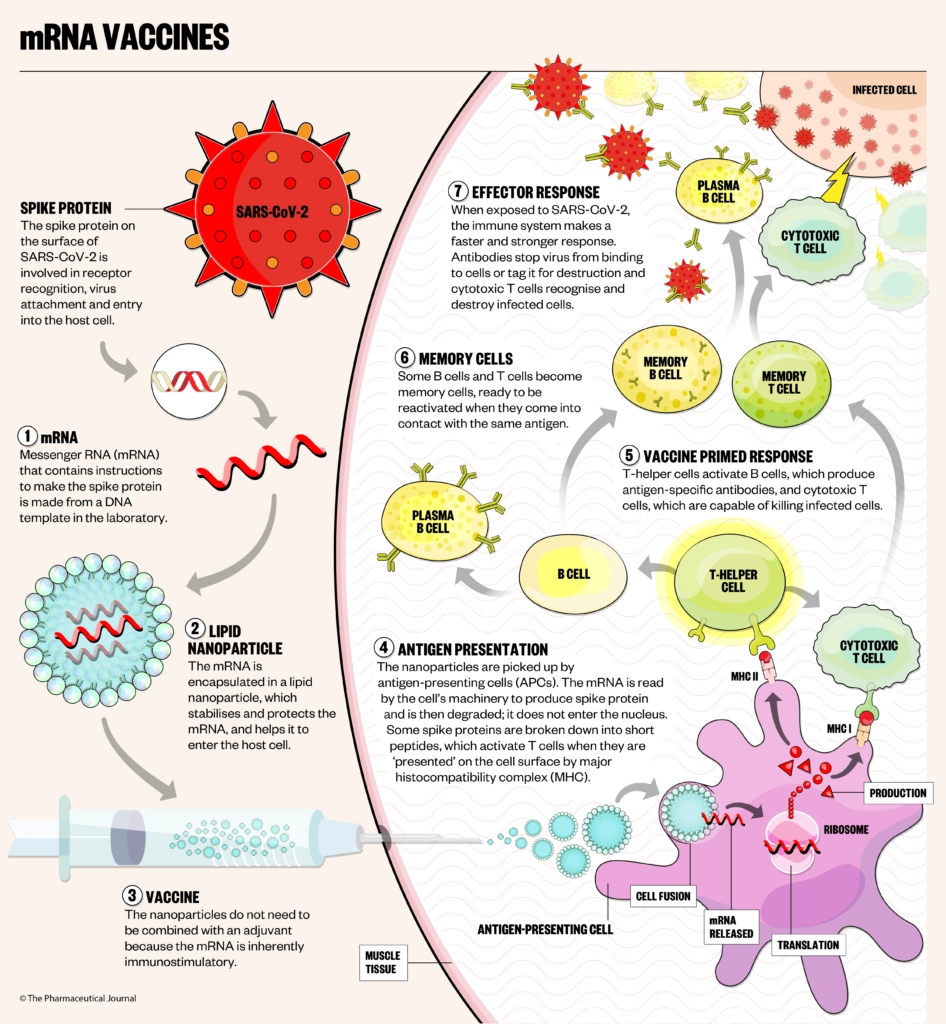
Pfizer/BioNTech (BNT162b2, brand name Comirnaty)
Is it approved in the UK?
Yes, it was authorised for temporary supply on 2 December 2020 and now has a conditional marketing authorisation.
How many doses have been ordered?
100 million.
What are the storage requirements?
- Store between -80°C and -60°C. Shelf life is 9 months.
- After thawing, undiluted vaccine can be stored for up to 31 days at 2–8°C, and up to 2 hours at temperatures up to 25°C.
- Once diluted, store at between 2–25˚C and use within 6 hours.
- Data support temperature excursion of -60°C to -15°C for up to 2 weeks, and vials that have been exposed to a single temperature excursion within these conditions can be returned to the recommended storage conditions. Unlike the European Medicines Agency and the US Food and Drug Administration, the Medicines and Healthcare products Regulatory Agency has not advised that the vaccine can be transported and stored at these higher temperatures.
What are the latest data on effectiveness?
Phase II/III study results, published on 31 December 2020, suggest that a two-dose regimen of BNT162b2 confers 94.6% protection against symptomatic COVID-19, at least 7 days following the second dose. One case of severe COVID-19 occurred in the vaccine group compared with 9 in the placebo group.
Since then, several ‘real-world’ studies have been published.
The latest consensus judgement of the Vaccine Effectiveness Expert Panel (27 August 2021), which comprises scientific and analytical specialists from academia and government in the UK, is that protection against symptomatic disease from the currently circulating variant (Delta) after two doses of the Pfizer/BioNTech vaccine is around 85%. Protection against hospitalisation and death is about 95% after two doses.
However, there is some evidence that vaccine efficacy begins to wane after six months. A pre-print published by Pfizer on 28 July 2021, suggests that efficacy peaks at 96.2% (95% CI 93.3–98.1) during the interval from 7 days to <2 months post-dose 2, and declines gradually to 83.7% (95% CI 74.7–89.9) from 4 months post-dose 2 to the data cut-off, an average decline of ∼6% every 2 month. The study was later published in The New England Journal of Medicine, along with two further studies, one global and one in Israel, looking at the benefits of a third ‘booster’ dose.
Waning against hospitalisation appears to be much more limited, with the Pfizer/BioNTech vaccine continuing at about 95% beyond 20+ weeks, according to PHE. Waning appears to be greater among those in the clinically extremely vulnerable group, though data beyond 20 weeks is limited.
Analysis carried out by the ZOE COVID study, which investigates real-world vaccine effectiveness, found that the protection provided by two doses of the Pfizer/BioNTech vaccine wanes within six months – from 88% protection against infection a month after the second dose, to 74% after five to six months.
A trial in adolescents has been published in The New England Journal of Medicine (27 May 2021), which found vaccine efficacy was 100% in over 2,000 participants aged 12 to 15 years old, with 18 cases in the placebo group and none in the vaccine group. Topline results from a phase II/III trial of 2,268 children aged 5 to 11 years old, announced by Pfizer/BioNTech on 20 September 2021, suggest a lower dose of the vaccine is effective in younger children. The antibody responses and side effect profile in the younger children given 10µg doses were comparable to those recorded in a previous study in people 16 to 25 years of age immunised with 30µg doses. The study was published in The New England Journal of Medicine on 9 November 2021.
Initial results from the OCTAVE trial, published as a pre-print on 23 August 2021, showed that 40% of clinically at-risk patients with specific immunocompromised or immunosuppressed conditions mounted a low or undetectable immune response after two doses of the Pfizer/BioNTech or Oxford/AstraZeneca vaccine.
A real-world study by the UK Health Security Agency, published on 15 November 2021, shows that, 2 weeks after receiving a booster dose, protection against symptomatic infection in adults aged 50 years and over was 93.1% (95% CI 91.7–94.3) in those who had the Oxford/AstraZeneca vaccine as their primary course and 94.0% (95% CI 93.4–94.6) for those who had Pfizer/BioNTech.
Is it effective against new variants?
Yes, but effectiveness may be reduced against some variants and more data are needed.
Alpha (B.1.1.7; first detected in UK): Data from the UK vaccination programme, published as pre-prints on 1 March 2021 and 23 April 2021, and the PHE SIREN study, suggest vaccine efficacy is still high against Alpha.
Observational data from the vaccine roll-out in Qatar, where 265,410 people have received two doses of the Pfizer/BioNTech vaccine, published in a letter to The New England Journal of Medicine on 5 May 2021, suggest vaccine efficacy is 89.5% (95% CI 85.9–92.3) at 14 or more days after the second dose. Vaccine efficacy against severe, critical, or fatal disease caused by either Alpha or Beta was high at 97.4% (95% CI 92.2–99.5).
And national-level data from Israel’s vaccination programme confirm that the vaccine is highly effective against the Alpha variant (see above).
Beta (B.1.351, first detected in South Africa): Analysis of 800 participants in Pfizer/BioNTech’s phase III trial, announced by the companies on 1 April 2021, shows nine cases of COVID-19 in the placebo group, six of which were due to Beta, and none in the vaccine group, suggesting vaccine efficacy of 100% (95% CI 53.5–100.0).
Observational data from the vaccine roll-out in Qatar, where 265,410 people have received two doses of the Pfizer/BioNTech vaccine, published in a letter to The New England Journal of Medicine on 5 May 2021, suggest vaccine efficacy is 75.0% (95% CI 70.5–78.9). Vaccine efficacy against severe, critical, or fatal disease caused by either Alpha or Beta was high at 97.4% (95% CI 92.2–99.5).
Gamma (P1, first detected in Japan/Brazil): A small laboratory study using vaccine sera suggests roughly equivalent neutralisation of this variant, although two other laboratory studies, published as preprints, suggest antibody titres were reduced by 2.6-fold and 14-fold. The impact is yet to be confirmed in clinical trials.
Delta (B.1.617.2, first detected in India): An observational study carried out by PHE, initially published as a pre-print but later published in The New England journal of Medicine on 22 July 2021, which included 4,272 people confirmed as having the Delta variant, suggested the Pfizer/BioNTech vaccine has an efficacy of 88.0% (95% CI 85.3–90.1) against symptomatic disease from the Delta variant 2 weeks after the second dose, compared with 93.7% (95% CI 91.6–95.3) against the Alpha variant. Vaccine efficacy after one dose was 35.6% (95% CI 22.7–46.4) against the Delta variant compared with 47.5% (95% CI 41.6–52.8) against the Alpha variant.
An updated analysis, including 14,019 symptomatic cases infected with the delta variant, was published as a pre-print by PHE on 14 June 2021. The analysis suggests vaccine efficacy against admission to hospital with the delta variant is 94% (95% CI 46–99) after one dose and 96% (95% CI 86–99) after two doses.
A Scottish study published on 14 June 2021 as a research letter in The Lancet, included 19,543 cases (7,723 with the Delta variant), of whom 377 were admitted to hospital (134 with the Delta variant). The vaccine offered 79% (955 CI 75–82) protection against the Delta variant and 92% (95% CI 90–93) against the Alpha variant. An updated analysis published as a letter in The New England Journal of Medicine on 20 October 2021 found that vaccine efficacy against death from the Delta variant 14 or more days after the second vaccine dose was 90% (95% CI 83–94).
Another letter in The New England Journal of Medicine, published on the same date, looked at vaccine efficacy against Delta infection in adolescents between the ages of 12 and 18 years in Israel. Estimated vaccine efficacy was 93% (95% CI 88–97) on days 7 to 21 after the second dose.
Omicron (B.1.1.529, first detected in South Africa): Early analysis of Omicron cases in the UK suggests vaccine effectiveness against symptomatic disease is significantly lower compared with the Delta variant (less than 10% and just under 40%, 25 weeks or more after an Oxford/AstraZeneca and Pfizer/BioNTech primary course, respectively). However, from 2 weeks after a Pfizer booster dose, vaccine effectiveness increases to around 71% among those who received Oxford/AstraZeneca as the primary course and around 76% among those who received Pfizer/BioNTech as the primary course.
Does it work in older people?
Yes.
Observational data from PHE, published as a pre-print on 10 May 2021, suggest that a single dose of the Pfizer/BioNTech vaccine reduces risk of death in over 70-year-olds by 44% (0.56, 95% CI 0.47–0.68), and two doses reduce the risk by 69% (0.31, 95% CI 0.14-0.69), compared with unvaccinated cases, which, when combined with the protection vaccines offer against becoming a case in the first place, is equivalent to approximately 80% protection against death with one dose and 97% protection with two doses. Another preprint, published by PHE on the same date, suggests, for the over 80s, two doses of the Pfizer/BioNTech vaccine reduce the risk of hospitalisation by 93% (95% CI 89–95). In 70- to 79-year-olds, one dose reduces the risk of hospitalisation by 81% (95% CI 73–87).
A large prospective cohort study (VIVALDI) of vaccine efficacy in 10,412 residents of long-term care facilities in England, all aged 65 years and older, was published in The Lancet Infectious Diseases on 23 June 2021. Vaccine efficacy for a single dose of either the Pfizer/BioNTech or Oxford/AstraZeneca vaccines was estimated to be 56% (95% CI 19–76) at 28–34 days, and 62% (23–81) at 35–48 days post-vaccination. Similar effect sizes at 35-48 days were seen for the Pfizer/BioNTech vaccine (65% reduced risk) and Oxford/AstraZeneca vaccine (68% reduced risk).
Does it reduce transmission?
Yes, for some variants. However, studies on the transmission blocking effect of vaccines against the Delta variant are ongoing.
A study led by Public Health Scotland and the University of Glasgow, published as a pre-print on 12 March 2021, examining Alpha infections in nearly 150,000 vaccinated and unvaccinated healthcare workers and 200,000 of their household members, provides the first direct evidence of a reduction in transmission. There was a 30% reduction in documented cases of infection in household members of vaccinated compared with unvaccinated healthcare workers (hazard ratio [HR] 0.70, 95% CI 0.63–0.78), which the researchers estimate translates to a 60% reduction in transmission since household members could be infected by other routes. A 54% reduction in documented cases in household members was observed ≥14 days after the second dose (HR 0.46, 95% CI 0.30—0.70). Participants received either the Pfizer/BioNTech vaccine or the Oxford/AstraZeneca vaccine. The study was published as a letter to the editor in NEJM on 8 September 2021.
The results of the Scottish research are backed up by a Public Health England study, published on 28 April 2021, which found that individuals who become infected 3 weeks after receiving one dose of the Pfizer/BioNTech or Oxford/AstraZeneca vaccine are between 38% and 49% less likely to pass the virus on to their household contacts than those who were unvaccinated. The study included over 57,000 contacts from 24,000 households in which there was a lab-confirmed case that had received a vaccination, compared with nearly 1 million contacts of unvaccinated cases. The study took place when the Alpha variant was circulating.
A study in Lancet Infectious Diseases, published on 29 October 2021, suggests that fully vaccinated people (either with Pfizer/BioNTech or Oxford/AstraZeneca) can contract and pass on the Delta variant of COVID-19 in the home, but at lower rates than unvaccinated people. The study found that around 25% (95% CI 18–33) of vaccinated household contacts tested positive for COVID-19 compared with roughly 38% ( 95% CI 24–53) of unvaccinated household contacts. The researchers also found that peak viral load was similar in vaccinated and unvaccinated people.
A study conducted by the UK Health Security Agency, published as a pre-print on 25 November 2021, suggests that transmission of the delta variant is reduced by 37% (95% CI 4–65) after two doses of Pfizer/BioNTech.
Are there any safety concerns?
The safety profile generally reflects that seen in clinical trials (injection-site reactions and generalised ‘flu-like’ symptoms). However, some safety signals have emerged.
On 7 May 2021, the Pharmacovigilance Risk Assessment Committee of the European Medicines Agency announced that it had concluded a review of a safety signal for facial swelling in people with a history of injections with dermal fillers, and found that there is at least a reasonable possibility of a causal association between the vaccine and the reported cases and requested that it should be added as a side effect in the summary of product characteristics. The PRAC stressed that the risk-benefit balance of the vaccine remains unchanged.
On 25 Jun 2021, the product information was updated to include a warning that there have been very rare reports of myocarditis and pericarditis occurring after vaccination, often in younger men and shortly after the second dose of the vaccine. As of 8 December 2021, the Medicines and Healthcare products Regulatory Agency had received 507 reports of myocarditis and 365 reports of pericarditis following use of the Pfizer/BioNTech vaccine.
Cases are typically mild and individuals tend to recover within a short time following standard treatment and rest. However, healthcare professionals should be alert to the signs and symptoms of myocarditis and pericarditis. Vaccinated individuals should also seek immediate medical attention should they experience new onset of chest pain, shortness of breath, palpitations or arrhythmias.
Full product information can be viewed here.
Moderna (mRNA-1273, brand name Spikevax)
Is it approved in the UK?
Yes, it was authorised for temporary supply on 3 January 2021, and given a conditional marketing authorisation on 1 April 2021.
How many doses have been ordered?
17 million.
What are the storage requirements?
- Store between -25º and -15ºC. Shelf life is 7 months.
- Once thawed, unopened vials can be stored at 2–8 °C for up to 30 days or at 8– 25°C for 12 hours.
- After opening, store at 2–25˚C and use within 6 hours.
What are the latest data on effectiveness?
Vaccine efficacy was 94.1% (95% CI 89.3–96.8) against symptomatic infection 14 days after the second dose in clinical trials. No cases of severe COVID-19 occurred in the vaccine group compared with 30 in the placebo group.
The latest consensus judgement of the Vaccine Effectiveness Expert Panel (27 August 2021), which comprises scientific and analytical specialists from academia and government in the UK, is that protection against symptomatic disease from the currently circulating variant (Delta) after two doses of the Moderna vaccine is around 85%.
The high efficacy is sustained for six months, according to updated results from the phase III COVE study announced by Moderna on 13 April 2021. A review of cases suggests vaccine efficacy starting two weeks following the second dose is greater than 90% against all cases of COVID-19, and greater than 95% against severe cases of COVID-19, with a median follow up of approximately six months following the second dose. Final results from COVE III, announced by Moderna on 5 August 2021, confirmed high efficacy of 93% is sustained for six months after the second dose. However, an analysis by Moderna of breakthrough infections in an open-label part of COVE suggested that they occur less frequently in those vaccinated more recently (8 months since the first dose compared with 13 months since the first dose).
Moderna has also announced topline results from a phase II/III trial in adolescents (25 May 2021), which found that vaccine efficacy was 100% in 3,732 participants aged 12 to 17 years old, with 4 cases in the placebo group and none in the vaccine group at least 14 days after the second dose. The full study was published in The New England Journal of Medicine on 11 August 2021.
Data on under 40 year olds, published by PHE on 1 July 2021, suggest vaccine efficacy is 72% (95% CI 46–86%) after 1 dose.
Does it work against new variants?
Yes, but effectiveness may be reduced against some variants and more data are needed.
Alpha (B.1.1.7; first detected in UK): A small laboratory study suggest no significant difference, but this is yet to be demonstrated in clinical trials.
Beta (B.1.351, first detected in South Africa): A small laboratory study suggest a 6.5-fold reduction in antibodies that neutralise this variant, but this is yet to be demonstrated in clinical trials.
Gamma (P1, first detected in Japan/Brazil): A small laboratory study suggests antibody titres are reduced by 2.6-fold. The impact is yet to be confirmed in clinical trials.
Delta (B.1.617.2, first detected in India): The latest consensus judgement of the Vaccine Effectiveness Expert Panel, published on 27 August 2021, is that vaccine efficacy against infection is about 75% after one dose and 85% after two doses.
Omicron (B.1.1.529, first detected in South Africa): No evidence yet.
Does it work in older people?
Yes, in clinical trials.
In clinical trials, vaccine efficacy was 86.4% (95% CI 61.4–95.2) against symptomatic infection 14 days after the second dose in those aged >65 years.
Does it reduce transmission?
More than likely, but we need more data. Studies on the transmission blocking effect of vaccines against the Delta variant are ongoing.
Data from swabs taken during clinical trials to check for asymptomatic infections, submitted to the US Food and Drug Administration by Moderna, suggest transmission could be reduced by two-thirds after one dose, but the number of asymptomatic infections was small and the data have not yet been peer reviewed.
An observational study in the US, published on 29 March 2021, also confirmed that both asymptomatic and symptomatic infections are reduced by 90% at least 14 days after the second dose.
Are there any safety concerns?
The safety profile generally reflects that seen in clinical trials (injection-site reactions and generalised ‘flu-like’ symptoms). However, some safety signals have emerged.
On 25 Jun 20201, the product information was updated to include a warning that there have been very rare reports of myocarditis and pericarditis occurring after vaccination, often in younger men and shortly after the second dose of the vaccine. As of 8 December 2021, the Medicines and Healthcare products Regulatory Agency had received 111 reports of myocarditis and 63 reports of pericarditis following use of the Moderna vaccine.
Cases are typically mild and individuals tend to recover within a short time following standard treatment and rest. However, healthcare professionals should be alert to the signs and symptoms of myocarditis and pericarditis. Vaccinated individuals should also seek immediate medical attention should they experience new onset of chest pain, shortness of breath, palpitations or arrhythmias.
Full product information can be viewed here.
CureVac (CV2CoV)
Is it approved in the UK?
No.
How many doses have been ordered/pre-ordered?
50 million.
What are the storage requirements?
- Store at 2–8ºC for up to 3 months.
- Store at room temperature for up to 24 hours.
What are the latest data on effectiveness?
A pre-clinical trial of CV2CoV, published as a pre-print on 13 May 2021, showed it induced high levels of antigen production as well as strong and dose-dependent immune responses in vaccinated animals.
Does it work against new variants?
We don’t know yet.
Alpha (B.1.1.7; first detected in UK): In a laboratory study, the serum of animals vaccinated with CV2CoV showed significant cross-neutralisation against Alpha variants. The impact is yet to be confirmed in clinical trials.
Beta (B.1.351, first detected in South Africa): In a laboratory study, the serum of animals vaccinated with CV2CoV showed significant cross-neutralisation against Beta variants. The impact is yet to be confirmed in clinical trials.
Gamma (P1, first detected in Japan/Brazil): No evidence yet.
Delta (B.1.617.2, first detected in India): No evidence yet.
Omicron (B.1.1.529, first detected in South Africa): No evidence yet.
Does it work in older adults?
We don’t know yet.
Data not yet available.
Does it reduce transmission?
We don’t know yet.
Data not yet available.
Are there any safety concerns?
Full product information is not yet available.
Adenoviral vector vaccines
Adenoviral vector vaccines are made using a relatively new technology, which uses a common cold virus to deliver the genetic instructions for human cells to make the SARS-CoV-2 spike protein, priming the immune system to attack the SARS-CoV-2 virus if it later infects the body (see Figure).
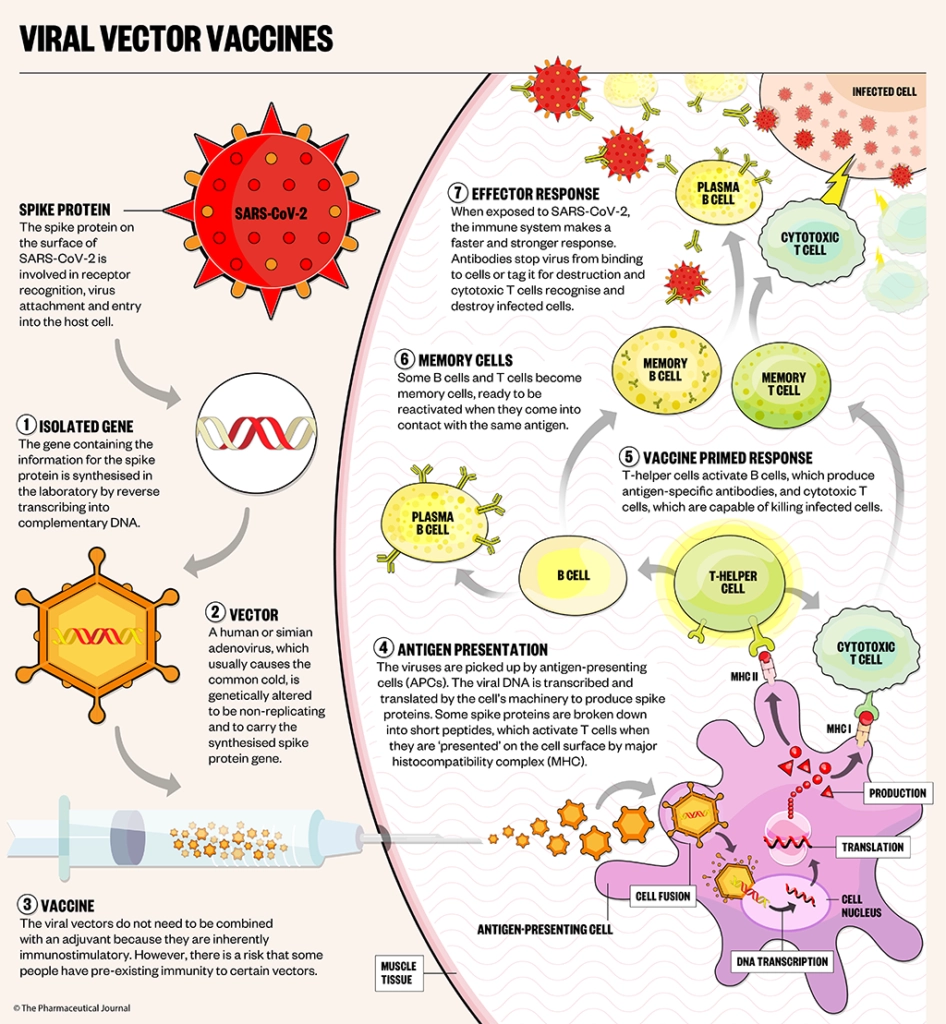
Oxford/AstraZeneca (ChAdOx1 nCoV-19 [AZD1222], brand name Vaxzevria)
Is it approved in the UK?
Yes, it was authorised for temporary supply on 30 December 2020 and now has a conditional marketing authorisation.
How many doses have been ordered?
100 million.
What are the storage requirements?
- Store at 2–8ºC. Shelf life is 6 months.
- After opening, store at 2–25˚C and use within 6 hours.
What are the latest data on effectiveness?
Data released as a pre-print in The Lancet on 2 February 2021 suggest vaccine efficacy against symptomatic infection is 76% (95% CI 59–86) after a single dose in individuals aged 18–55 years, increasing to 82.4% (95% CI 62.7–91.7) after a second dose administered 12+ weeks later. There were no hospitalisations or deaths in the vaccine group.
The latest consensus judgement of the Vaccine Effectiveness Expert Panel (27 August 2021), which comprises scientific and analytical specialists from academia and government in the UK, is that protection against symptomatic disease from the currently circulating variant (Delta) after two doses of the Oxford/AstraZeneca vaccine is around 70%. Protection against hospitalisation and death is about 95% after two doses.
However, there is some evidence that vaccine efficacy begins to wane after six months. Vaccine efficacy against symptomatic infection wanes to just over 50% after 20+ weeks Waning against hospitalisation appears to be more limited, with the Oxford/AstraZeneca vaccine efficacy falling to just under 80% beyond 20+ weeks, according to PHE. There is some suggestion of greater waning in the oldest age groups from 20+ weeks, however confidence intervals are wide. For those in the the 40-64 years age group, the waning is restricted to those in clinical risk groups. Waning appears to be greater among those in the clinically extremely vulnerable group, though data beyond 20 weeks is limited.
Analysis carried out by the ZOE COVID study, which investigates real-world vaccine effectiveness, found that the protection provided by two doses of the Oxford/AstraZeneca vaccine wanes within six months – from 77% protection against infection a month after the second dose to 67% after four to five months.
Initial results from the OCTAVE trial, published as a pre-print on 23 August 2021, showed that 40% of clinically at-risk patients with specific immunocompromised or immunosuppressed conditions mounted a low or undetectable immune response after two doses of the Pfizer/BioNTech or Oxford/AstraZeneca vaccine.
Does it work against new variants?
Yes and no.
Alpha (B.1.1.7; first detected in UK): Real-world data from the UK vaccination programme, published as pre-prints on 1 March 2021 and 23 April 2021, suggest there is high vaccine efficacy against the UK variant. And study data published in The Lancet on 30 March 2021 show that, although neutralising antibody titres generated by the vaccine were nine-fold lower for the UK variant, vaccine efficacy against symptomatic cases was similar for Alpha and non-Alpha lineages (70.4% [95% CI 43·6–84·5] and 80.5% [95% CI 67·9–89·4], respectively).
Beta (B.1.351, first detected in South Africa): A study published in The New England Journal of Medicine on 16 March 2021 suggests that a two-dose regimen has an efficacy of only 10.4% (95% CI −76.8 to 54.8) against mild to moderate COVID-19 infection in almost 1,500 HIV-negative young adults. Efficacy against severe COVID-19 infection from this variant was not assessed.
Gamma (P1, first detected in Japan/Brazil): A small laboratory study using vaccine sera suggests antibody titres were reduced by 2.9-fold against this variant. The impact is yet to be confirmed in clinical trials.
Delta (B.1.617.2, first detected in India): An observational study carried out by PHE, initially published as a pre-print but later published in The New England journal of Medicine on 22 July 2021, which included 4,272 people confirmed as having the Delta variant, suggested the Oxford/AstraZeneca vaccine has an efficacy of 67.0% (95% CI 61.3–71.8) against symptomatic disease from the Delta variant 2 weeks after the second dose, compared with 74.5% (95% CI 68.4–79.4) against the Alpha variant. Vaccine efficacy after one dose was 30.0% (95% CI 24.3–35.3) against the Delta variant compared with 48.7% (95% CI 45.2–51.9) against the Alpha variant.
An updated analysis, including 14,019 symptomatic cases infected with the Delta variant, was published as a pre-print by PHE on 14 June 2021. The analysis suggests vaccine efficacy against admission to hospital with the delta variant is 71% (95% CI 51–83) after one dose and 92% (95% CI 75–97) after two doses.
A Scottish study published on 14 June 2021 as a research letter in The Lancet, included 19,543 cases (7,723 with the Delta variant), of whom 377 were admitted to hospital (134 with the Delta variant). The vaccine offered 60% (95% CI 53–66) protection against the Delta variant and 73% (95% CI 66–78) against the Alpha variant. An updated analysis published as a letter in The New England Journal of Medicine on 20 October 2021 found that vaccine efficacy against death from the Delta variant 14 or more days after the second vaccine dose was 91% (95% CI 86–94).
Omicron (B.1.1.529, first detected in South Africa): Early analysis of Omicron cases in the UK suggests vaccine effectiveness against symptomatic disease is significantly lower compared with the Delta variant (less than 10% and just under 40%, 25 weeks or more after an Oxford/AstraZeneca and Pfizer/BioNTech primary course, respectively). However, from 2 weeks after a Pfizer booster dose, vaccine effectiveness increases to around 71% among those who received Oxford/AstraZeneca as the primary course and around 76% among those who received Pfizer/BioNTech as the primary course.
Does it work in older people?
Yes.
Observational data from PHE, published as a preprint on 10 May 2021, suggests that a single dose of the Oxford/AstraZeneca vaccine reduces risk of death in over 70-year-olds by 55% (0.45, 95% CI 0.34–0.59), compared with unvaccinated cases, which, when combined with the protection vaccines offer against becoming a case in the first place, is equivalent to approximately 80% protection against death. Another preprint, published by PHE on the same date, suggests, for the over 80s, one dose of the Oxford/AstraZeneca vaccine reduces the risk of hospitalisation by 73% (95% CI 60–81). In 70- to 79-year-olds, one dose reduces the risk of hospitalisation by 84% (95% CI 74–89).
A large prospective cohort study (VIVALDI) of vaccine efficacy in 10,412 residents of long-term care facilities in England, all aged 65 years and older, was published in The Lancet Infectious Diseases on 23 June 2021. Vaccine efficacy for a single dose of either the Pfizer/BioNTech or Oxford/AstraZeneca vaccines was estimated to be 56% (95% CI 19–76) at 28–34 days, and 62% (23–81) at 35–48 days post-vaccination. Similar effect sizes at 35-48 days were seen for the Pfizer/BioNTech vaccine (65% reduced risk) and Oxford/AstraZeneca vaccine (68% reduced risk).
Does it reduce transmission?
Yes, for some variants. However, studies on the transmission blocking effect of vaccines against the Delta variant are ongoing.
A study led by Public Health Scotland and the University of Glasgow, published as a pre-print on 12 March 2021 and examining Alpha infections in nearly 150,000 vaccinated and unvaccinated healthcare workers and 200,000 of their household members, provides the first direct evidence of a reduction in transmission. There was a 30% reduction in documented cases of infection in household members of vaccinated compared with unvaccinated healthcare workers (HR 0.70, 95% CI 0.63–0.78), which the researchers estimate translates to a 60% reduction in transmission since household members could be infected by other routes. A 54% reduction in documented cases in household members was observed ≥14 days after the second dose (HR 0.46, 95% CI 0.30—0.70). Participants received either the Pfizer/BioNTech vaccine or the Oxford/AstraZeneca vaccine. The study was published as a letter to the editor in NEJM on 8 September 2021.
The results of the Scottish research are backed up by a Public Health England study, published on 28 April 2021, which was carried out in a larger and wider cross-section of the population. The study found that individuals who become infected 3 weeks after receiving one dose of the Oxford/AstraZeneca or Pfizer/BioNTech vaccine are between 38% and 49% less likely to pass the virus on to their household contacts than those who were unvaccinated. The study included over 57,000 contacts from 24,000 households in which there was a lab-confirmed case that had received a vaccination, compared with nearly 1 million contacts of unvaccinated cases. The study took place when the Alpha variant was circulating.
A study in Lancet Infectious Diseases, published on 29 October 2021, suggests that fully vaccinated people (either with Pfizer/BioNTech or Oxford/AstraZeneca) can contract and pass on the Delta variant of COVID-19 in the home, but at lower rates than unvaccinated people. The study found that around 25% (95% CI 18–33) of vaccinated household contacts tested positive for COVID-19 compared with roughly 38% ( 95% CI 24–53) of unvaccinated household contacts. The researchers also found that peak viral load was similar in vaccinated and unvaccinated people.
A study conducted by the UK Health Security Agency, published as a pre-print on 25 November 2021, suggests that transmission of the delta variant is reduced by 42% (14–69) after two doses of Oxford/AstraZeneca.
Are there any safety concerns?
The safety profile generally reflects that seen in clinical trials (injection-site reactions and generalised ‘flu-like’ symptoms).
However, there is a link between the vaccine and blood clotting cases alongside low levels of platelets, particularly in younger people.
The Joint Committee on Vaccination and Immunisation (JCVI) advised adults aged 18 to 39 years old should be offered an alternative vaccine, if available.
The MHRA advises careful consideration before administering the vaccine to people at higher risk of blood clots because of a medical condition, and that a second dose should not be given to those who experienced this adverse event after the first dose. Vaccine recipients should also be advised to look out for symptoms four days or more after vaccination, including new onset of severe or persistent headache, blurred vision, confusion or seizures, shortness of breath, chest pain, leg swelling or persistent abdominal pain, and unusual skin bruising or pinpoint round spots beyond the injection site.
As of 8 December 2021, the MHRA had received Yellow Card reports of 428 cases of blood clots with low platelets following the vaccine, with a case fatality rate of 17%. The estimated number of first doses administered in the UK by 8 December 2021 was 24.9 million, giving an overall case incidence of 15.3 per million doses.
On 11 June 2021, the European Medicines Agency announced that its safety committee has concluded that people who have previously had capillary leak syndrome must not be vaccinated with the Oxford/AstraZeneca vaccine and that capillary leak syndrome should be added to the product information as a new side effect of the vaccine. The Medicines and Healthcare products Regulatory Agency said that it has received 14 reports of capillary leak syndrome in the context of more than 49.0 million doses of the vaccine given, and the current evidence does not suggest that the syndrome is caused by the vaccine. However, the MHRA is carefully considering whether or not precautionary advice is warranted for patients who have previously experienced capillary leak syndrome. On 16 July 2021, the MHRA added previous episodes of capillary leak syndrome as a contraindication.
The MHRA has added a precautionary warning to product information that Guillain-Barré Syndrome (GBS) has very rarely been reported following vaccination. As of 8 December 2021, the MHRA had received Yellow Card reports of 472 cases of GBS following vaccination with the Oxford/AstraZeneca vaccine. The EMA listed GBS as a very rare side effect on 8 September 2021.
Full product information can be viewed here.
Janssen (Ad26.COV2.S)
Is it approved in the UK?
Yes, it was granted a conditional marketing authorisation on 28 May 2021.
How many doses have been ordered?
30 million.
What are the storage requirements?
Store at -20ºC for 2 years or 2–8ºC for 3 months.
What are the latest data on effectiveness?
Results from a phase III trial published in The New England Journal of Medicine on 21 April 2021 suggest vaccine efficacy is 66.1% (95% CI 55.01; 74.80) overall against moderate to severe/critical disease (72.0% [95% CI 58.2–81.7] in the United States) 28 days after vaccination. It was 85.4% (95% CI 54.15–96.90) effective in preventing severe/critical disease. Vaccine efficacy against COVID-19 requiring medical intervention (defined as hospitalisation, intensive care unit admission, mechanical ventilation, extracorporeal membrane oxygenation) was 100.0% (95% CI 74.3–100.0) at least 28 days after vaccination.
In a letter to the editor, published in The New England Journal of Medicine on 14 July 2021, data from a small number of individuals suggests that the vaccine elicited durable humoral and cellular immune responses with minimal decreases for at least 8 months after immunisation.
Real-world data from the US vaccination programme, published as pre-print on 16 September 2021, show that, among 390,517 vaccinated and 1,524,153 matched unvaccinated individuals, vaccine efficacy was 79% (95% CI 77–80) for COVID-19 and 81% (79–84) for COVID-19-related hospitalisations. Among states with high Delta variant incidence, vaccine efficacy remained high, at 78% (73–82) for infections and 85% (95% CI 73–91) for hospitalisations.
Interim data from a phase I/IIa study, announced by Janssen on 21 September 2021, show that, when a second dose is given six months after the single shot, antibody levels increased nine-fold one week after the booster and continued to climb to 12-fold higher four weeks after the booster, with all rises irrespective of age. And recent results from the phase III ENSEMBLE 2 study that found a booster dose given 56 days after the primary dose provided 94% (95% CI 58–100) protection against symptomatic (moderate to severe/critical) COVID-19 and 100% (95% CI 33–100) protection against severe/critical COVID-19, at least 14 days post-booster.
Does it work against new variants?
Yes, although more data are needed.
Alpha (B.1.1.7; first detected in UK): A real-world study, published as a pre-print on 30 April 2021 and conducted in the US when Alpha was the predominant variant circulating, suggests that, among 1,779 vaccinated individuals matched with 17,744 unvaccinated controls, vaccine effectiveness against SARS-CoV-2 infection was 76.7% (95% CI 30.3–95.3) two weeks after vaccination.
Beta (B.1.351, first detected in South Africa): Phase III trial data suggest vaccine efficacy is 64.0% (95% CI 41.2–78.7) against moderate to severe/critical disease, and 81.7% (95% CI 46.2–95.4) against severe/critical disease.
Gamma (P1, first detected in Japan/Brazil): Phase III trial data suggest vaccine efficacy is 68.1% (95% CI 48.8–80.7) against moderate to severe/critical disease and 87.6% (95% CI 7.8–99.7) against severe/critical disease, although Gamma was detected in 30.6% of sequences and P2 was detected in 69.4% of sequences.
Delta (B.1.617.2, first detected in India): A small laboratory study, published as a pre-print on 1 July 2021, using vaccine sera from eight participants suggests neutralising antibody titres against the Delta variant are reduced 1.6-fold compared with the B1 strain. However, a different laboratory study, published as a pre-print on 19 July 2021, showed a more pronounced reduction in neutralising antibody titres of 5.5-fold.
Data in a letter to the editor, published in The New England Journal of Medicine on 14 July 2021, reveal an expansion of neutralising antibodies against the Delta variant over eight months, which the company says suggests maturation of B-cell responses even without further boosting.
And results from a South African study called Sisonke, announced by South Africa’s ministry of health on 6 August 2021, suggest the vaccine was highly effective in preventing severe illness and death from both Delta and Beta variants in almost 500,000 health care workers, with an efficacy of up to 95% against death from the Delta variant, and up to 71% against hospitalisation.
Omicron (B.1.1.529, first detected in South Africa): No evidence yet.
Does it work in older adults?
Yes, although more data are needed.
Phase III trial data suggest a vaccine efficacy of 67.9% (95% CI 38.2–82.8) 28 days after vaccination in participants aged ≥60 years, although participants aged ≥60 years without comorbidities had a higher vaccine efficacy estimate than participants aged ≥60 years with comorbidities. Further follow-up data are needed to help understand these potential differences.
Does it reduce transmission?
Probably, but we need more data.
Phase III trial data show the vaccine reduced the risk of infection in a subgroup of 2,650 participants by 65.5% (95% CI 39.91–81.08), suggesting an impact on transmission, but this finding needs to be further investigated with additional data.
Are there any safety concerns?
The safety profile in clinical trials includes injection-site reactions and generalised ‘flu-like’ symptoms. However, some safety signals have emerged.
Administration of the Janssen vaccine in the US was temporarily paused on 13 April 2021 while the US Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC) review data on six reported cases of blood clots with low platelets after receiving the vaccine among more than 6.8 million doses administered. Use of the vaccine resumed on the recommendation of the FDA and CDC on 24 April 2021, which said that a warning should now be included on the vaccine label about an elevated, but rare, risk of blood clots with low platelets. As of 12 May 2021, the CDC had identified 28 cases of blood clots with low platelets among more than 8.7 million people vaccinated.
The European Medicines Agency (EMA) has also reviewed four cases of blood clots and low platelets among vaccine recipients in clinical trials and the US vaccination programme. On 20 April 2021, the EMA concluded that the overall benefit/risk ratio for the vaccine remains positive but said that a warning about unusual blood clots with low platelets should be added to the product information and that these events should be listed as very rare side effects of the vaccine.
On 9 July 2021, the EMA announced that its safety committee has concluded that people who have previously had capillary leak syndrome must not be vaccinated with the Janssen vaccine and that capillary leak syndrome should be added to the product information as a new side effect of the vaccine. And on 22 July 2021, the EMA’s safety committee recommended that Guillain-Barré syndrome be added as a vary rare side effect to product information after assessing available data and concluding that a causal relationship between the vaccine and GBS is possible. On 7 August 2021, the EMA recommended adding immune thrombocytopenia, dizziness and tinnitus as adverse reactions. And on 1 October 2021, the EMA recommended listing venous thromboembolism (VTE) as a rare side effect together with a warning to raise awareness among healthcare professionals and people taking the vaccine, especially those who may have an increased risk of VTE.
Full product information can be viewed here.
Protein-based vaccines
Protein-based vaccines are made using an older technology, which delivers SARS-CoV-2 spike proteins directly into the body, along with an adjuvant, priming the immune system to attack the SARS-CoV-2 virus if it later infects the body (see Figure).
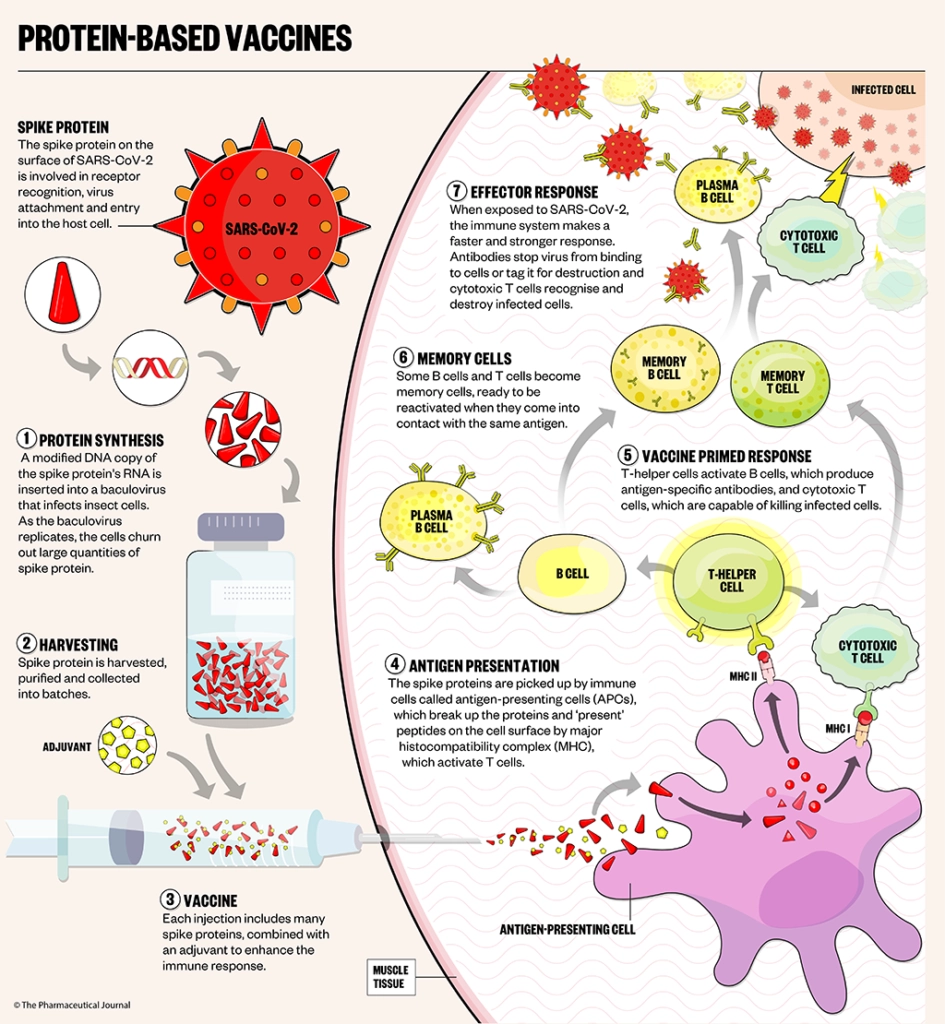
Novavax (NVX-CoV2373)
Is it approved in the UK?
No.
How many doses have been ordered/pre-ordered?
60 million.
What are the storage requirements?
Store at 2–8 ºC.
What are the latest data on effectiveness?
Data from a final analysis of a phase III UK trial involving more than 15,000 participants, announced by Novavax on 15 March 2021, suggest vaccine efficacy is 89.7% (95% CI 80.2–94.6) against symptomatic infection at least seven days after the second dose. No severe cases occurred in the vaccine group compared with five in the placebo group. The study was published in The New England Journal of Medicine on 30 June 2021.
Novavax announced preliminary data from a phase III trial of its vaccine in almost 30,000 participants in Mexico and the US on 14 June 2021. The study found that the vaccine demonstrated 90.4% efficacy overall (95% CI 82.9–94.6), with 63 mild cases in the placebo group and 14 mild cases in the vaccine group. Protection was 100% (95% CI 87.0–100) against moderate and severe disease, with ten moderate cases and four severe cases, all in the placebo group. Efficacy against variants of concern/interest was 93.2% (95% CI 83.9–97.1). The study was published in The New England Journal of Medicine on 15 December 2021.
On 5 August 2021, Novavax announced that a single booster dose at 6 months increased wild-type neutralising antibodies more than 4-fold, and cross-reactive functional antibodies to Delta 6-fold, versus primary vaccination series.
Does it work against new variants?
Yes, but vaccine effectiveness may be reduced against some variants and more data are needed.
Alpha (B.1.1.7; first detected in UK): Data from Novovax suggest a vaccine efficacy of 86.3% (95% CI 71.3–93.5) against symptomatic infection.
Beta (B.1.351, first detected in South Africa): Data from Novovax suggest a vaccine efficacy of 48.6% (95% CI 28.4–63.1) against symptomatic infection, 55.4% (95% CI 35.9–68.9) in HIV-negative participants.
Data from a phase IIb study published in The New England Journal of Medicine on 5 May 2021, suggest a vaccine efficacy among 2,684 participants of 49.4% (95% CI 6.1–72.8) at least 7 days after the second dose, with all cases classed as mild-to-moderate except one severe case in the placebo group. Among HIV-negative participants, vaccine efficacy was 60.1% (95% CI 19.9–80.1). The B.1.351 variant accounted for 92.7% of sequenced cases.
Gamma (P1, first detected in Japan/Brazil): No evidence yet.
Delta (B.1.617.2, first detected in India): No evidence yet.
Omicron (B.1.1.529, first detected in South Africa): No evidence yet.
Does it work in older people?
Yes, although more data are needed.
Just over a quarter (27%) of trial participants were aged >65 years; there was one case of COVID-19 in the vaccine group compared with nine in the placebo group.
The results of a phase III trial of almost 30,000 participants in Mexico and the US, announced by Novavax on 14 June 2021, indicated vaccine efficacy was 91.0% (95% CI 83.6–95.0) among “high-risk” populations (defined as over age 65, under age 65 with certain comorbidities or having life circumstances with frequent COVID-19 exposure), with 62 cases in the placebo group and 13 in the vaccine group.
Does it reduce transmission?
We don’t know yet.
Animal studies suggest transmission can be prevented entirely but this has not yet been confirmed in humans.
Are there any safety concerns?
Analyses of the UK and South Africa trials showed that the vaccine is well-tolerated, with low levels of severe, serious and medically attended adverse events at day 35, balanced between vaccine and placebo group. The same was found for the US/Mexico trial.
Full product information is not yet available.
GSK/Sanofi (CoV2 preS dTM)
Is it approved in the UK?
No.
How many doses have been ordered/pre-ordered?
60 million.
What are the storage requirements?
Store at 2–8ºC.
What are the latest data on effectiveness?
GSK and Sanofi announced on 17 May 2021 that their vaccine candidate has demonstrated strong immune responses across all adult age groups in a phase II trial involving 722 participants. Seroconversion after two doses was between 95% and 100%, with higher levels of neutralising antibodies observed in younger adults (18 to 59 years old). After a single injection, high neutralising antibody levels were generated in participants with evidence of prior SARS-CoV-2 infection.
The news comes after the partnership was forced to reformulate the vaccine following a weaker response in older people owing to an insufficient concentration of the antigen in the vaccine.
Does it work against new variants?
We don’t know yet.
Alpha (B.1.1.7; first detected in UK): No evidence yet.
Beta (B.1.351, first detected in South Africa): No evidence yet.
Gamma (P1, first detected in Japan/Brazil): No evidence yet.
Delta (B.1.617.2, first detected in India): No evidence yet.
Omicron (B.1.1.529, first detected in South Africa): No evidence yet.
Does it work in older adults?
Not in early trials.
Phase I/II trial results announced by GSK/Sanofi showed insufficient neutralising antibody titres in adults over the age of 50 years. The interim analysis was published in The Lancet on 19 April 2021.
Does it reduce transmission?
We don’t know yet.
Data not yet available.
Are there any safety concerns?
Data not yet available.
Full product information is not yet available.
Panel: Advice on vaccinating children, and on the booster campaign
The Joint Committee on Vaccination and Immunisation (JCVI) advises that all young people aged 12 to 15 years of age should be offered two doses of the Pfizer/BioNTech vaccine, 12 weeks apart, reduced to at least 8 weeks between doses if the emerging epidemiological data supports this. Children aged 16 and 17 years should be offered a second dose 12 weeks after the first. Children who have had a proven SARS-CoV-2 infection should wait at least 12 weeks before receiving a dose of the vaccine.
Children aged 5 to 11 years in a clinical risk group or who are a household contact of someone who is immunosuppressed should be offered two 10mcg doses of the Pfizer/BioNTech with an interval of 8 weeks between the first and second doses. The minimum interval between any vaccine dose and recent COVID-19 infection should be 4 weeks.
All children aged 16 and 17 years old, children aged 12 to 15 who are in a clinical risk group or who are a household contact of someone who is immunosuppressed, and children aged 12 to 15 years who are severely immunosuppressed and have had a third primary dose, should be offered a 30mcg booster dose of Pfizer/BioNTech no earlier than 3 months after completion of the primary vaccine course.
The JCVI advises that a booster vaccine dose is offered no earlier than 3 months after completion of the primary vaccine course to all adults aged 18 years or over, with Pfizer/BioNTech and Moderna being given equal preference.
Severely immunosuppressed individuals who have completed their primary course (3 doses) should be offered a booster dose with a minimum of 3 months between the third primary and booster dose.
Latest:
- The panel on vaccinating children, and on the booster campaign, was updated on 22 December 2021 to reflect new JCVI advice.
- The Oxford/AstraZeneca, Pfizer/BioNTech and Moderna entries were updated on 22 December 2021 to include the latest data on cases of blood clots with low platelets, and cases of myocarditis and pericarditis, and Guillain-Barré syndrome
- The Oxford/AstraZeneca and Pfizer/BioNTech entries were updated on 15 December 2021 to include the latest analysis on vaccine effectiveness against the Omicron variant.
- The Oxford/AstraZeneca and Pfizer/BioNTech entries were updated on 7 December 2021 to include a new study on vaccine effectiveness at reducing transmission of the Delta variant.
- Advice on vaccinating children and on the booster campaign was updated on 7 December 2021 to reflect recent JCVI guidance.
- The Pfizer/BioNTech entry was updated on 22 November 2021 to include a study on the effect of a booster dose.
- The Pfizer/BioNTech and Oxford/AstraZeneca entries were updated on 5 November 2021 to include a study on vaccine efficacy against transmission of the Delta variant.
Illustrations: Alisdair Macdonald
Editorial advisers for the illustrations: Stephen Griffin, associate professor and chair, Virus Division, Microbiology Society, University of Leeds; Charles Bangham, chair in immunology and co-director of the Institute of Infection at Imperial College London.
Clearing the COVID-19 backlog
For views from pharmacists and nurses on the impact of COVID-19 on cancer care and treatment, see: Clearing the COVID-19 patient treatment backlog through advancing efficient and safe delivery of care (promotional content)
2 comments
You must be logged in to post a comment.



clobetasone cream is a medication used on the skin to regale enlarging, tingling, and irritation. It can assist with skin issues, for example, skin inflammation, including contact dermatitis. psoriasis. You should always use the lowest strength corticosteroid that is effective for your skin condition for the fastest possible time. It should also not be used to treat: Acne.
Very helpful in understanding mode of actions of various vaccineso