European Medicines Agency (EMA)
What can be done to boost paediatric research in the European Union?
One of the problems is a lack of financing. Paediatric research networks, which involve academic and healthcare professional groups, are usually based in hospitals. They would like to do their own research but are reliant on industry funding for trials. If they are reliant on industry support, they may not be free to research areas they are interested in.
If there is more public support for paediatric research networks, it would help to increase research into off-patent medicines that are commonly used in children, an area that has seen very little research to date.
Some funding into paediatric research is provided by the EU’s main instrument for funding research and innovation, known as the ‘Horizon 2020’ project, which will run from 2014 to 2020. But unlike its predecessor — the seventh framework programme for research and technological development (FP7) — Horizon 2020 contains no specific fund dedicated to paed-iatric medicinal research.
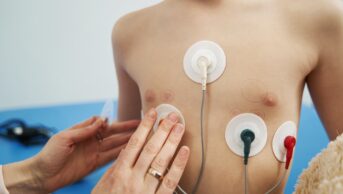
Paediatricians are hesitating in encouraging parents to volunteer their children into studies because there is, understandably, a societal reluctance to include children in research.
Over the past year, the Paediatric Committee (PDCO) [part of the European Medicines Agency] has worked with paediatricians to raise awareness of the benefits of doing clinical research on children rather than give them off-label medicines, i.e. drugs that don’t have an approved licensed indication in children because of the risks of inefficacy and side effects. Paediatricians are hesitating in encouraging parents to volunteer their children into studies because there is, understandably, a societal reluctance to include children in research.
We have to educate patients and children’s organisations more about the benefits of participating in clinical trials so that they are more accepting of them. Some member states have addressed this by improving awareness through education programmes for healthcare professionals, but paediatricians can do more, too, by persuading parents to ask for clinical trials for their children.
Since the 2007 Paediatric Regulation, companies filing applications for new drugs gain an extra six months of patent protection if they do studies in children according to what is agreed in a paediatric investigation plan (PIP). Are PIPs a success story?
More than 1,600 PIPs have been submitted to the PDCO in nearly all system organ classes (according to the MedDRA classification). The PIP is a success story as companies have been submitting them because they are mandated by law to consider the needs of children every time they develop any new medicine for an adult.
Large pharmaceutical companies have been actively considering paediatric needs for any new drug developments because they do submit the PIPs. But it will be seven to ten years (starting from 2007) before those new drugs come on to the market after completing a PIP. Therefore, the real effect of our work will come in the next few years.
The 2007 regulation also brought in an incentive for companies developing orphan medicines for diseases affecting fewer than 5 in 10,000 children. Companies receive an extra two years of
marketing exclusivity added to the existing ten years awarded
under the EU’s orphan regulation. Is that a success?
For orphan medicines, we are seeing the number of applications increasing, but I don’t think the two years’ additional marketing exclusivity is such a successful incentive for the industry because orphans are not blockbuster drugs that have huge sales.
More than 50% of the medicines used for children have not been tested in this age group. Through the Paediatric Regulation, companies were offered an incentive of eight years of data and ten years of marketing exclusivity to undertake research into older, off-patent medicines used in children through a paediatric use marketing authorisation (PUMA). How successful is the scheme?
The return on investment for pharmaceutical companies to develop an off-patent medicinal product for a PUMA application is not clear. The companies face uncertainties in the reimbursement of these older medicines by EU member states. To date, only one PUMA has been granted: for midazolam (Buccolam), an oromucosal solution licensed for status epilepticus, in 2011.
Although the paediatric committee scientifically assesses a PUMA application for an old drug that has lost its patent and may be used in children off-label in the EU, it has little say on its market access. It is the national reimbursement bodies in the EU that decide whether the drug will be funded or not.
The risk of this uncertainty may be too high for the company not knowing whether the expenses for developing its drug will be at least covered by obtaining full reimbursement in the EU.
There is a gap between what is approved by the PDCO and what is decided by member states when it comes to reimbursement. The gap needs to be closed.
Is it because companies fear that getting marketing exclusivity
will not prevent doctors from continuing to use competitor products with the same active ingredient off-licence at lower costs or that substitution for cheaper adult forms takes place at the pharmacy level?
Yes, it seems that this is a concern. For instance, in Germany, when you go to the pharmacy and ask for a drug on prescription, the pharmacist can actually give out the cheapest drug of the same active substance without seeking the doctor’s approval.
Although you have a drug, which is the best for the child, it may not be given out because of the generic substitution by the pharmacist. The paediatricians prescribing these drugs have no influence on this. And of course the companies realise this in terms of their return of investment.
Is it important that the PUMA scheme works?
The largest proportion of medicines used in children are off-patent drugs. At least 50% of medicines used in children are off-patent medicines, which have been rarely tested in children. We are only now starting to see new medicines with approved paediatric indications coming to the market, but this has been a slow process.
What can we do to make PUMAs work?
There needs to be some reassurance made to companies that are developing paediatric off-patent drugs that they will see some return on their investment. We also need to reduce the regulatory requirements for PUMAs, so they are less burdensome compared with drug applications of new molecules, while still obtaining the necessary data on efficacy, quality and safety.
The PDCO/EMA plans to include these concepts in the next report on the success of the paediatric regulation in 2017. One issue the PDCO would like to raise with the commission is about new innovative drugs (for example, first in class) that are identified at the clinical trial phase I stage. For some of these drugs, little scientific information is available at this stage and the PDCO needs to find ways to agree on a paediatric investigation plan (PIP) with the company without causing delays by over-analysing inclusion criteria, endpoints and study populations at this early stage.
Regulators will never stop off-label use, which includes a product that has received a PUMA and is not being reimbursed. I think what we need to do is to pinpoint this problem to the lawmakers of the member states, who are looking at the reimbursement of medicines. If this is not being aligned to what the PDCO is working for at the European level, then the PUMA will never be a success story.
Interview by
Elizabeth Sukkar