John Bavosi / Science Photo library
Chronic obstructive pulmonary disease (COPD) is the only major cause of death with an increasing incidence. It is expected to be the third leading cause of death worldwide by 2030 and 2020 projections rank it fifth worldwide in terms of disease burden[1]
. In the UK, there are around 835,000 people who have been diagnosed with COPD and an estimated 2.2 million people with COPD who remain undiagnosed[2]
,[3]
. This underdiagnosis is partly because of a patient’s acceptance of the condition as a normal part of ageing; it has aptly been described as “a story with no beginning, a middle that is a way of life and an uncertain and unlooked for end”[4]
. Underdiagnosis is also partly a result of the complexity of reaching a definitive diagnosis.
COPD is characterised by the presence of incompletely reversible airflow obstruction (defined as a post-bronchodilator forced expiratory volume in one second [FEV1]/forced vital capacity [FVC] <0.70) and, while this may underestimate the presence of airflow obstruction in young people and overestimate it in older people[5]
, it remains current practice in the UK. This airflow limitation is usually progressive and was first described by Fletcher and Peto in 1977 as “an accelerated lung decline”[6]
. This has been challenged by more recent cohort data that suggest patients display differences in their rate of decline, some declining very quickly and others not declining faster than age would predict[7]
. These data have also added complexity by reinforcing the concept of susceptibility to COPD through parental smoking and its effect on lung development.
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) collaboration[8]
suggest that a clinical diagnosis of COPD should be considered in any patient who has dyspnoea (breathlessness), chronic cough and/or sputum production, but confirmation of COPD always requires spirometry[8]
. Current guidelines from the National Institute for Health and Care Excellence (NICE)[9]
, England’s health technology assessment body, use spirometric classification of severity, as determined by a comparison of the patient’s FEV1 to that predicted for their age, height and gender. It describes their disease, ranging from “mild” to “very severe” (see ‘Table 1: Post-bronchodilator FEV1 value and associated COPD stage’).
| Table 1: Post-bronchodilator FEV1 value and associated COPD stage | |
|---|---|
| Post-bronchodilator FEV1 | COPD stage |
| >80% predicted | Mild |
| 50–80% predicted | Moderate |
| 49–30% predicted | Severe |
| <30% predicted | Very severe |
The correlation between FEV1 and health status is weak[10]
,[11]
and yet most therapeutic intervention studies focus on improvement of FEV1 as a primary end point. Staging of COPD based on FEV1 misses important features that impact on the health status of patients, such as frequency of exacerbations (acute worsening of symptoms that necessitate oral prednisolone and occasionally antibiotic therapy), functional limitation, and presence of comorbidities. This has been addressed somewhat by the GOLD group[8]
, but adoption in the UK has been slow.
COPD is an umbrella term encompassing a range of disorders: chronic bronchitis, emphysema and, in some definitions, variants of asthma[12]
. Combining these subgroups may make diagnosis simpler, but it adds significant complexity to the resultant management because the pharmacological strategies for each type differ. It also makes the interpretation of clinical trials difficult, as they usually include a heterogeneous population under the umbrella of COPD.
Phenotypes in COPD
The development of different options for pharmacological and non-pharmacological treatments for patients with COPD has led to the realisation that clinical response differs according to the characteristics of the disease. The concept of phenotype (defined as the observable characteristics of a subject as determined by the interaction of its genotype with the environment) applied to COPD has resulted in the definition of different types of patients with prognostic and therapeutic significance.
Therefore, in the same way that a patient would not be described as having arthritis without further clarification, there is a move towards better characterisation of patients using their phenotype[13]
.
In this way, we may take on a more personalised approach to treatment according to not only the severity of the airflow obstruction, but also modified by the clinical phenotype. Therefore, from a clinical viewpoint, a COPD phenotype should separate patients into distinct categories that differentiate their prognosis and response to treatment that can produce clinically meaningful outcomes.
This concept has been described by Agusti et al.[10]
and is presented in ‘Figure 1: COPD populations, the subsequent concept of phenotypes and the real world model of treatable characteristics’. COPD is a heterogeneous disease (as represented by the different colours in figure 1A), so the concept of a clinical phenotype attempted to provide some order by grouping patients with similar clinically relevant characteristics (figure 1B). In real life, patients with COPD are not only heterogeneous but also complex, indicating that different clinical characteristics can occur in varying proportions in any given patient (figure 1C). However, recognition of the predominant characteristics within an individual patient using phenotypes is a useful tool in better understanding their disease and tailoring treatment to it.
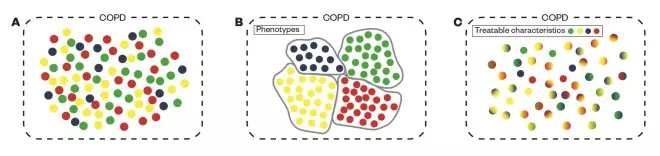
Figure 1: COPD population, the subsequent concept of phenotypes and the real world model of treatable characteristics
Source: Reproduced with permission from http://thorax.bmj.com/content/69/9/857
Current practice is illustrated in figure A, the idealistic separation of COPD types per phenotype in B, then the real life situation of patients with multiple features of the disease in C. However, recognition of the predominant characteristics within an individual patient using phenotypes is a useful tool in better understanding their disease and tailoring treatment to it.
There are many potential COPD phenotypes, possibly as many as there are patients with COPD[14]
, and there is no consensus currently. Potential classifications can be based on disease attributes, such as symptoms[15]
, long-term prognosis[16]
, by cluster analysis[17]
,[18]
or combining different features[19]
. Several recent reviews have covered the extensive literature around phenotyping[20]
,[21]
. However, for a classification of subgroups of COPD to be clinically useful, it should be simple for the healthcare professional to apply and be potentially responsive to different therapeutic interventions (so called ‘treatable traits’). Three basic phenotypes based on the predominant symptom profile have been proposed as a possible simple classification[22]
,[23]
:
- Predominant chronic breathless phenotype (may have emphysema-hyperinflation);
- Predominant frequent exacerbator phenotype (may have chronic bronchitis +/- chronic sputum production);
- Asthma overlap phenotype (mixed COPD-asthma).
This phenotypic classification may be useful because the predominant characteristic helps determine the main therapeutic options.
Predominant chronic breathless phenotype (emphysema-hyperinflation)
Emphysema is permanent destructive enlargement of the airspaces distal to the terminal bronchioles without obvious fibrosis[24]
. The main feature of this phenotype is that the main symptom is breathlessness on exertion but there may not be frequent exacerbations (≤2 exacerbations/year requiring treatment with oral steroids and/or antibiotics). Patients with emphysema who do have frequent exacerbations would be classified as predominant frequent exacerbators.
These patients usually have a significant smoking history and full lung function shows reduced gas transfer capacity (DLCO) and hyperinflation (increased residual volume [RV]/total lung capacity [TLC] ratio). However, studies have demonstrated that up to 24% of smokers who have a normal FEV1/FVC ratio may have evidence of emphysema or gas trapping on high resolution computed tomography (CT)[25]
,[26]
and therefore would not be considered to have COPD, but may still have symptoms of breathlessness (and for these people there are no guidelines).
Many studies have demonstrated that breathlessness[27]
, reduced exercise[28]
and hyperinflation[29]
predict mortality independent of FEV1 defined severity[30]
. Therefore, reduction in hyperinflation and gas trapping may be a more relevant therapeutic target than just improvement in FEV1.
Hyperinflation can be reduced by bronchodilation with only minor improvements to flow[31]
,[32]
. It is also improved by long-acting muscarinic antagonists (LAMA)[33]
and a long-acting B2 -agonist (LABA)[34]
. This strategy may have additional benefits in terms of improvement in FEV1, reduction in need for rescue treatment and improvement in quality of life, particularly when used together[35]
,[36]
,[37]
,[38]
. Long acting bronchodilators are the cornerstone of pharmacological treatment of COPD, improving breathlessness, exercise capacity and quality of life.
In addition, non-drug treatments, such as pulmonary rehabilitation[23]
,[39]
,[40]
, education on breathing techniques[41]
aimed at reducing dynamic hyperinflation, or surgical techniques aimed at reducing the volume of localised areas of hyperinflated lung, may also be useful to specially selected patients within this phenotype[42]
,[43]
.
Predominant exacerbator phenotype
An exacerbation of COPD can be defined as a change in the patient’s baseline dyspnoea, cough, and/or sputum that is beyond normal day-to-day variations, being acute in onset, and warranting a change in regular medication — usually a short course of oral steroids and occasionally antibiotics. Exacerbation frequency and severity vary between patients. The exacerbator phenotype is defined as patients with fixed airflow obstruction who have two or more exacerbations per year (with the exacerbations at least four weeks apart to ensure that they are separate episodes rather than a persisting single event) or one hospitalised exacerbation[44]
. A frequent exacerbator is considered to be someone who has two or more exacerbations per year[8]
.
The risk of an exacerbation is poorly correlated with their severity of disease (as classified by FEV1), but highly correlated with having had previous exacerbations[44]
. The study also found that up to 23% of patients had no exacerbations, further confirming the need to ascertain exacerbation history, rather than rely on severity as a prompt for treatment[44]
. The importance of exacerbations is three-fold: exacerbations adversely affect the patient’s quality of life and risk deterioration to the extent the patient may need hospital treatment, but also damage the lungs so much that they may never return to pre-exacerbation levels (as seen as in an abrupt decline in FEV1). Suissa et al. reported that, with advanced disease, the frequency of exacerbations increases, so targeting treatment to reduce exacerbation frequency and severity will have beneficial long-term effects on the rate of decline in lung function, morbidity and mortality[45]
.
The risk of frequent exacerbations is more closely associated with chronic mucus hypersecretion in the airways, rather than severity of airflow obstruction[46]
,[47]
. Patients with predominant exacerbator phenotype were previously described as having chronic bronchitis (expectorating purulent sputum daily for at least three consecutive months for two consecutive years). A proportion of these patients may also have associated bronchiectasis[48]
or colonisation of the airways with pathogens such as Pseudomonas complicating treatment[49]
, so antibiotic courses must be adjusted accordingly.
Treatment modalities focused on exacerbation reduction, such as flu vaccination, stopping smoking and pulmonary rehabilitation, are the cornerstones in the management of this phenotype[39]
,[50]
. In terms of medications, both LAMAs[51]
,[52]
and LABAs[53]
,[54]
have been shown to reduce the risk of exacerbations by around 25%. It should be noted that interpretation of studies of LAMAs and LABAs in COPD is complicated by allowing the continuation of inhaled steroids during the studies. The addition of inhaled corticosteroid (ICS) therapy has also been demonstrated to be beneficial in those patients who exacerbate[55]
,[56]
, and the current COPD guidelines only recommend the use of ICS in combination with a LABA. However, the benefits of adding an ICS to LAMA or LABA may only be marginal[57]
.
Long-term, low-dose macrolides seem to have an anti-inflammatory and immunomodulatory role and may be effective in reducing exacerbations in addition to triple therapy, although more data is needed to support this[58]
. Roflumilast, an oral phosphodiesterase IV inhibitor with anti-inflammatory properties, has been shown to reduce exacerbations in those with severe COPD and chronic mucus hypersecretion[59]
and is approved for use in some countries, but not in the UK.
Mixed COPD-asthma phenotype
This phenotype is the most controversial and prompts discussion. If COPD is considered a disease where the airflow limitation is irreversible or poorly reversible, it should be simple to determine which patients have COPD and which have asthma. Both the American Thoracic Society (ATS) and European Respiratory Society (ERS) define COPD as a disease state that is characterised by airflow limitation that is not fully reversible[60]
— but there is disagreement on what constitutes reversibility. Using GOLD criteria[8]
, a greater than 12% (or 200ml improvement) in FEV1 following a bronchodilator is suggestive of asthma (but does not rule out COPD). In the UK, the British Thoracic Society (BTS)/Scottish Intercollegiate Guidelines Network (SIGN) asthma guidelines suggest that >200ml or a >15% increase in FEV1 is suggestive of asthma[61]
, while the NICE COPD guidelines use an increase of >400ml in FEV1 (“marked” reversibility) as suggestive of asthma[9]
. There is also no accepted method of conducting a reversibility test, which can be either 200µg salbutamol, 400µg salbutamol, 160µg ipratropium, or both, two weeks of high dose inhaled corticosteroids, or two weeks of 30mg prednisolone.
There is no single objective test for asthma and the distinction between asthma and COPD has to be made by clinical history, the presence of atopy and/or blood/sputum eosinophilia or high concentration of exhaled nitric oxide (FeNO)[62]
. In reality, some patients may have features of both asthma and COPD, the so-called ‘asthma-COPD overlap syndrome’ (ACOS)[63],
[64]
. It is interesting to note that the majority of studies of medications in COPD (apart from TORCH[55]
) include patients who have significant response to inhaled bronchodilator but do not fully reverse.
Current estimates suggest that, depending on age, 10–52% (pooled estimate 27%) of patients classified as having COPD may actually have a mixed asthma-COPD phenotype
[63]
,[65]
. The importance of determining which patients have a mixed phenotype or predominant asthma phenotype is that these patients may benefit from inhaled corticosteroid (ICS) treatment[66]
,[67]
. Consensus currently is that patients who have features of ACOS should be treated early with a combination of LABA and ICS. Monotherapy with ICS alone is not recommended[68]
. Patients who have significant bronchodilator reversibility seem to do better with ICS[69]
,[70]
. With more widespread use of FeNO measurement, a marker of eosinophilic inflammation in lung tissue, it may become easier to identify those who have an asthmatic element that should respond to ICS.
Considering the implications of COPD phenotypes on practice
Having pooled obstructive lung diseases under the umbrella term COPD and not taking into account differences between patients to guide treatment, the disease is now being deconstructed into its component parts. Defining specific phenotypes with treatable characteristics will help guide more appropriate drug management. However, it must be understood that many patients exhibit more than one characteristic, so it is suggested that the most difficult element is identified to prioritise treatment. This has led to each phenotype being accompanied by the qualifier ‘predominant’, as other characteristics may be present. In order to classify the patient into the most appropriate phenotype, the first consideration is whether the patient has had two or more exacerbations in the past year. If they have, they fall into the predominant frequent exacerbator phenotype and treatment should prioritise the reduction of exacerbations. If the patient has not had two or more exacerbations in the past year, it should be considered whether or not they have features of asthma with significant reversibility, which will help differentiate those with predominant mixed asthma-COPD phenotype from those who may have a predominant breathlessness phenotype.
These individual COPD phenotypes are likely to respond differently to different drug therapies. This has implications for how we interpret clinical trials, in particular those based on statistically significant changes in FEV1, and importantly how the results are applied to a population with undifferentiated COPD. While some treatments may only benefit specific COPD phenotypes, few studies have investigated the subgroups of disease phenotypes in this way and although many studies have subgroup analyses available on request, most cannot provide meaningful conclusions.
In the future, studies may focus on disease phenotypes rather than COPD as a homogenous disease, but at the moment, treatment should be individualised to the patient’s treatable characteristics and address the high value interventions of influenza vaccination, support for smoking cessation and pulmonary rehabilitation.
Vincent Mak is consultant physician, respiratory integrated care, Imperial College Healthcare NHS Trust, London, and Grainne d’Ancona is principal pharmacist, respiratory/sleep medicine and clinical lecturer, Guy’s and St Thomas’ NHS Foundation Trust and King’s College London. Correspondence to:
v.mak@nhs.net
Financial and conflicts of interest disclosure:
Vincent Mak has received honoraria from AstraZeneca, GSK, Boehringer, Almirall, Novartis, TEVA and Chiesi. Grainne d’Ancona has received honoraria from AstraZeneca, GSK, Boehringer, Almirall, Pfizer, TEVA and Chiesi. No writing assistance was utilised in the production of this manuscript.
Reading this article counts towards your CPD
You can use the following forms to record your learning and action points from this article from Pharmaceutical Journal Publications.
Your CPD module results are stored against your account here at The Pharmaceutical Journal. You must be registered and logged into the site to do this. To review your module results, go to the ‘My Account’ tab and then ‘My CPD’.
Any training, learning or development activities that you undertake for CPD can also be recorded as evidence as part of your RPS Faculty practice-based portfolio when preparing for Faculty membership. To start your RPS Faculty journey today, access the portfolio and tools at www.rpharms.com/Faculty
If your learning was planned in advance, please click:
If your learning was spontaneous, please click:
References
[1] World Health Organization. Chronic obstructive pulmonary disease. Fact Sheet No. 315 (2015). Available at: http://www.who.int/mediacentre/factsheets/fs315/en/ (accessed May 2016).
[2] Shahab L, Jarvis MJ, Britton J et al. Prevalence, diagnosis and relation to tobacco dependence of chronic obstructive pulmonary disease in a nationally representative population sample. Thorax 2006;61(12):1043–1047. doi: 10.1136/thx.2006.064410
[3] Department of Health. An outcomes strategy for chronic obstructive pulmonary disease and asthma in England 2011. Available at: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/216139/dh_128428.pdf (accessed May 2016).
[4] Pinnock H, Kendall M, Murray SA et al. Living and dying with severe chronic obstructive pulmonary disease: multi-perspective longitudinal qualitative study. The BMJ 2011;342:142–152. doi: 10.1136/bmj.d142
[5] Mohamed Hoesein FA, Zanen P & Lammers JW. Lower limit of normal or FEV1/FVC <0.70 in diagnosing COPD: an evidence-based review. Respiratory Medicine 2011;105(6):907–915. doi: 10.1016/j.rmed.2011.01.008
[6] Fletcher C & Peto R. The natural history of chronic airflow obstruction. The BMJ 1977;1(6077):1645–1648. doi: 10.1136/bmj.1.6077.1645
[7] Lange P, Celli B, Agustà A et al. Lung-function trajectories leading to chronic obstructive pulmonary disease. New England Journal of Medicine 2015;373(2):111–122. doi: 10.1056/NEJMoa1411532
[8] Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global strategy for diagnosis, management, and prevention of COPD — 2016. Available at: http://www.goldcopd.org/ (accessed May 2016).
[9] The National Institute for Health and Care Excellence. Chronic obstructive pulmonary disease: management of chronic obstructive pulmonary disease in adults in primary and secondary care 2010. Available at: http://publications.nice.org.uk/chronic-obstructive-pulmonary-disease-cg101 (accessed May 2016).
[10] Agusti A. The path to personalised medicine in COPD. Thorax 2014;69(9):857–864. doi: 10.1136/thoraxjnl-2014-205507
[11] Vestbo J, Agusti A, Wouters EF et al. Should we view chronic obstructive pulmonary disease differently after ECLIPSE? A clinical perspective from the study team. American Journal of Respiratory and Critical Care Medicine 2014;189(9):1022–1030. doi: 10.1164/rccm.201311-2006PP
[12] Marsh SE, Travers J, Weatherall M et al. Proportional classifications of COPD phenotypes. Thorax 2008;63(9):761–767. doi: 10.1136/thx.2007.089193
[13] Fingleton J, Weatherall M & Beasley R. Towards individualised treatment in COPD. Thorax 2011;66(5):363–364. doi: 10.1136/thx.2010.155564
[14] Rennard SI & Vestbo J. The many “small COPDs”: COPD should be an orphan disease. Chest 2008;134(3):623–627. doi: 10.1378/chest.07-3059
[15] Miravitlles M, Soler-Cataluna JJ, Calle M et al. A new approach to grading and treating COPD based on clinical phenotypes: summary of the Spanish COPD guidelines (GesEPOC). Primary Care Respiratory Journal 2013;22(1):117–121. doi: 10.4104/pcrj.2013.00016
[16] Garcia-Aymerich J, Gomez FP, Benet M et al. Identification and prospective validation of clinically relevant chronic obstructive pulmonary disease (COPD) subtypes. Thorax 2011;66(5):430–437. doi: 10.1136/thx.2010.154484
[17] Burgel PR, Paillasseur JL, Caillaud D et al. Clinical COPD phenotypes: a novel approach using principal component and cluster analyses. The European Respiratory Journal 2010;36(3):531–539. doi: 10.1183/09031936.00175109
[18] Weatherall M, Travers J, Shirtcliffe PM et al. Distinct clinical phenotypes of airways disease defined by cluster analysis. The European Respiratory Journal 2009;34(4):812–818. doi: 10.1183/09031936.00174408
[19] Rennard SI, Locantore N, Delafont B et al. Identification of five chronic obstructive pulmonary disease subgroups with different prognoses in the ECLIPSE cohort using cluster analysis. Annals of the American Thoracic Society 2015;12(3):303–312. doi: 10.1513/AnnalsATS.201403-125OC
[20] Han MK, Agusti A, Calverley PM et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. American Journal of Respiratory and Critical Care Medicine 2010;182(5):598–604. doi: 10.1164/rccm.200912-1843CC
[21] Turner AM, Tamasi L, Schleich F et al. Clinically relevant subgroups in COPD and asthma. European Respiratory Review 2015;24(136):283–298. doi: 10.1183/16000617.00009014
[22] Izquierdo-Alonso JL, Rodriguez-Gonzalezmoro JM, de Lucas-Ramos P et al. Prevalence and characteristics of three clinical phenotypes of chronic obstructive pulmonary disease (COPD). Respiratory Medicine 2013;107(5):724–731. doi: 10.1016/j.rmed.2013.01.001
[23] Miravitlles M, Calle M & Soler-Cataluna JJ. Clinical phenotypes of COPD: identification, definition and implications for guidelines. Archivos de Bronconeumologia 2012;48(3):86–98. doi: 10.1016/j.arbres.2011.10.007
[24] Symposium CG. Terminology, definitions, and classification of chronic pulmonary emphysema and related conditions. Thorax 1959;14:286–299.
[25] Bhatt SP, Sieren JC, Dransfield MT et al. Comparison of spirometric thresholds in diagnosing smoking-related airflow obstruction. Thorax 2014;69(5):409–414. doi: 10.1136/thoraxjnl-2012-202810
[26] Regan EA, Lynch DA, Curran-Everett D et al. Clinical and radiologic disease in smokers with normal spirometry. JAMA Internal Medicine 2015;175(9):1539–1549. doi: 10.1001/jamainternmed.2015.2735
[27] Martinez FJ, Foster G, Curtis JL et al. Predictors of mortality in patients with emphysema and severe airflow obstruction. American Journal of Respiratory and Critical Care Medicine 2006;173(12):1326–1334. doi: 10.1164/rccm.200510-1677OC
[28] Pinto-Plata V, Cote C, Cabral H et al. The 6â€min walk distance: change over time and value as a predictor of survival in severe COPD. European Respiratory Journal 2004;23(1):28–33. doi: 10.1183/09031936.03.00034603
[29] Diaz O, Villafranca C, Ghezzo H et al. Role of inspiratory capacity on exercise tolerance in COPD patients with and without tidal expiratory flow limitation at rest. European Respiratory Journal 2000;16(2):269–275. doi: 10.1034/j.1399-3003.2000.16b14.x
[30] Casanova C, Cote C, de Torres JP et al. Inspiratory-to-total lung capacity ratio predicts mortality in patients with chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine 2005;171(6):591–597. doi: 10.1164/rccm.200407-867OC
[31] Diaz O, Villafranca C, Ghezzo H et al. Role of inspiratory capacity on exercise tolerance in COPD patients with and without tidal expiratory flow limitation at rest. The European Respiratory Journal 2000;16(2):269–275. doi: 10.1034/j.1399-3003.2000.16b14.x
[32] Newton MF, O’Donnell DE & Forkert L. Response of lung volumes to inhaled salbutamol in a large population of patients with severe hyperinflation. Chest 2002;121(4):1042–1050. doi: 10.1378/chest.121.4.1042
[33] O’Donnell DE, Fluge T, Gerken F et al. Effects of tiotropium on lung hyperinflation, dyspnoea and exercise tolerance in COPD. European Respiratory Journal 2004;23(6):832–840. doi: 10.1183/09031936.04.00116004
[34] O’Donnell DE, Casaburi R, Vincken W et al. Effect of indacaterol on exercise endurance and lung hyperinflation in COPD. Respiratory Medicine 2011;105(7):1030–1036. doi: 10.1016/j.rmed.2011.03.014
[35] Bateman ED, Ferguson GT, Barnes N et al. Dual bronchodilation with QVA149 versus single bronchodilator therapy: the SHINE study. European Respiratory Journal 2013;42(6):1484–1494. doi: 10.1183/09031936.00200212
[36] D’Urzo AD, Rennard SI, Kerwin EM et al. Efficacy and safety of fixed-dose combinations of aclidinium bromide/formoterol fumarate: the 24-week, randomized, placebo-controlled AUGMENT COPD study. Respir Res 2014;15(1):123. doi: 10.1186/s12931-014-0123-0
[37] Decramer M, Anzueto A, Kerwin E et al. Efficacy and safety of umeclidinium plus vilanterol versus tiotropium, vilanterol, or umeclidinium monotherapies over 24 weeks in patients with chronic obstructive pulmonary disease: results from two multicentre, blinded, randomised controlled trials. The Lancet Respiratory Medicine 2014;2(6):472–486. doi: 10.1016/S2213-2600(14)70065-7
[38] Singh D, Ferguson GT, Bolitschek J et al. Tiotropium+ olodaterol shows clinically meaningful improvements in quality of life. Respiratory Medicine 2015;109(10):1312–1319. doi: 10.1016/j.rmed.2015.08.002
[39] Seymour JM, Moore L, Jolley CJ et al. Outpatient pulmonary rehabilitation following acute exacerbations of COPD. Thorax 2010;65(5):423–428. doi: 10.1136/thx.2009.124164
[40] Yoshimi K, Ueki J, Seyama K et al. Pulmonary rehabilitation program including respiratory conditioning for chronic obstructive pulmonary disease (COPD): Improved hyperinflation and expiratory flow during tidal breathing. Journal of Thoracic Disease 2012;4(3):259–264. doi: 10.3978/j.issn.2072-1439.2012.03.17
[41] de Araujo CL, Karloh M, Dos Reis CM et al. Pursed-lips breathing reduces dynamic hyperinflation induced by activities of daily living test in patients with chronic obstructive pulmonary disease: a randomized cross-over study. Journal of Rehabilitation Medicine 2016;47(10):957–962. doi: 10.2340/16501977-2008
[42] Hopkinson NS, Toma TP, Hansell DM et al. Effect of bronchoscopic lung volume reduction on dynamic hyperinflation and exercise in emphysema. American Journal of Respiratory and Critical Care Medicine 2005;171(5):453–460. doi: 10.1164/rccm.200407-961OC
[43] Lammi MR, Marchetti N & Criner GJ. Reduced dynamic hyperinflation after LVRS is associated with improved exercise tolerance. Respiratory Medicine 2014;108(10):1491–1497. doi: 10.1016/j.rmed.2014.07.015
[44] Hurst JR, Vestbo J, Anzueto A et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. New England Journal of Medicine 2010;363(12):1128–1138. doi: 10.1056/NEJMoa0909883
[45] Suissa S, Dell’Aniello S & Ernst P. Long-term natural history of chronic obstructive pulmonary disease: severe exacerbations and mortality. Thorax 2012;67(11):957–963. doi: 10.1136/thoraxjnl-2011-201518
[46] Dewan NA, Rafique S, Kanwar B et al. Acute exacerbation of COPD: factors associated with poor treatment outcome. Chest 2000;117(3):662–671. doi: 10.1378/chest.117.3.662
[47] Foreman MG, DeMeo DL, Hersh CP et al. Clinical determinants of exacerbations in severe, early-onset COPD. European Respiratory Journal 2007;30(6):1124–1130. doi: 10.1183/09031936.00009307
[48] Hurst JR, Elborn JS, De Soyza A et al. COPD-bronchiectasis overlap syndrome. European Respiratory Journal 2015;45(2):310–313. doi: 10.1183/09031936.00170014
[49] Miravitlles M, Marin A, Monso E et al. Colour of sputum is a marker for bacterial colonisation in chronic obstructive pulmonary disease. Respiratory Research 2010;11:58. doi: 10.1186/1465-9921-11-58
[50] Man SF, McAlister FA, Anthonisen NR et al. Contemporary management of chronic obstructive pulmonary disease: clinical applications. JAMA 2003;290(17):2313–2316. doi: 10.1001/jama.290.17.2313
[51] Tashkin DP, Celli B, Senn S et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. New England Journal of Medicine 2008;359(15):1543–1554. doi: 10.1056/NEJMoa0805800
[52] Vogelmeier C, Hederer B, Glaab T et al. Tiotropium versus salmeterol for the prevention of exacerbations of COPD. New England Journal of Medicine 2011;364(12):1093–1103. doi: 10.1056/NEJMoa1008378
[53] Rossi A, Khirani S & Cazzola M. Long-acting beta2-agonists (LABA) in chronic obstructive pulmonary disease: efficacy and safety. International Journal of Chronic Obstructive Pulmonary Disease 2008;3(4):521–529. doi: 10.2147/COPD.S1353
[54] Sin DD, McAlister FA, Man SF et al. Contemporary management of chronic obstructive pulmonary disease: scientific review. JAMA 2003;290(17):2301–2312. doi: 10.1001/jama.290.17.2301
[55] Calverley PM, Anderson JA, Celli B et al. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. New England Journal of Medicine 2007;356(8):775–789. doi: 10.1056/NEJMoa063070
[56] Kardos P, Wencker M, Glaab T et al. Impact of salmeterol/fluticasone propionate versus salmeterol on exacerbations in severe chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine 2007;175(2):144–149. doi: 10.1164/rccm.200602-244OC
[57] Wedzicha JA, Calverley PM, Seemungal TA et al. The prevention of chronic obstructive pulmonary disease exacerbations by salmeterol/fluticasone propionate or tiotropium bromide. American Journal of Respiratory and Critical Care Medicine 2008;177(1):19–26. doi: 10.1164/rccm.200707-973OC
[58] Pomares X, Monton C, Espasa M et al. Long-term azithromycin therapy in patients with severe COPD and repeated exacerbations. International Journal of Chronic Obstructive Pulmonary Disease 2011;6:449–456. doi: 10.2147/COPD.S23655
[59] Wedzicha JA, Calverley PM & Rabe KF. Roflumilast: a review of its use in the treatment of COPD. International Journal of Chronic Obstructive Pulmonary Disease. 2016;11:81–90. doi: 10.2147/COPD.S89849
[60] Celli BR, MacNee W & Force AET. Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. European Respiratory Journal 2004;23(6):932–946. doi: 10.1183/09031936.04.00014304
[61] British Thoracic Society (BTS)/Scottish Intercollegiate Guidelines Network (SIGN). British Guideline on the Management of Asthma: a national clinical guideline 2014 [13/12/14]. Available at: https://www.brit-thoracic.org.uk/document-library/clinical-information/asthma/btssign-asthma-guideline-2014/ (accessed May 2016).
[62] Essat M, Harnan S, Gomersall T et al. Fractional exhaled nitric oxide for the management of asthma in adults: a systematic review. The European Respiratory Journal 2016;47(3):751–768. doi: 10.1183/13993003.01882-2015
[63] Alshabanat A, Zafari Z, Albanyan O et al. Asthma and COPD Overlap Syndrome (ACOS): a systematic review and meta analysis. PloS One 2015;10(9):e0136065. doi: 10.1371/journal.pone.0136065
[64] Postma DS & Rabe KF. The Asthma-COPD Overlap Syndrome. New England Journal of Medicine 2015;373(13):1241–1249. doi: 10.1056/NEJMra1411863
[65] Nielsen M, Barnes CB & Ulrik CS. Clinical characteristics of the asthma-COPD overlap syndrome — a systematic review. International Journal of Chronic Obstructive Pulmonary Disease 2015;10:1443–1454. doi: 10.2147/COPD.S85363
[66] Dummer JF, Epton MJ, Cowan JO et al. Predicting corticosteroid response in chronic obstructive pulmonary disease using exhaled nitric oxide. American Journal of Respiratory and Critical Care Medicine 2009;180(9):846–852. doi: 10.1164/rccm.200905-0685OC
[67] Leigh R, Pizzichini MM, Morris MM et al. Stable COPD: predicting benefit from high-dose inhaled corticosteroid treatment. European Respiratory Journal 2006;27(5):964–971. doi: 10.1183/09031936.06.00072105
[68] GOLD. Diagnosis of diseases of chronic airflow limitation: asthma, COPD and Asthma-COPD Overlap Syndrome (ACOS) 2014. Available at: http://ginasthma.org/asthma-copd-and-asthma-copd-overlap-syndrome-acos/ (accessed May 2016).
[69] Bleecker ER, Emmett A, Crater G et al. Lung function and symptom improvement with fluticasone propionate/salmeterol and ipratropium bromide/albuterol in COPD: response by beta-agonist reversibility. Pulmonary Pharmacology & Therapeutics 2008;21(4):682–688. doi: 10.1016/j.pupt.2008.04.003
[70] Lee JH, Lee YK, Kim EK et al. Responses to inhaled long-acting beta-agonist and corticosteroid according to COPD subtype. Respiratory Medicine 2010;104(4):542–549. doi: 10.1016/j.rmed.2009.10.024