dpa picture alliance archive / Alamy
Summary
Patients receiving dialysis will typically require additional medicines to mimic kidney functions. These include phosphate binders and erythropoiesis-stimulating agents.
As many medicines rely on the kidneys for clearance and excretion, it is important that any medicines a patient is taking are reviewed. The effects of dialysis are more significant for medicines used in the long term due to the risk of accumulation; medicines given as a single or loading dose very rarely require adjustment.
Patients who are treated with dialysis for chronic kidney disease have a higher risk of morbidity and mortality than the general population. Patients receiving dialysis are much more likely to suffer from cardiovascular events such as myocardial infarction.
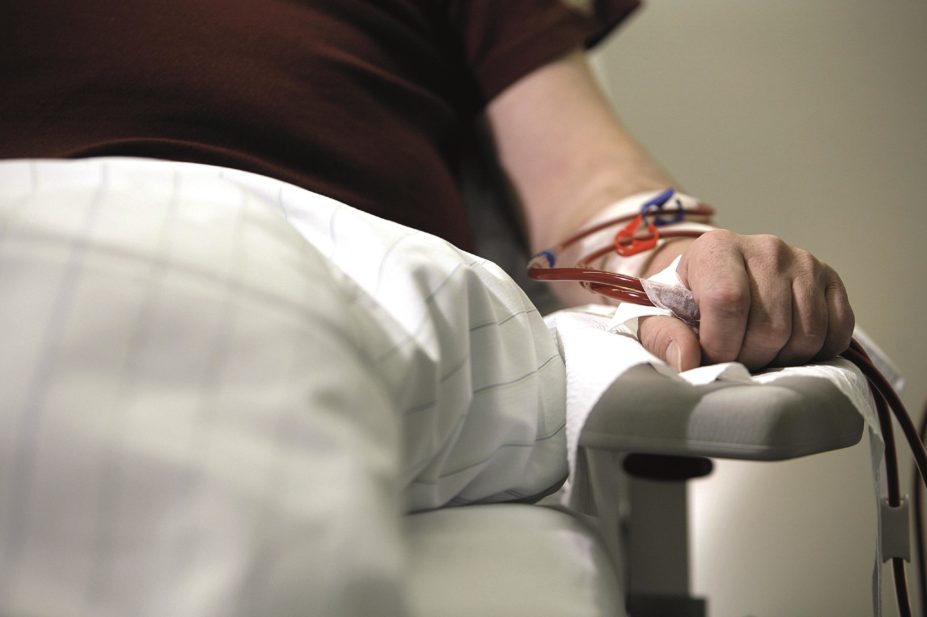
Patients with end-stage renal disease require renal replacement therapy, such as dialysis, to replace the function of the kidneys. However, dialysis cannot replicate all processes, and patients will require clinical management to provide a number of small functions.
This usually includes additional medicines which mimic other functions of the kidney.
For example, dialysis will only partially control blood pressure, the clearance of middle molecular range solutes (E.g. beta2 -microglobulin) and uraemic toxins, and the calcium and phosphate balance in the body[1]
. Functions that dialysis cannot perform include the synthesis of local hormones in the kidney, such as erythropoietin (EPO, a hormone required to produce new red blood cells, or erythropoiesis), the metabolism of filtered proteins and activation of vitamin D.
Therefore, dialysis patients are commonly prescribed a number of additional medicines such as antihypertensives, phosphate binders and erythropoiesis-stimulating agents (ESAs) and the active form of vitamin D.
Phosphate binders along with a restricted-phosphate diet are used to control the phosphate levels of patients receiving dialysis. Dietary phosphorus levels should be restricted to 5,500–7,000mg per week, of which around 60% is absorbed. Each session of haemodialysis (HD) will remove around 800mg of phosphorus; therefore, a standard three times a week HD regimen will remove on average more than half of the weekly intake of phosphate. The remainder will need to be removed and managed through the use of phosphate binders[2]
. Patients will usually start with a calcium-based binder (e.g. calcium carbonate or calcium acetate). The overall daily elemental calcium level should not exceed 1,500mg per day.
If this is not sufficient to control the phosphate levels, then other phosphate binders (e.g. sevelamer hydrochloride or lanthanum carbonate) can be used, either in addition to or as replacement for the calcium-based binder. Calcium-based binders are contraindicated if there is a raised serum calcium level.
Erythropoiesis-stimulating agents (e.g. epoetin alfa, epoetin beta and darbepoetin) may be used in patients receiving dialysis to increase EPO levels[3]
. The effect of these agents is measured by assessing a patient’s haemoglobin level, which should be checked at least monthly at the start of treatment, and once every three months when the patient is stable[4]
.
The patient’s haemoglobin level should be maintained between 10–12 g/dl, and the ESA dose adjusted if the level is below 10.5g/dl or greater than 11.5g/dl. If haemoglobin levels are high, the ESA dose should be adjusted rather than omitting doses of the ESA.4
To ensure ESA treatment works effectively, the patient’s iron stores will need to be sufficient to enable the production of new red blood cells. Therefore, serum ferritin levels should be measured. If the concentration is below 100mcg/l then iron replacement should be given instead of initiating ESA treatment.2,4
Dialysis patients can experience anaemia relating to their chronic kidney disease (CKD). Possible causes of anaemia include blood loss, deficiency of vitamin B12 or folic acid. Patients with CKD who have symptoms of anaemia (e.g. fatigue, pallor) or have a haemoglobin level of less than 11g/dl should have investigations to determine the cause.
HD patients often have lower levels of iron and require intravenous iron replacement. Oral iron therapy is not effective for patients receiving HD due to poor absorption relating to high urea levels. Smaller iron doses can be given at each session. For patients receiving peritoneal dialysis (PD), it is more practical for them to receive a higher dose much less frequently if intravenous iron therapy is required. Generally PD patients can be managed with oral iron therapy[5]
.
Ferritin levels should be checked every one to three months, and iron management should be assessed if the serum ferritin level is more than 500mcg/l to prevent iron overload from occurring (>800mcg/l).
Iron and ESA treatment decreases patient fatigue, and has also been found to reduce hospitalisation and the need for blood transfusions.(
[2],
[3],
5
)
Blood samples should be taken from the patient during a midweek HD session, with monitoring of biochemical and haematological parameters every month for HD delivered in hospitals and at least every three months for home HD patients.
Pre-dialysis blood pressure should be less than 140/90 and following dialysis this should be less than 130/80.
Pre-dialysis serum level targets in HD[6]
| Biochemistry/haematology measures | Serum concentration |
|---|---|
Bicarbonate | 18–24 mmol/l |
Potassium | 4.0–6.0 mmol/l |
Phosphate | Less than 1.8 mmol/l |
Calcium | 2.2–2.55 mmol/l |
Haemoglobin | 10–12g/dl |
Drug dosing in dialysis
Once a patient starts receiving any form of dialysis, it is important to consider how any medicines they are taking will be cleared, as many medicines are dependent on the kidneys for elimination (see ‘Factors affecting drug clearance via dialysis’).
The effects of dialysis are more significant for medicines used in the long term due to the risk of accumulation; medicines given as a single or loading dose very rarely require adjustment.
It is important to consider the mode of dialysis as certain drugs may be used in one mode but not another. For example, methotrexate is contraindicated in PD but can be used with caution in HD; however, it is dialysed in HD and therefore the dose should be given at least 12 hours before the next HD session to ensure effectiveness.
Drug parameters affecting clearance via dialysis[3],
[4]
| Drug parameters | Less likely to be removed via dialysis |
|---|---|
Molecular weight (Daltons) | >500 Da* |
Protein binding | >80% protein binding** |
Volume of distribution | >1l/kg |
Solubility | Lipid soluble |
Usual elimination process | Not normally cleared by the kidneys |
*A molecule with a low molecular weight is more likely to be removed; however, the chemistry of the molecule could reduce the clearance of the drug3 **The patient’s current albumin and urea status needs to be considered. | |
Factors affecting drug clearance via dialysis
| Haemodialysis | Peritoneal dialysis |
|---|---|
|
|
|
|
|
|
|
|
|
To be removed by dialysis, a drug needs to be small enough to be filtered by the dialyser and be concentrated in the plasma and unbound from proteins to be ‘free’ to be filtered; if the kidneys would normally filter the drug, the chances of dialysis doing the same is increased3. If the renal elimination of a medicine in a normal functioning kidney is less than 30% of its total clearance from the body, then removal via dialysis is not usually clinically relevant to the patient[7]
.
Dialysis only replicates glomerular filtration, which can lead to increased clearance of the medicines that are normally filtered out in the glomerulus if they would normally be reabsorbed at a later stage of kidney filtration (e.g. fluconazole is reabsorbed in the tubules of the nephron)7
.
Guidance on the most appropriate dose of a medicine in a patient receiving dialysis is available from a number of reference sources, such as the Renal Drug Handbook (available at www.renaldrugdatabase.com). It is essential that all medicines are reviewed, including over-the-counter and herbal medicines, prior to initiation of dialysis and that the patient informs the dialysis unit of any changes in medicines.
Reference sources are a guide only, and a patient’s response to treatment should be monitored closely (and dose adjusted accordingly). This is particularly important when dealing with medicines that have a narrow therapeutic index where serum drug level monitoring is required.
Medicines that should not be used in dialysis patients include metformin, fenofibrate, bendroflumethiazide and nitrofurantoin.
Continuous ambulatory peritoneal dialysis (CAPD) is equivalent to a creatinine clearance of 5–10ml/min, while HD is equivalent of a clearance of 150–200ml/min during dialysis sessions, but drops to the patient’s previous level between sessions. Due to the high clearance during haemodialysis, medicines may be administered post-dialysis to ensure that the drug remains at a therapeutic level in the body.
In practice, drug dosing should be based on the patient’s creatinine clearance being <10ml/min unless stated otherwise in the Renal Drug Handbook or the medicine’s summary of product characteristics[3]
. However, it is important to note that the patient’s own residual kidney function can also contribute to the elimination process[3]
. Patients receiving HD should take any drug likely to be dialysed after the session to ensure it has the desired therapeutic effect[3]
.
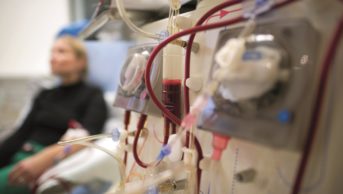
The number of different dialysers available makes it hard to predict the effect dialysis will have on drug clearance, as there can be significant variation in the amount of medicine removed. It is therefore important to know the dialyser type before making any dose decisions. The shape, surface area, material and membrane permeability are all important factors to consider.
Complications of haemodialysis
Patients who are treated with dialysis for chronic kidney disease have a higher risk of morbidity and mortality than the general population. Patients receiving dialysis are much more likely to suffer from cardiovascular events such as myocardial infarction[1]
, and risk of mortality is also increased in patients not receiving sufficient dialysis or with a poor nutritional profile[3]
.
Disequilibrium syndrome is a potential complication in patients starting HD. This occurs on account of rapid changes in the blood’s composition, as the body has become accustomed to the build-up of toxic substances. Symptoms can include headaches, nausea, vomiting and, rarely, confusion and convulsions. Dialysis sessions at the start of treatment are therefore usually shorter in duration and more frequent than in established therapy[3]
.
Hypotension is a common problem for dialysis patients with at least 25% of patients affected[2] ,[3]
. It can occur if fluid is removed too quickly during dialysis. This is most likely to happen in patients who struggle to maintain a low fluid intake between dialysis sessions[2] ,[3]
.
Acute management focuses on restoring circulatory blood volume through the administration of sodium chloride 0.9% bolus injection (100ml) and raising the patient’s feet.
If hypotension occurs, various steps can be taken to minimise the risk at future sessions. These include switching to daily HD; using a bicarbonate-based dialysate or sodium modelling (mapping changes to the tonicity of the dialysate as the session progresses); or cooling the dialysate to 35.5–36.0o C, which will cause a higher release of catecholamines and thus raise the blood pressure through vasoconstriction. Patients taking antihypertensive medicines can also omit their dose on dialysis days.
The rate of clearance can also be reduced during future sessions; however, this has not been proven to be of benefit and could lead to insufficient dialysis if the session time is not lengthened to compensate.
If low pressure remains an issue, then the use of fludrocortisone or midodrine can be considered. Fludrocortisone as a mineralocorticoid will increase the reabsorption of sodium into the body, and therefore increase fluid into the intravascular space. It is commonly used to treat orthostatic hypotension and has been used with limited evidence of benefit in HD patients.
Midodrine is an alpha agonist that increases vascular tone. It can be taken orally, starting at 2.5mg and titrated if necessary up to 10mg. It is usually given 30–60 minutes prior to HD therapy[2] ,[3]
.
Cramps occur in 5–20% of patients undergoing dialysis, mainly in the legs, again related to lowering the blood pressure or too much fluid being removed. Cramps can also be caused by low sodium levels, which may be depleted by the end of the session, hypomagnesaemia or carnitine deficiency.
The choice of treatment depends on the cause. A bolus injection of fluids may be sufficient to ease cramps caused by low sodium levels, or the patient can be prescribed 200–300mg quinine sulphate one to two hours prior to dialysis. Patients experiencing cramp can also be given carnitine and vitamin E supplements[2] ,[3]
.
Nausea and vomiting can also occur in patients due to the hypotensive effect of dialysis. This can be treated symptomatically with an antiemetic[3]
.
Dialysis-associated steal syndrome can occur in patients with fistulas due to a reduced perfusion of blood to the hand. The common symptoms associated with the syndrome are numbness and pain in the hand, coldness in the hand and a faint pulse[2]
. In severe cases, urgent surgical intervention may be required to prevent loss of digits and function of the hand[2]
.
Pruritus affects half the patients on dialysis (both HD and PD) and can occur for a number of reasons related to kidney disease and dialysis. Possible causes include uraemic toxins within the blood; a high phosphate level; adverse reactions to heparin; or a reaction to the semi-permeable membrane[3]
. Treatment involves identifying the cause and taking corrective steps, for example optimising dialysis therapy to reduce toxins in the blood; use of an alternative anticoagulant; or altering the membrane within the dialysis system.
Thrombosis relating to the patient’s catheter (sometimes fistula) is more likely in patients who have a higher haemoglobin level. Thrombolytics such as alteplase or urokinase can be used if the catheter becomes thrombosed.
Patients with raised haemoglobin levels receiving epoetin or iron should have their treatment reviewed and possibly stopped[3]
. However, this needs to be balanced against the potential risk of anaemia caused by blood loss during and after dialysis, for which erythropoietin and iron replacement therapy is required.
Arrythmias can occur in patients receiving HD on account of changes in the patient’s fluid, electrolyte and acid-base balance, as well as co-morbidities such as left ventricular hypertrophy. This is important for potassium, which is removed by dialysis. If serum potassium levels fall too quickly or by too much, then arrhythmias are more likely to occur. This is of particular concern if the patient is taking digoxin, where a lowering of potassium to less than 3.5mmol/l could lead to an increased sensitisation of the cardiac cells, resulting in arrhythmia.
If potassium levels after dialysis have been lowered more than expected, the dialysate used in the next session should be altered to have a higher potassium content, so the concentration gradient is reduced and less potassium is lost from the blood[2] ,[3]
.
C
ardiac arrest can occur during haemodialysis, which can result in sudden death. It is more likely to occur in older patients, patients with diabetes, or patients who use a catheter rather than a fistula. There are seven cardiac events per 100,000 HD sessions[2]
.
Peritoneal dialysis complications
Peritonitis is a serious and significant complication of peritoneal dialysis (PD), which can result in death[3]
. Symptoms of peritonitis include abdominal pain, tenderness, nausea, vomiting, diarrhoea, chills and fever. Peritonitis accounts for up to 35% of all hospital admissions in patients receiving PD, and is the main cause of the patient losing their catheter and changing to haemodialysis. Peritonitis also causes damage to the membrane and reduces the efficiency of dialysis; the permeability of the peritoneal membrane can be increased by changing the dialysate solution.
Gram-positive organisms (particularly Staphylococcus aureus and S. epidermidis) account for 75% of cases of peritonitis in patients receiving PD. However, the risk of death is higher in cases caused by a gram-negative organism[2] ,[3]
.
The risk of peritonitis can be reduced with good aseptic, non-touch techniques when changing the dialysate, and regular inspections of the dialysate after emptying.
Patients are advised to immediately contact their dialysis unit if a bag is cloudy[2]
. Although a cloudy bag is a good indicator of an infection, it is not always present and there could be other reasons (e.g. caused by the patient’s abdomen being dry during the day)[2]
. If the bag of dialysate is cloudy, heparin can be added to dissolve the build-up of fibrin in the peritoneum to prevent the catheter becoming blocked.
Patients with suspected peritonitis should be started on broad spectrum antibiotics that cover both gram-positive and gram-negative organisms
[3],
5
. Common first-line treatments include vancomycin or cephalosporins for gram-positive organisms, and aminoglycosides or third-generation cephalosporins for gram-negative organisms5
.
These should be administered via the intraperitoneal route, usually once daily for at least six hours, and continued for at least two to three weeks depending on the patient’s response and causative organism[3],
5
. Penicillin-based antibiotics and aminoglycosides are chemically not compatible and cannot be mixed together; however, cephalosporins and aminoglycosides are compatible[3],
5
.
Patients with peritonitis that do not respond to treatment within five days are classed as having refractory peritonitis, and their catheters may need to be removed[3],5
.
Repeated or extensive use of antibiotics increases the risk of a fungal infection causing peritonitis. Fungal peritonitis is associated with a high mortality rate, causing death in 25% of affected patients. To ensure prompt initiation of antifungal therapy, a gram stain is taken as part of the initial assessment and can be used to identify yeast growth.
Patients with fungal peritonitis should have the catheter removed immediately (keeping it in place is associated with an increased risk of mortality), and antifungal treatment should be started and continued for up to four weeks after catheter removal
[2],
[3]
.
Depending on local protocols, initial therapy could be combined or single therapy. A number of antifungals can be used, including amphotericin, fluconazole, flucytosine, voriconazole, caspofungin or anidulafungin.
The route of administration is dependent on the antifungal chosen; initial treatment will be through intravenous or intraperitoneal administration. For example, fluconazole can be used via the intraperitoneal route at a dose of 200mg every 24–48 hours, while amphotericin should not be given via the intraperitoneal route on account of the risk of causing chemical peritonitis and pain to the patient.
Exit site infections predominantly occur because of poor aseptic technique, and may lead to peritonitis. The diagnosis is based upon a visual inspection of the exit site, with marked erythema or discharge suggestive of an infection[2]
.
Exit site infections are usually treated with antibiotics for at least two weeks. The most common cause of exit site infections is S. aureus; therefore, an antibiotic effective against S. aureus is required. Flucloxacillin is commonly used for S. aureus infections and can be given orally, at a dose of 500mg four times a day, if no systemic symptoms are present. Otherwise, the patient may need admission to receive intravenous antibiotics. If the culture shows a gram-negative organism, such as a Pseudomonas infection, then oral ciprofloxacin 500mg twice a day could be used if appropriate[3]
.
Hernias may occur in patients receiving PD due to the pressure of the dialysate fluid held in the abdominal cavity. Large hernias can affect the flow of solutes[3]
, and may require patients to be moved to HD while the hernia is surgically repaired3 5.
Fluid leakage in PD can result from poor placement of the catheter, damage to the abdomen or the patient’s anatomy. If a leak does occur, the underlying cause must be identified and treated; during this stage PD should be withheld for as long as possible, or continuous ambulatory PD can be used2 5.
Alan Green is an academic pharmacist practitioner at the University of Sunderland and Gateshead NHS Foundation Trust.
References
[1] Field M, Pollock C & Harris D. The Renal System: Basic science and clinical conditions, 2nd Edition. London: Churchill Livingstone Elsevier 2010.
[2] Floege J, Johnson RJ & Feehally J. Comprehensive Clinical Nephrology, 4th edition. London: Elsevier Saunders 2010.
[3] Ashley C & Morlidge C. Introduction to Renal Therapeutics. London: Pharmaceutical Press 2008.
[4] Renal Association. Anaemia in CKD Guideline 2010.
[5] Leung R. What factors need to be considered when dosing patients on renal replacement therapies? January 2013. UK Medicines Information.
[6] The Renal Association. Haemodialysis Guideline. 2013.
[7] Gilbert SJ & Weiner DE. National Kidney Foundation’s Primer on kidney diseases, 6th Edition. London: Elsevier Saunders 2014.
You might also be interested in…
Dialysis: principles and treatment options
Managing ANCA-associated vasculitis