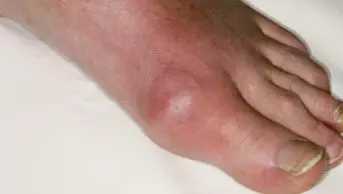
Mr Jones, a 56-year old man with a history of myocardial infarction, and taking aspirin, metoprolol, lisinopril and simvastatin presents with an acute episode of gout. Acute gout is usually treated with a non-steroidal anti-inflammatory drug or colchicine but what would be best for this patient?
The use of NSAIDs in patients with ischaemic heart disease has been the source of much debate in recent years. Non-selective NSAIDs can antagonise the antiplatelet effects of aspirin and reduce its cardioprotective effects, and some NSAIDs (particularly coxibs) are also associated with an increased thrombotic risk, particularly when used at high doses and for long-term treatment. The Commission on Human Medicines in the UK advises that the combination should only be if absolutely necessary. In addition, the European Society of Cardiology guidelines advise that NSAIDs and coxibs should not be given to patients resistant to antiplatelet treatment.
They also advise avoiding NSAIDs and the immediate period myocardial infarction. Therefore, although the duration of use of the NSAID is likely to be short, an NSAID is not likely to be the first choice in this particular patient, and other options should be considered.
If an NSAID must be given, it should be considered that the combined use of NSAIDs (including coxibs) and aspirin (even in antiplatelet doses) increases the risk of gastrointestinal bleeds. If the patient has any other risk factors for gastrointestinal bleeding, such as a past ulcer, it would be prudent to consider giving him some form of gastroprotection, such as a proton pump inhibitor.
Given the patient’s history, colchicine may be a more suitable option.
However, this drug also interacts with his other medicines. Mr Jones is taking simvastatin and cases of myopathy and rhabdomyolysis have been reported in patients taking colchicine with a statin. Colchicine alone can, rarely, cause myopathy, and because the statins can also cause myopathy, an additive or synergistic effect may occur. An adverse interaction seems to be more common in those given colchicine long-term or at high-dose, or in those with renal impairment. Although this interaction is rare it is serious.
Patients taking statins should always be made aware of the risks of myopathy and rhabdomyolysis, and asked to promptly report muscle pain, tenderness or weakness, especially if this is accompanied by malaise, fever or dark urine. This advice should be reinforced if a patient is also given colchicine. However, despite this possible interaction, colchicine would appear to be a better choice than an NSAID for this patient.
Blackan/Dreamstime.com
Long-term maintenance The longer term management of gout often includes the prophylactic use of allopurinol. Mr Jones is also taking an angiotensin-converting enzyme inhibitor and the concurrent use of allopurinol can lead to severe adverse effects. There have been a small number of cases of Stevens-Johnson syndrome (including fatalities) and cases of hypersensitivity attributed to the use of captopril with allopurinol.
he combination of ACE inhibitors and allopurinol can also increase the risk of blood dyscrasias (eg, leucopenia, neutropenia), resulting in serious infection, and patients with renal impairment are at greater risk. However, these adverse effects are rare and unpredictable, and do not preclude the use of both medicines. If this patient is given this combination long-term, he should be closely monitored for signs of adverse effects, with frequent checks of white blood cell counts, particularly in the first three months of treatment. He should be advised to report any signs or symptoms of hypersensitivity (eg, skin reactions) or low white cell count (such
as any unexplained sore throat or fever).
The treatment of gout in patients with other co-morbidities is often problematic but, with monitoring and good patient counselling, can be undertaken safely.