CDC / SCIENCE PHOTO LIBRARY
Invasive fungal infections (IFI) that cause life-threatening diseases, such as invasive aspergillosis, cryptococcosis and
Candida infections, carry a high mortality rate despite optimal treatment[1]
. Globally, fungal-associated mortality is comparable to tuberculosis and is more than three times that of malaria, with an estimated 1.6 million deaths annually[2]
.
Selective pressure on these pathogens through indiscriminate antifungal usage has isolated resistant fungal pathogens to one or more of the main antifungal classes. Widespread agricultural antifungal exposure and the global movement of produce has accelerated the spread of antifungal resistance (AFR).
Antifungal stewardship (AFS) is an ethic that incorporates responsible usage and conservation of antifungals and its aims overlap with antimicrobial stewardship (AMS) by focusing upon optimising patient outcomes, while minimising healthcare costs and preserving the future effectiveness of the limited treatment options. AFS poses unique challenges to improving antifungal usage; patients presenting with IFI have greater comorbidities and high case-mortality rates, and the treatments are associated with higher costs and greater risk of toxicity.
With AFR becoming increasingly recognised as a major concern for healthcare, this article aims to highlight the current trends in AFR, review the treatment options available and the importance of diagnostic capabilities. It will also put this in the context of pharmacy and how pharmacy teams can support AFS through selecting appropriate antifungal therapies and performing ongoing reviews of these therapies.
Availability of antifungal therapies
Unlike bacteria, fungi are eukaryotes and similar to their host human cells and, therefore, have fewer unique cell targets for antifungal therapeutics[3]
. Antifungal therapies for IFIs are limited to five distinct classes:
- Polyenes (e.g. amphotericin B)
—
the polyenes bind to ergosterol (the major sterol component of fungal membranes) destabilising the permeability of the cell membrane and resulting in cell death. Toxicity of human cells may cause tubular damage and kidney dysfunction, which is negated by using liposome formulations[4]
. - Pyrimidine analogues (e.g. flucytosine) — pyramidine analogues, such as flucytosine, inhibit pyrimidine metabolism and DNA synthesis and are the mainstay of Cryptococcus meningitis treatment in combination with amphotericin. A narrow therapeutic window necessitates therapeutic monitoring to reduce risk of bone marrow suppression[5]
. - Echinocandins (e.g. anidulafungin, caspofungin and micafungin)
—
the main mechanism of action of echinocandins is to inhibit (2-4)-beta-D-glucan synthase, disrupting cell wall biosynthesis. Echinocandins are only active against Aspergillus spp. and Candida spp.; however, anidulafungin and micafungin are not used for the treatment of aspergillosis[6]
. - Azoles (e.g. fluconazole, isavuconazole, itraconazole, posaconazole and voriconazole)
—
the azoles block ergosterol biosynthesis through inhibition of lanosterol 14-alpha-demethylase. Fluconazole has high oral availability and tolerability, most likely owing to its high oral availability and tolerability, however, emergent azole-resistant Candida is making empiric therapy less effective. - Allylamines (e.g. terbinafine)
—
a newer class of antifungal drugs, allylamines inhibit ergosterol synthesis and are currently limited to treating superficial dermatophyte infections only.
Despite increasing AFR and the growing burden of IFIs, the development pipeline for new antifungals is limited (see Figure 1)[7]
. Amphotericin B still remains the most potent fungicidal therapy (in vitro) with the broadest antifungal activity, often used to treat a wide range of systemic infections. Owing to its toxicity and related side effects, it is often reserved for severe infections in critically ill or immunocompromised patients. Newly licensed triazoles (e.g. isavuconazole and posaconazole) provide some more robust fungicidal activity against Aspergillus, including mucorales coverage, however isavuconazole is not licensed for empiric Candida coverage owing to increasing prevalence of non-Candida (non-C.) albicans azole-resistant infections.
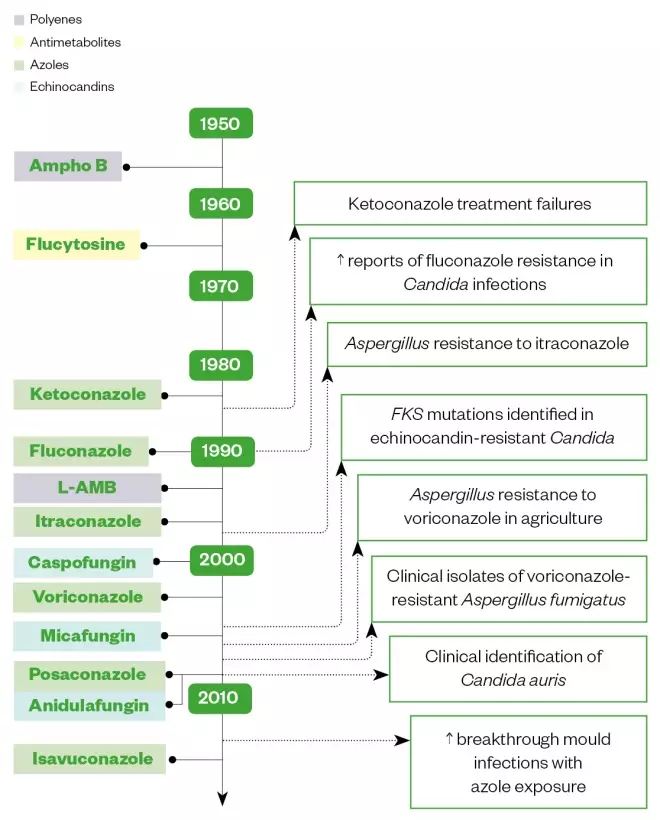
Figure 1: Antifungal drug development and emergence of antifungal resistance isolates
Source: Reproduced with permission from https://link.springer.com/article/10.1007/s12281-018-0307-z
Ampho B: Amphotericin B; L-AMB: Liposomal amphotericin B.
Timeline by decade of antifungal drug development and the correlation with emergence of antifungal resistance isolates
Impact of antifungal resistance
Candida species form part of the natural mucosal microbiome, but through opportunistic infection, can cause life-threatening disease with mortality around 40%, despite optimum therapy[8]
. Candidaemia incidence in the UK is increasing with non-C. albicans species, particularly C. glabrata becoming more frequently associated with infections contracted while under medical care. UK surveillance data indicates that 7% of Candida bloodstream infections tested are resistant to fluconazole, with 70% of these being C. glabrata or C. krusei species[9]
. Echinocandins are recommended as first-line therapy for candidaemia owing to increased azole resistance, however, 3% of C. glabrata isolates are now resistant to echinocandins (see Table)[10]
.
| Antifungal agent | 2015 | 2016 | 2017 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| S (%) | I (%) | R (%) | S (%) | I (%) | R (%) | S (%) | I (%) | R (%) | |
| Amphotericin B | 96 | 1 | 3 | 99 | <1 | <1 | 98 | 0 | 2 |
| Caspofungin | 98 | <1 | 1 | 94 | 3 | 3 | 94 | 3 | 3 |
| Fluconazole | 61 | 21 | 18 | 51 | 28 | 20 | 61 | 28 | 12 |
| Flucytosine | 99 | <1 | 0 | 95 | 0 | 5 | 95 | <1 | 5 |
| Voriconazole | 83 | 4 | 13 | 76 | 7 | 17 | 72 | 15 | 13 |
S: susceptible; I: intermediate; R: resistant Source: Reproduced with permission from Public Health England | |||||||||
C. auris, a multidrug-resistant yeast first isolated in Japan in 2009, has spread globally, resulting in dozens of hospital outbreaks[11]
. It poses a public health concern owing to the ease at which it spreads in the healthcare setting and the difficulty in isolate identification using standard laboratory methods. All isolates from the UK to date have been found to be resistant to fluconazole and often cross-resistant to other azoles, with variable resistance to amphotericin B (around 20%) and echinocandins (around 10%)[12]
.
Continuous lung exposure to Aspergillus mould can result in opportunistic invasive infections in at-risk patient groups, such as those with impaired immune function, solid organ transplant recipients and patients on immunosuppressive therapies[2]
. Azole-resistant Aspergillus may develop in patients receiving long-term azole therapy for chronic aspergillosis, however, the greatest burden of azole resistant A. fumigatus originates from spore-producing species secondary to environmental fungicide use. Azoles are increasingly failing as front line therapies, with associated patient mortality approaching 100%[13]
. Variations in amphotericin B susceptibility may also be seen with other Aspergillus species; A. flavus and A. terreus can tolerate higher concentrations of amphotericin B compared with A. fumigatus, resulting in reduced fungicidal activity[14]
. Aspergillosis, its causes, types and treatment is covered in more detail in the accompanying learning article.
Antifungal stewardship in practice
While there is overall acceptance of AMS programmes and their initiatives are well documented, AFS is less established and is not universally practiced, despite growing concerns about AFR. Therefore, having an AFS team comprising of microbiology/infectious disease clinicians, as well as an antimicrobial pharmacist is vital in the implementation of AFS in the healthcare setting (see Box 1).
Box 1: The role of the antifungal stewardship team
The role of the antifungal stewardship (AFS) team is to ensure that antifungal usage is restricted to patients that require treatment for invasive fungal infections and should include:
- Prescription review for discontinuing unrequired therapy;
- De-escalating where appropriate;
- Prompting intravenous to oral switch when suitable;
- Optimising drug therapy;
- Monitoring, which should comprise:
- Ensuring appropriate dosing;
- Pharmacokinetic/pharmacodynamic interaction management;
- Therapeutic drug monitoring (TDM);
- Dose adjustments in hepatic and renal dysfunction;
- Allergy status;
- Optimising non-drug treatment, such as source control;
- Restoring the immune response.
This team plays an essential role in educating healthcare professionals on AFS, local and national resistance patterns and awareness of new invasive fungal infections and appropriate treatments. They play an important role in developing and implementing evidence-based antifungal guidance and in encouraging appropriate prescribing. Guidance will need to be updated regularly in response to local and national resistance patterns[15],[16]
.
The team should monitor antifungal usage, to determine prescribing trends, including whether it is in line with local resistance patterns. Expenditure reporting will also be a key aspect in ensuring that appropriate regimens are used where indicated and fed back to local teams[16]
.
How pharmacists can implement patient-level strategies to improve AFS
Presentation with symptoms indicative of acute IFI necessitates early diagnosis and prompt initiation of antifungals where indicated. Delays in initiating effective therapy result in unacceptable patient morbidity and mortality. Initiation of therapy must be timely, follow local evidence-based antifungal prescribing guidelines and be tailored to individual patient needs.
Assess the need for initiation of antifungal therapy
IFIs can be difficult to recognise, so treatment may often be delayed or not given. Colonisation with yeast is common in hospitalised patients and does not necessarily represent invasive infection. Patients with a suspected IFI should undergo a physical examination and blood cultures, which are insensitive markers of IFIs but are still relied upon for diagnosis, as well as microbiology tests. Microbiological confirmation of fungal pathogen in a sterile site should trigger immediate antifungal treatment[6]
.
In the absence of confirmed microbiology for IFIs, patient assessment for risk factors for candidiasis, which include broad-spectrum antimicrobial usage, central intravascular devices, gastrointestinal tract perforations or surgical intervention, or mould infection, which include an immunocompetent host with radiological or clinical evidence of infection, is required to assess risk versus benefit of empiric antifungal usage. Biomarkers and polymerase chain reaction detection may be used to exclude infection, where available (see Box 2)[6]
.
Box 2: Biomarker and polymerase chain reaction techniques in diagnosis
Modern techniques for diagnosing invasive fungal infections include biomarker testing to provide evidence of a fungal infection, as well as molecular tools, such as polymerase chain reaction to identify the fungi present. Biomarker tests available include detection of Aspergillus galactomannan, cryptococcal antigen, and pan-fungal 1,3-beta-D-glucan. An array of species-specific and pan-fungal polymerase chain reaction diagnostic tests are also available[17]
. These tests are used for screening, diagnosis and prognosis with varying degrees of sensitivity and specificity.
For the diagnosis of aspergillosis, imaging, such as a chest X-ray or CT scan for the regions of the body where infection is suspected, may be required. In cases where a respiratory source is suspected, further samples such as sputum or a bronchoalveolar lavage should be collected and, in some cases, tissue biopsies may need to be taken for microscopy.
Lack of access to and reduced sensitivity from diagnostic tools, as well as delayed results, culminate in uncertainty of diagnosis in many complex patients and introduction of empiric antifungals treatment where diagnosis remains unclear[18]
. An essential part of AFS programmes is to improve the availability of diagnostic tests to enable earlier diagnosis and earlier targeted treatment in high-risk patients.
Select appropriate antifungal therapies
Patients at risk of IFIs belong to special populations (e.g. neonates, paediatric patients, those in intensive care, and frail, older patients) who show substantial pathology-mediated pharmacokinetic variations[17]
, where detailed pharmacokinetic knowledge of antifungal drugs is essential to optimise outcomes while minimising treatment complications. Polypharmacy also complicates treatment options with drug interactions common with antifungal therapies[19]
.
Pharmacists should have a detailed knowledge of a drug’s pharmacokinetic properties, its activity against targeted organisms, relevant tissue penetration, and local resistance rates. National and international guidelines provide evidence-based practice for the management of IFI including Candida, Aspergillus, other moulds, histoplasmosis, Cryptococcus and P. jiroveci pneumonia (PJP).
Echinocandins remain the treatment of choice for candidaemia but poor penetration to the eye, central nervous system and urine may require alternative drug choice once the source of IFI is identified[6]
.
Voriconazole is a the first-line therapy for Aspergillus infections, however, drug–drug interactions and adverse effects may preclude its use first line in some patients[16]
. In IFIs, where the causative pathogen is unclear, amphotericin B remains the drug of choice owing to its robust Candida and mould activity.
Perform an ongoing review of antifungal therapy
Antimicrobial therapy review is advised daily and by 48–72 hours to assess antimicrobial effectiveness[19]
. In AFS, the slow time to culture, as well as the host’s expected slower disease response to antifungals would suggest that a 48–72 hour review may be too early to measure treatment response.
Drug interactions, which can influence drug levels and risk of toxicity, are important considerations to optimise antifungal use. Potent interactions may preclude first-line antifungal therapies (e.g. anti-mould azoles may be problematic in patients on concurrent chemotherapy or anti-retrovirals owing to unpredictable drug interactions)[19],[20]
.
Therapeutic drug monitoring (TDM) is performed for antifungal drugs exhibiting a narrow therapeutic range; large inter-individual variation in pharmacokinetic can cause sub-optimal treatment response or severe adverse effects. This approach is most relevant for the mould-active triazoles (i.e. itraconazole, voriconazole and posaconazole) and flucytosine[21],[22]
. TDM of antimould azole should be taken after three to five days to guide ongoing dosing schedule and optimise treatment. Concomitant use of proton pump inhibitors, food intake and gastric pH can all influence oral azole absorption.
Kidney and hepatic dysfunction may impact antifungal metabolism and clearance. Dose adjustments for fluconazole and flucytosine are advised for patients with kidney failure, while the voriconazole vehicle, sulfobutylether-beta-cyclodextrin sodium (SBECD), may accumulate if prolonged intravenous treatment is administered. Amphotericin B remains one of the most nephrotoxic therapies, even when administered as a liposomal form with kidney tubular acidosis with associated electrolyte disturbance common after prolonged treatment. Azoles and echinocandins have been associated with hepatotoxicity and dose adjustment of caspofungin is advised if hepatic function is present (Child–Pugh C score)[23]
.
De-escalation of broad-spectrum antifungals to targeted therapies is advised once sensitivities are recognised. This can reduce antifungal selective pressures of broad-spectrum therapies and can facilitate an intravenous-to-oral (IV-PO) switch, reducing the cost of treatment and improving patient tolerability. De-escalation of empiric treatment in candidaemia is advised after five to seven days, when clinical response and sensitivities are both expected[6]
. Microbiology results and patients’ clinical response should be used to inform this review.
National and organisational strategies to improve antifungal stewardship
National surveillance of fungal pathogens and an overview of nation antifungal resistance is coordinated by Public Health England. Antifungal usage, as well as both primary and secondary care is reported under the umbrella of the annual English Surveillance Programme for Antimicrobial Utilisation and Resistance[10]
.
In the UK, AFS programmes are underused within acute trusts[24]
, with a lack of resources being a major obstacle to starting local programmes. As the majority of antifungal spend is commissioned centrally by NHS England, acute trusts may lack the financial incentives to reduce antifungal usage and invest in new biomarker and/or molecular diagnostic tests.
AMS-related incentives, such as the Commissioning for Quality and Innovation scheme, have primed antimicrobial activities and quality improvement projects across England with great success[25]
.
A coordinated effort is required from all prescribers across an organisation. Engagement by healthcare leaders, microbiology/infectious disease specialists and specialist pharmacists with all clinical areas of healthcare is required for antifungal quality improvements (see Box 1). Initiatives should be structured to reduce suboptimal prescriptions, unnecessary antifungal use, unsuitable broad-spectrum antifungals (e.g. amphotericin B), and inappropriate dosage or duration[26]
.
Corrective feedback from real-time practice is associated with sustainable improvements in prescribing habits and should be used in combination with any AFS quality improvement initiative. Local surveillance and epidemiological monitoring is advised to identify trends in prescribing and resistance, with results influencing local developments as international guidance is incorporated into practice (see Box 1).
References
[1] Bongomin F, Gago S, Oladele RO & Denning DW. Global and Multi-National Prevalence of Fungal Diseases – Estimate Precision. J Fungi (Basel) 2017;3(4):57. doi: 10.3390/jof3040057
[2] Brown GD, Denning DW, Gow NAR et al. Hidden Killers: Human Fungal Infections. Science Translational Medicine 2012;4(165):165rv13. doi: 10.1126/scitranslmed.3004404
[3] Srinivasan A, Lopez-Ribot JL & Ramasubramanian AK. Overcoming antifungal resistance.Drug Discov Today Technol 2014;11:65–71. doi: 10.1016/j.ddtec.2014.02.005
[4] Fanos V & Cataldi L. Amphotericin B-induced nephrotoxicity: a review. J Chemother 2013;12(6):463–470. doi: 10.1179/joc.2000.12.6.463
[5] Vermes A, Guchelaar H-J & Dankert J. Flucytosine: a review of its pharmacology, clinical indications, pharmacokinetics, toxicity and drug interactions. J Antimicrob Chemother 2000;46:171–179. doi: 10.1093/jac/46.2.171
[6] Pappas PG, Kauffman CA, Andes DR et al. Clinical practice guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis 2016;62(4):e1–50. doi: 10.1093/cid/civ933
[7] Miller RA. A Case for antifungal stewardship. Current Fungal Infection Reports 2018;12(165):33–43. doi: 10.1007/s12281-018-0307-z
[8] Pfaller M, Neofytos D, Diekema D et al. Epidemiology and outcomes of candidemia in 3648 patients: data from the Prospective Antifungal Therapy (PATH Alliance®) registry, 2004–2008. Diagn Microbiol Infect Dis 2012;74(4):323–331. doi: 10.1016/j.diagmicrobio.2012.10.003
[9] Public Health England. Laboratory surveillance of candidaemia in England, Wales and Northern Ireland: 2017. 2018. Available at: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/742602/hpr3418_cnddm17.pdf (accessed July 2019)
[10] Public Health England. English Surveillance Programme for Antimicrobial Utilisation and Resistance (ESPAUR). 2017. Available at: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/656611/ESPAUR_report_2017.pdf (accessed July 2019)
[11] Chowdhary A, Sharma C & Meis JF. Candida auris: a rapidly emerging cause of hospital-acquired multidrug-resistant fungal infections globally. PLoS Pathog 2017;13(5):e1006290. doi: 10.1371/journal.ppat.1006290
[12] Public Health England. Guidance for the laboratory investigation, management and infection prevention and control for cases of Candida auris. 2017. Available at: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/637685/Updated_Candida_auris_Guidance_v2.pdf (accessed July 2019)
[13] Fisher MC, Hawkins NJ, Sanglard D & Gurr SJ. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 2018;360(6390):739–742. doi: 10.1126/science.aap7999
[14] Robbins N, Caplan T & Cowen LE. Molecular evolution of antifungal drug resistance. Annual Review of Microbiology 2017;71(1):753–775. doi: 10.1146/annurev-micro-030117-020345
[15] Whitney L, Al-Ghusein H, Glass S et al. Effectiveness of an antifungal stewardship programme at a London teaching hospital 2010–16. J Antimicrob Chemother 2018;74(1):234–241. doi: 10.1093/jac/dky389
[16] Agrawal S, Barnes R, Brüggemann RJ et al. The role of the multidisciplinary team in antifungal stewardship. J Antimicrob Chemother 2016;71:ii37–42. doi: 10.1093/jac/dkw395
[17] Patterson TF, Thompson GR, Denning DWet al. Practice guidelines for the diagnosis and management of aspergillosis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis 2016;63:e1–60. doi: 10.1093/cid/ciw326
[18] Micallef C, Ashiru-Oredope D, Hansraj S et al. An investigation of antifungal stewardship programmes in England. J Med Microbiol 2017;66:1581–1589. doi: 10.1099/jmm.0.000612
[19] Albengres E, Louet L & Tillement J. Systemic antifungal agents. Drug Saf 1998;18:83–97. doi: 10.2165/00002018-199818020-00001
[20] Baxter K (ed), Marshall A & Sharp J. Drug interactions that can occur with azole antifungals.Pharm J 2008;280:280. URI: 10006194
[21] Wheat LJ, Freifeld AG, Kleiman MB et al. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin Infect Dis 2007;45:807–825. doi: 10.1086/521259
[22] Ashbee HR, Barnes RA, Johnson EMet al. Therapeutic drug monitoring (TDM) of antifungal agents: guidelines from the British Society for Medical Mycology. J Antimicrob Chemother 2014;69: 1162–1176. doi: 10.1093/jac/dkt508
[23] eMC. Caspofungin 50 mg powder for concentrate for solution for infusion – Summary of Product Characteristics (SmPC) – (eMC). 2018. Available at: https://www.medicines.org.uk/emc/product/8955/smpc (accessed July 2019)
[24] Perfect JR, Dismukes WE, Dromer F et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis 2010;50:291–322. doi: 10.1086/649858
[25] Islam J, Ashiru-Oredope D, Budd Eet al. A national quality incentive scheme to reduce antibiotic overuse in hospitals: evaluation of perceptions and impact. J Antimicrob Chemother 2018;73:1708–1713. doi: 10.1093/jac/dky041
[26] British HIV Association. British HIV Association and British Infection Association guidelines for the treatment of opportunistic Infection in HIV-seropositive individuals 2011. 2011. Available at: https://www.bhiva.org/file/SwhaEzgXmAGOt/hiv_v12_is2_Iss2Press_Text.pdf (accessed July 2019)

