This content was published in 2009. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
For more recent content on this topic, please visit this article: Liver function tests: indication and interpretation
In short
Pharmacists are likely to come across patients whose liver function tests are abnormal. However, assessing LFTs alone will not offer pharmacists the full story.
Knowing the likely cause of abnormal LFTs can help determine whether the abnormalities are caused by a medicine, or whether any doses of medicines need to be changed to compensate.
Many pharmacists will, at some stage, have needed to interpret a patient’s liver function tests (LFTs) and decide whether any of his or her medicines required dose reduction or cessation.
Although drug-induced liver disease is relatively uncommon, medicines should always be considered as a possible cause of liver dysfunction (and pharmacists should be vigilant for such events and report all suspected reactions to the Medicines and Healthcare products Regulatory Agency using the yellow card scheme). Over 600 medicines have been associated with hepatotoxic reactions.
Liver function assessment
LFTs involve detecting the levels of several biochemical markers in the bloodstream. While advice on adjusting the doses of medicines for patients with renal disease is plentiful (eg, ‘The renal drug handbook’), there is no equivalent publication for patients with abnormal LFTs or hepatic dysfunction. Furthermore, LFTs cannot quantify liver function in the same way that creatinine clearance quantifies renal function. Instead, practitioners rely on information in summaries of product characteristics, the British National Formulary and, where available, in published guidelines for treatment of specific diseases.
Although abnormal LFTs can indicate a hepatobiliary problem that warrants further investigation, they should not be assessed in isolation. The presence of other signs and symptoms of liver disease (eg, ascites, varices) should also be considered.
The common biochemical markers measured in LFTs are explained below.
Bilirubin
A by-product of haem metabolism, bilirubin is conjugated by hepatocytes for excretion. When hepatocytes are damaged (due to acute hepatitis or cirrhosis), they are unable to perform this function sufficiently, resulting in raised levels of unconjugated bilirubin and, ultimately, jaundice.
Serum bilirubin levels can also be raised as a result of cholestasis — a condition where the movement of bile from the liver to the gallbladder is inhibited (this can be caused by intrahepatic factors, such as viral hepatitis or a bacterial abscess, or extrahepatic factors, such as gallstones). Haemolysis, as seen in autoimmune haemolytic anaemia, can also raise bilirubin levels.
Transaminase enzymes
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) enzymes are most commonly produced by liver cells. In response to acute hepatocellular damage (eg, acute hepatitis), the levels of these enzymes in the bloodstream can rise significantly. Transaminase levels are not usually affected for patients with cholestasis; however, gallstones lodged in the common bile duct can result in transaminase enzymes and biliary enzymes (alkaline phosphatase [ALP] and gamma glutaryltransferase [GGT]) all being raised — indicating hepatitis and cholestasis.
Transaminase levels may be normal (or only marginally raised) in patients with cirrhosis since there is little remaining healthy liver tissue to produce them.
Gamma glutaryltransferase
Produced by hepatocytes and bile ducts, GGT is an enzyme for which levels can be raised 10- to 20-fold in patients with cholestasis. Its levels can also be raised, but often less so, in patients with cirrhosis.
Alkaline phosphatase
ALP isoenzymes are found in hepatocytes, bile canaliculi epithelial cells, bone and intestine. Raised ALP levels are commonly seen in both intra- and extrahepatic cholestasis, and in Paget’s disease. If a patient’s GGT is also raised, this can help to confirm whether a raised ALP is of hepatobiliary origin.
Albumin
The level of serum carrier protein albumin is useful for assessing a liver’s synthetic function. Since albumin’s plasma half-life is approximately 20 days, it will take at least a week for levels to fall below the reference range as a result of liver damage or dysfunction. Consequently, albumin levels are a useful indicator of chronic liver dysfunction.
Other conditions can cause a more rapid reduction in albumin levels, such as the catabolic effects of severe infection.
Coagulation measures
Raised prothrombin time and international normalised ratio can indicate hepatobiliary disease in patients for two reasons. First, vitamin K absorption might be impaired by the absence of bile in the gut (eg, because of cholestasis). Second, the damaged hepatocytes (eg, in cirrhosis or acute hepatitis) will be unable to synthesise adequate clotting factors.
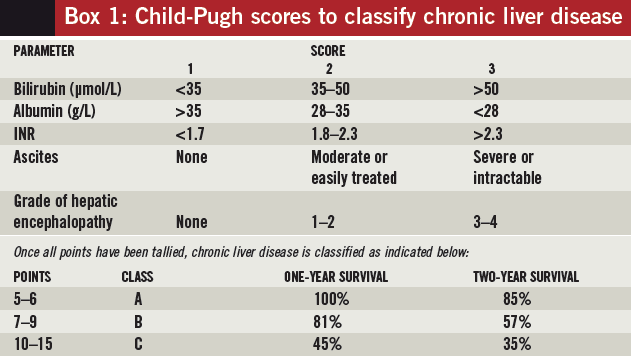
Child-Pugh score
If a patient has been diagnosed with chronic liver disease, the Child-Pugh score (see Box 1) can be used to assess the severity of his or her disease. However, its use in guiding drug dosing is limited because two patients with the same severity of chronic liver disease (as determined using Child-Pugh) could have markedly different metabolic capacities.
As a result, few drugs use this scoring system in their product information to suggest specific dose reductions — caspofungin is an exception which recommends a dose of 35mg daily (instead of the usual 50mg daily) for patients with Child-Pugh class B disease.
Effects of medicines on the liver
Medicines can affect the liver in many ways, ranging from minor abnormalities of reported LFTs to significant hepatic necrosis. In some cases, the reaction has been serious enough to warrant a medicine’s withdrawal from the market (eg, troglitazone — the first marketed thiazolidinedione).
Some reactions are predictable and occur in a dose-related manner (eg, in paracetamol overdose) whereas others are idiosyncratic (eg, cholestasis caused by chlorpromazine). For most drugs, pre-existing liver dysfunction does not generally increase the risk of developing drug-induced liver disease, although sodium valproate and methotrexate are notable exceptions.
Polypharmacy may increase the risk of hepatotoxic reactions. For example, nonsteroidal anti-inflammatory drugs are more likely to cause liver disease if they are prescribed at the same time as drugs that are hepatotoxic. Also, inducers of cytochrome P450 2E1 (eg, alcohol, rifampicin) raise the risk of hepatotoxicity caused by paracetamol and isoniazid.
Appropriate action
Since abnormal LFTs do not always indicate hepatic dysfunction and may not be drug-induced, changes to a patient’s drug therapy are not always needed. Nonetheless, if abnormal LFTs (with or without clinical jaundice) are observed, the following should be considered:
Are the abnormal values significant?
If enzyme levels are more than twice the upper limit of normal (ULN), this is generally considered to be significant. However, for some drugs larger increases may be considered acceptable (see examples, Box 2).

When were the patient’s medicines started?
Predictable hepatotoxic reactions usually have a latency period ranging from hours to weeks, whereas idiosyncratic reactions can take several months to occur.
Does the patient have a history of hepatotoxic drug reactions?
There is known crosssensitivity between some groups of medicines (eg, between phenothiazines, tricyclic antidepressants and NSAIDs).
Have any other tests been performed that offer alternative explanations for the abnormal LFTs?
When there is another explanation for deranged LFTs (eg, gallstones or pancreatic cancer), many medicines can be continued as normal. If medicines are stopped and the patient’s LFTs improve, the changes are likely to be drug-induced.
References
- North-Lewis P (editor). Drugs and the Liver. London: Pharmaceutical Press; 2008.
- Walker R, Whittlesea C. Clinical Pharmacy and Therapeutics. 4th edition. London: Churchill Livingstone; 2007.
- Joint tuberculosis committee of the British Thoracic Society. Chemotherapy and management of tuberculosis in the United Kingdom: recommendations 1998. Thorax 1998;53:536–48.
NOTE
Clinical Pharmacist PRACTICE TOOLS do not constitute formal practice guidance. Articles in the series have been commissioned from independent authors who have summarised useful clinical skills.
You might also be interested in…
How to support patients taking new oral anticoagulant medicines
