This content was published in 2008. We do not recommend that you make any clinical decisions based on this information without first ensuring you have checked the latest guidance.

In 2007, a National Patient Safety Agency alert (“Promoting safer use of injectable medicines”) focused attention on the safety and effectiveness of intravenous treatment.
So far, most discussions about implementing the alert have focused on injectable drugs, but IV fluids should not be overlooked.
Pharmacists review prescribed medicines critically, but their reviewing and monitoring of IV fluids is less consistent. However, this is an area where pharmaceutical expertise could, and should, be systematically applied.
IV fluids are often administered with little regard for actual fluid and electrolyte needs, however both under- and overdosing can cause serious adverse effects.
Research shows that junior doctors are responsible for most IV fluid prescribing, yet many are unaware of the sodium and potassium contents of common IV fluids. Recent advances in critical care have underlined the importance of tailoring fluid input to physiological needs and haemodynamic performance.
When it comes to prescribing of IV fluids, one size definitely does not fit all.
In this article, we have confined our discussion to IV treatment of adults, not children.
Fluid resuscitation
The situations where fluid resuscitation is required are discussed on p273.
It is generally agreed that large volumes of fluid are required in the short term. Isotonic crystalloid solutions (see p273) are often recommended in the first instance (unless a patient has had a significant haemorrhage and a haemoglobin level below 7g/dl, in which case a blood transfusion may be necessary).
However, 80–90 per cent of the infused volume of sodium chloride (NaCl) 0.9 per cent will eventually leave the intravascular compartment.
Panel 1 shows the amount of volume expansion of each compartment that can be expected from common fluids.
Crystalloid solutions rapidly leak into the interstitial space and can cause significant tissue oedema. This can be uncomfortable, but can also reduce mobility and compromise organ function. For example, there is evidence that crystalloid infusions cause small bowel oedema and this may impair gastrointestinal function by causing intolerance to enteral feeding and postoperative nausea and vomiting.
Also, increased cerebral oedema can worsen the prognosis for patients who have undergone brain surgery or head trauma.
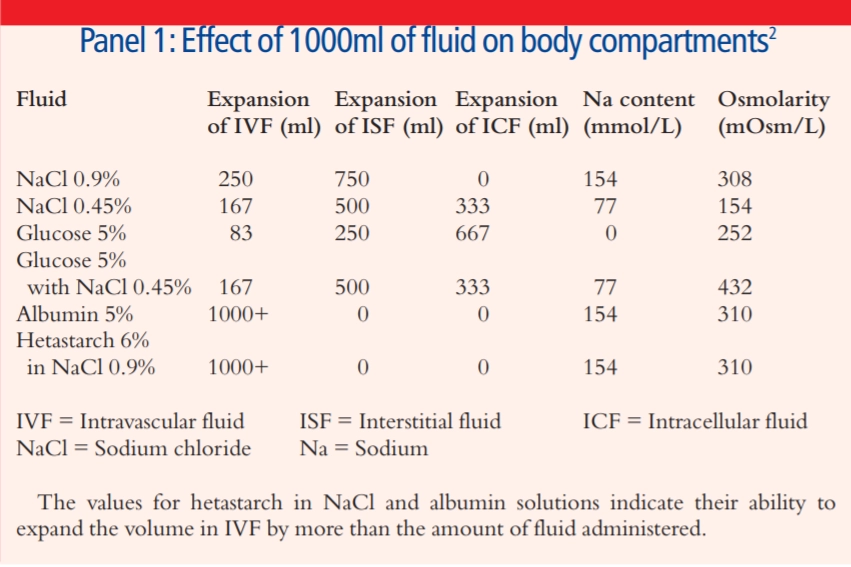
Colloid solutions (eg, starches, gelatin, albumin) are also effective for volume replacement. They are advantageous because they provide volume expansion without the risk of fluid overload and oedema.
In the past, the use of colloids has been limited by adverse effects and cost. Newer colloids have better side effect profiles, however studies comparing the two have not shown any differences in patient survival.
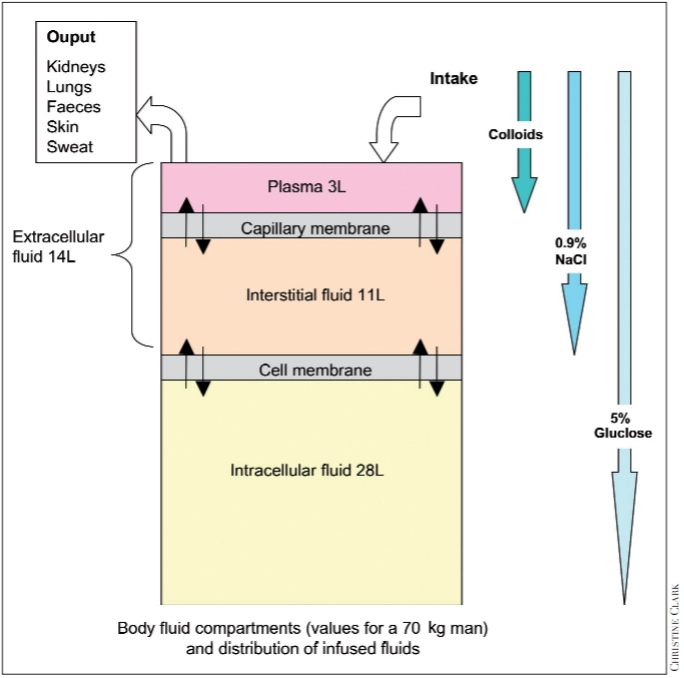
Figure 1 illustrates the different fluid compartments that exist in the human body, and how common IV fluids distributes within the body.
Fluid maintenance
Once the goals of fluid resuscitation have been achieved, the level of fluid administration should be adjusted for maintenance treatment.
Overloading with salt and water during resuscitation is often unavoidable and can take days or weeks to resolve, so it is important to switch as soon as possible to a regimen that matches physiological needs more closely, to avoid further overloading.
The basal requirements for young adults are approximately[3]:
- 1–1.4mmol/kg/day of sodium;
- 0.7–0.9mmol/kg/day of potassium;
- 30ml/kg/day of water.
A worked example of how these requirements can be met for a 60kg and an 80kg patient is shown in Panel 2.
These maintenance regimens are designed to provide typical amounts of sodium, potassium and water for a healthy young adult. Some caution is required because these requirements are based on body weight, therefore they tend to overestimate the requirements for obese and elderly patients.
Adjustments need to be made to correct pre-existing deficits and replace unusual losses (eg, from drains or fistulae — see below). In addition, pre-existing medical problems need to be taken into account.
Elderly patients are more likely to have cardiac, respiratory and renal impairment, and have a lower reserve capacity than younger patients.
Other at-risk groups include patients with:
- Cardiac disease — tissue perfusion maybe reduced and there is a risk of fluid retention and overload;
- Renal disease — it may be difficult to remove excess fluid, so there is a risk of fluid overload;
- Cerebrovascular disease — patients are likely to have complex requirements and it may be difficult to maintain electrolyte and fluid balance.

Replacing losses
Where patients are losing fluid through gastrointestinal (GI) drains or fistulae, maintenance regimens need to be adjusted accordingly to account for additional losses. Panel 3 (p281) lists the typical contents of the various GI fluids, and explains how these are used to calculate an additional requirement. Many replacement regimens involve administering 1L bags of NaCl 0.9 per cent with added potassium.
However, excessive administration of these bags provides large amounts of chloride which may contribute to metabolic acidosis (see p273). This may be aggravated if the fluid lost also contains large amounts of bicarbonate.
Response to injury
Normal fluid and electrolyte homeostasis isdisturbed after injury (eg, trauma, surgery) and this can affect the way in which infused electrolytes and fluids are handled[4].
The changes are complex and there is still much to learn. However, two important elements of the physiological response to surgery are now understood and need to be taken into account when planning or reviewing treatment.
Vascular leakage
Inflammation, resulting from sepsis, trauma or burns, causes a sharp increase in vascular permeability. Consequently, there is leakage of plasma proteins, electrolytes and water from the intravascular compartment into the interstitial space. Vascular permeability returns to normal over the next 12 hours, but affected patients may need large amounts of IV fluids to maintain adequate tissue perfusion.
Crystalloids are commonly used although high molecular weight colloid solutions can also be given, in smaller quantities, to restore intravascular volume.
Antidiuretic hormone
In the immediate postoperative period, there is intense activation of antidiuretic hormone and the renin-aldosterone-angiotensin system.
This results in active sodium retention and a low urine output. The capacity to excrete salt and water returns to normal over the next few days. If urine output is used to determine organ perfusion, it will be underestimated and excess fluids may be prescribed as a result.

Perioperative management
Two approaches commonly used during perioperative fluid management are goal-directed therapy and restricted fluid therapy.
Goal-directed therapy
The process of optimising a patient’s fluid status is known as goal-directed therapy. This approach involves adjustments of cardiac preload, afterload, and contractility, using vasodilators and inotropes, to balance oxygen delivery with oxygen demand[5].
Goal-directed protocols are intended to ensure the patient’s heart is producing its maximum stroke volume. This approach usually requires invasive monitoring techniques, such as central venous pressure monitoring.
Restricted fluid therapy
Restricted fluid therapy describes the process of replacing only the fluid that is lost and maintaining constant body weight, instead of following a standard recipe (usually 2–2.5L fluid) for all patients.
This approach requires careful monitoring of fluid input and output, and body weight.
Reviewing IV prescriptions
Pharmacists are required to review drug therapy for patients, but if this review is undertaken without access to all clinical information related to the patient, it is simply a prescription review. This only allows pharmacists to identify basic problems withthe prescription, and does not enable them to make a full contribution to patient care.
This principle also applies to IV prescriptions. A comprehensive treatment review is only undertaken with knowledge of a patient’s diagnosis and pathophysiological status.
IV prescriptions should be reviewed at the same time as all other prescribed medicines, including parenteral and non-parenteral medicines and fluids.
Reviewing one prescribed item or sectionof a prescription in isolation does not provide a clear picture of the patient’s treatment.
A treatment review is likely to identify more significant clinical problems than a simple prescription review and pharmacists should aim to review patients comprehensively whenever possible.
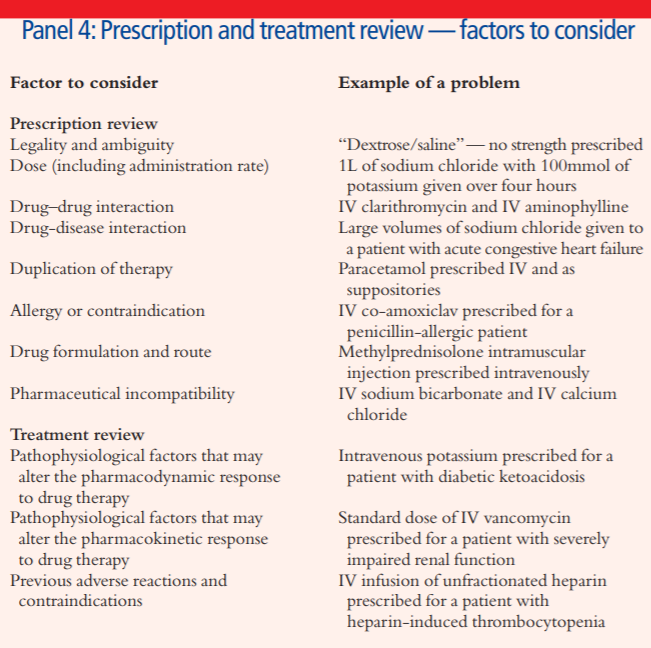
Panel 4 is a reminder of the aspects to consider when reviewing a prescription that includes IV medicines and examples of problems that can be identified.
It also is helpful for a pharmacist to consider:
- How the drugs are to be administered (ie, what type of pump will be used);
- Whether the drugs are to be administered through a central line or a peripheral line;
- Whether more than one drug is intended to be administered at the same time.
Some systems are complex and sound knowledge of different giving sets, connectors and taps is essential (further information on IV administration sets can be found in a bulletin from the Medical Devices Agency or an article by Quinn)[6,7].
Experienced pharmacists often recognise patterns in prescribing. Being familiar with common regimens helps when undertaking a prescription review.
Factors affecting blood pressure
Low blood pressure can be an early indicator that a patient does not have enough circulating volume (an indication for being prescribed more IV fluids). However, other factors that affect blood pressure may need to be considered.
Concurrent drugs
Some drugs that affect blood pressure (eg, diuretics, antihypertensives) can potentiate postoperative hypotension and influence IV fluid treatment.
Practitioners can accidentally prescribe more fluid than necessary for these patients, in the belief that hypovoleamia is the cause of their low blood pressure.
Analgesia
Low blood pressure can be caused by epidural analgesia. It is easy to assume for hypotensive patients who have recently received an epidural that their low blood pressure is drug-induced.
However, hypovoleamia cannot be excluded, especially for patients who have suffered large preoperative fluid losses.
Sodium content of medicines
When calculating the daily requirement of sodium for a patient, pharmacists should remember that several IV medicines deliver a considerable sodium load.
In addition, many IV medicines are added to infusion bags, often containing NaCl.
For example:
- Vancomycin 1g in 250ml NaCl 0.9 percent infusion, given twice a day —77mmol of sodium per day;
- Benzylpenicillin 1.2g in 100ml NaCl0.9 per cent infusion, given four times a day — 75mmol;
- Clarithromycin 500mg in 250ml NaCl0.9 per cent, given twice a day — 78mmol;
- Tazocin 4.5g in 50ml NaCl 0.9 percent, given three times a day — 51.3mmol;
- Metronidazole 500mg in 100ml NaCl0.9 per cent, given three times a day — 39mmol (Flagyl brand, other brands may differ).
Drug monographs for many IV medicines, which include the sodium content of the drug after reconstitution, can be accessed at www.injguide.nhs.uk.
Any enquiries regarding access to this website should be sent to jill.bullock@imperial.nhs.uk. Some case examples, highlighting some of the problems that can occur with patients receiving IV fluid therapy and the lessons that can be learnt from them, are described in Panel 5.
Panel 5: Lessons learnt through case examples
Lesson 1: Prescribe a full fluid regimen, not “bag-by-bag”
A 78-year-old male is admitted to a surgical admissions ward with acute abdominal pain.
The consultant instructs a junior doctor to start intravenous fluids.
After three days, the patient’s biochemistry results are reviewed — he has a plasma sodium level of 154mmol (normal range: 135–145mmol). On investigation, it is found that he has only received sodium chloride (NaCl) 0.9 per cent as his fluid maintenance regimen. The infusion had been commenced three days earlier, and the patient had received a new 1L bag every 8–10 hours thereafter.
Different doctors had repeated the prescription of the infusions, most likely at the request of the nurses managing the patient, who had replaced each bag with another bag of the same fluid. This case demonstrates the need to plan fluid replacement regimens and not prescribe it on a “bag by bag” basis.
This case was compounded by different doctors prescribing retrospectively and without referring to daily blood results. Such events can easily occur in a busy clinical area.
Lesson 2: Always use infusion pumps for continuous infusions
An 84-year-old female is admitted to hospital with pneumonia.
She is started on IV fluid maintenance treatment shortly after her admission because she is unable to eat or drink.
Three days later, she is found to be in acute heart failure and her fluid balance was 4L positive (her fluid output was 4L less than the amount of fluid she had received) since her admission.
Diuretic therapy was started and her heart failure resolved. The initial IV fluid regimen had been for 2L of glucose 4%/NaCl 0.9% with 20mmol/L of potassium every 24 hours, but the charts showed that a higher volume of fluid had been administered.
On investigation, it was found that all infusions had been administered without the aid of an infusion device. When administered in this way, some infusions are not delivered accurately. It appears that infusions intended for administration over 12 hours had been given over 8–10 hours, resulting in more fluid than intended being administered.
This case demonstrates the importance of using infusion pumps for all continuous infusions, including IV fluids. It is extremely difficult to infuse at accurate rates with gravity lines, and many end up being given at significantly different rates to those prescribed.
This is not critical for intermittent infusions, but can result in serious problems if used for longer periods when therapy is not adequately supervised.
Lesson 3: Do not interpret any individual biochemical results in isolation
An 83-year-old, 60kg male undergoes uneventful hemicolectomy for bowel cancer.
During the procedure, he loses 300ml of blood, but is given 500ml of a colloid solution and 2L of a crystalloid solution.
At the end of the operation, his blood pressure is 100/60mmHg and his haemoglobin is 7.2g/dl (normal: 11.5–16.0g/dl). He is prescribed two units of packed red blood cells (red blood cells in a reduced volume of plasma).
He is moved to the high dependency unit where he is found to have a 6L positive fluid balance. He is also oliguric, producing 10–15ml of urine per hour instead of the expected 30ml (0.5ml/kg/h), hypotensive and oedematous.
This was interpreted as being due to low renal perfusion, therefore he is prescribed 1L of NaCl 0.9 per cent and 2L of glucose 5%/NaCl 0.45% every 24 hours for the next three days.
During this time he remains oliguric, but also starts to vomit.
This case illustrates the danger of interpreting single measurements in isolation. It is likely that the low haemoglobin measured at the end of the operation was the result of plasma dilution, as his blood loss had been modest. He was oliguric in the postoperative phase, so he was prescribed fluids to increase his urine output. However, this was probably due to his body retaining large amounts of sodium in response to trauma, rather than low renal perfusion.
Consequently, the additional prescribed fluids will have worsened his condition. The full clinical picture should have been evaluated.
Andrew Staples is a surgical pharmacist, John Dade is a specialist pharmacist for critical care and Chris Acomb is lead pharmacist for professional development, all at Leeds Teaching Hospital NHS Foundation Trust.
References
- National Patient Safety Agency. Patient safety alert 20: Promoting safer use of injectable medicines. London: The Agency; 2007.
- Aber TS, Hosac AM, Veach MP, Pierre YW. Fluid therapy in the critically ill patient. Journal of Pharmacy Practice 2002;15:114.
- Scottish Intercollegiate Guidelines Network. Postoperative management in adults (guideline 77). Edinburgh: The Network; 2004.
- Lobo DN. Fluid, electrolytes and nutrition: physiological and clinical aspects. Proceedings of the Nutrition Society 2004;63:453–66.
- Rivers E, Nguyen B, Hanstad S, Ressler J, Muzzin A, Knoblich B, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. New England Journal of Medicine 2001;345:1368–77
- Medical Devices Agency (now Medicines and Healthcare products Regulatory Agency). Devices Bulletin — Infusion Systems (MDA DB2003(D2)). London: The Agency; 2003.
- Quinn C. Infusion devices: Risks, functions, and management. Nursing Standard 2000;14:35–41.