Ink Drop / Alamy Stock Photo
After reading this article, you should be able to:
- Understand the clinical indications and mechanism of action of isotretinoin;
- Understand appropriate initiation of isotretinoin for acne vulgaris treatment;
- Safely implement pregnancy prevention programmes;
- Identify, prevent and manage common adverse effects.
Acne vulgaris is defined as a chronic inflammatory skin condition1. Prevalence among adolescents and young adults in the UK has steadily increased over the past three decades. Between 1990 and 2021, prevalence in individuals aged 10–24 years rose by around 7.4%. In 2021, an estimated 14.6% of this age group were affected, placing the UK among the top ten countries globally for acne prevalence2. Acne predominantly affects adolescents and young adults but can persist into adulthood, impacting quality of life and self-esteem2,3.
Management of acne in the UK follows a stepwise approach based on severity and response to treatment. Mild cases are typically managed with topical therapies such as benzoyl peroxide, topical antibiotics or retinoids. For moderate acne, combination therapy, often benzoyl peroxide with a topical retinoid or a topical antibiotic is recommended, and oral antibiotics may be introduced for more persistent cases. Hormonal treatments, such as combined oral contraceptives, can be considered for suitable patients4.
In severe or treatment-resistant acne, oral isotretinoin is the gold standard, offering long-term remission but requiring strict monitoring owing to its safety profile. In addition to its well-known teratogenic risk, isotretinoin has been a subject of public concern regarding its potential psychiatric adverse effects. These concerns had resulted in regulatory bodies, such as the Medicines and Healthcare products Regulatory Agency (MHRA), issuing warnings and implementing enhanced monitoring requirements.
Pharmacists play a pivotal role in ensuring the safe and effective use of isotretinoin across both primary and secondary care. In community pharmacy, pharmacists are often the first point of contact for patients seeking advice on side effects, skincare adjustments and safe use of other over-the-counter (OTC) products. They also play a critical role in reinforcing the pregnancy prevention programme (PPP) and ensuring adherence to treatment plans. In hospital settings, pharmacists are integral to monitoring blood tests, managing drug interactions and supporting dermatology teams in dose adjustments. Through proactive counselling and vigilance, pharmacists can optimise patient outcomes and maintain compliance.
Retinoids in acne
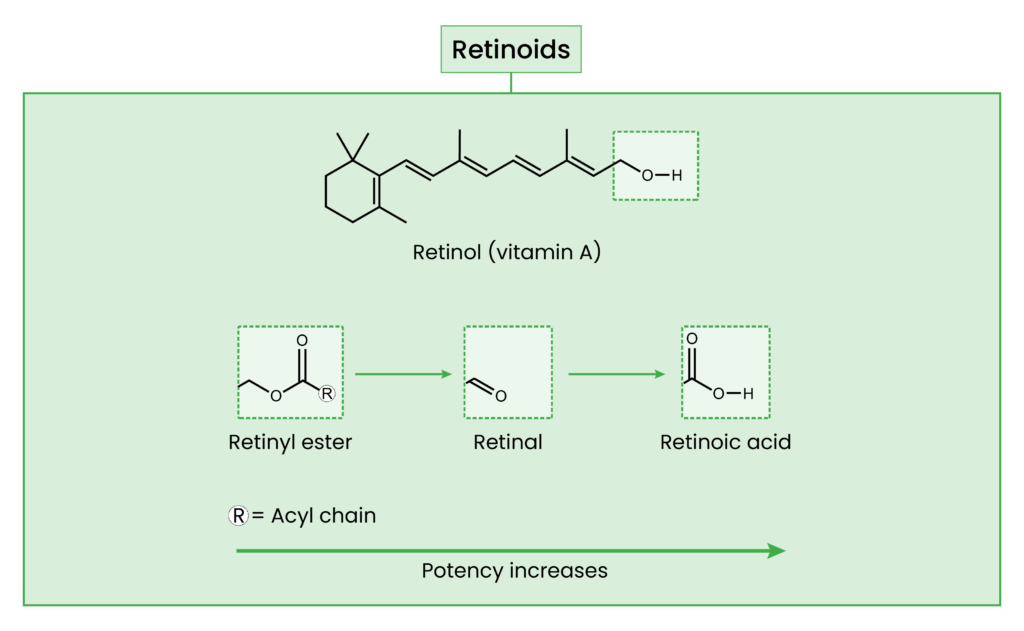
Retinoids are an umbrella term for all compounds that are chemically related to vitamin A. Retinol is the alcohol form of vitamin A and is a specific member of that group. It is the natural form of vitamin A and found in many skincare products. It is one of the most commonly sold OTC retinoids5,6.
Figure 1: Retinoids
PubChem / The Pharmaceutical Journal / J Lipid Res. 2013 Jul;54(7):1731–1743. doi: 10.1194/jlr.R037648
Retinoic acid is the biologically active form that directly affects the skin. Other retinoids are inactive and must be converted into retinoic acid through a series of enzymatic oxidation steps. This conversion pathway generally progresses from retinyl esters to retinol, then to retinaldehyde, and finally to retinoic acid. With each step closer to retinoic acid, both efficacy and the likelihood of irritation increase. Isomers of retinoic acid do not require conversion to exert its effects. One such isomer is tretinoin (the all-trans-retinoic acid isomer)7. In the UK, tretinoin is used topically and is seen in formulations such as gels or creams8. It acts locally on the skin, binding to retinoic acid receptors, which increases cell turnover and unclogs pores. Tretinoin is used for mild-to-moderate acne.
Isotretinoin is a 13-cisoretinoic acid isomer and is available as an oral formulation. In contrast to antibiotics or topical agents that address one or two aspects of acne, isotretinoin targets all the important drivers known to cause acne: sebum suppression, normalising keratinisation, reducing Cutibacterium acnes (C. acnes) and providing an anti-inflammatory effect9–13. Isotretinoin is a very potent, systemic retinoid with long-lasting effects. As such, it is reserved for severe acne and is a last-line treatment. Isotretinoin is therapeutically indicated for severe forms of acne that have not improved with standard treatments, such as oral antibiotics and creams. This includes acne with large, painful lumps (i.e. nodular acne) or clusters of interconnected spots (i.e. conglobate acne), especially when there’s a high risk of permanent scarring4,14. Patients can be considered for isotretinoin initiation if they have moderate-to-severe acne and have not responded to prior treatments, have scarring or experience a high psychosocial impact1.
Isotretinoin initiation
Initiating isotretinoin requires strict protocols and careful patient education. Patients will have an assessment of their mental health prior to starting treatment. Clinicians should screen for symptoms of depression, mood changes, self-harm and suicidal ideation. This assessment is essential because acne itself can affect mental wellbeing and — although rare — isotretinoin has been associated with neuropsychiatric side effects15. These factors should be considered when weighing the benefits versus risks of treatment. Ongoing monitoring at follow-up appointments supports early detection and intervention should any mental health concerns arise. Baseline blood results are required, which include hepatic function and serum lipids. Patients should receive full information regarding the possible side-effects, as well the benefits of this treatment, allowing them to make an informed decision.
For further details, see the later section on psychiatric side effects.
As part of the updated MHRA guidance, the previous requirements for an independent second prescriber before initiating isotretinoin in patients aged under 18 years has now been removed. Healthcare professionals may initiate isotretinoin to this age group without obtaining agreement from a second prescriber; however, all other risk minimisation measures remain in place, and the revised acknowledgement of risk form reflects this change. Importantly, the form also highlights that patients retain the right to seek a second opinion if they choose. In addition, a new patient information video produced to explain the risks of isotretinoin in a clear and accessible format should now be signposted to all patients, who are advised to watch it prior to starting treatment16.
The initial isotretinoin dose is 0.5mg/kg daily in one-to-two divided doses orally. It is increased to 1mg/kg daily based on tolerance and response and is continued until a total cumulative dose of 120–150 mg/kg is achieved. The duration of treatment usually lasts 16–24 weeks, depending on the daily dose and response. Alternatively, treatment can be stopped once there has been sufficient response with no new lesions for at least 4 weeks, even if the cumulative dose is slightly lower14,17.
Isotretinoin is contraindicated in pregnancy and breastfeeding owing to the severe risk of birth defects (teratogenicity). It cannot be commenced in patients of child-bearing potential unless all of the conditions outlined in the PPP are met. All patients of child-bearing potential must complete the acknowledgement of risk form and be enrolled in the PPP.
Isotretinoin commonly affects serum lipids, with triglycerides and total cholesterol rising and HDL-cholesterol occasionally falling during treatment. These changes are often dose-related and are reversible with dose reduction or discontinuation. If hypertriglyceridemia cannot be controlled, treatment should be stopped. As isotretinoin is metabolised in the liver, aspartate aminotransferase (AST) and alanine aminotransferase (ALT) elevations can occur. This is why isotretinoin is contraindicated in hepatic insufficiency. Co-administration with tetracyclines is contraindicated owing to the risk of intracranial hypertension. There should be no overlap when transitioning from antibiotics to isotretinoin18.
Allergies and dietary considerations
The capsules contain a highly refined soya oil as an ingredient. In the UK, medicines containing soya typically carry a warning advising people with peanut or soya allergies to avoid them, owing to the potential risk of an allergic reaction. The precaution includes peanuts, even though isotretinoin does not contain peanut protein, as peanuts and soya belong to related plant families. However, after reviewing the evidence, UK allergy specialists, on behalf of the British Association of Dermatologists (BAD), have confirmed that individuals with a peanut allergy — but no soya allergy — can safely take isotretinoin19.
Each isotretinoin capsule contains a small amount of highly processed soya oil, meaning that people with mild-to-moderate soya allergy can usually take the medication safely. Even though those with severe soya allergy may tolerate treatment, prescribers should seek advice from an allergy specialist before commencing isotretinoin in these20.
The capsules of all available brands of isotretinoin contain gelatine18,21,22. There are no gelatine-free alternatives and, as such, should be highlighted to patients factoring in if they are vegetarian or vegan. The gelatine ingredient may be derived bovine (i.e. beef) or porcine (i.e. pork). Therefore, prior to initiating, ascertaining the patient’s religion and dietary requirements is an important factor to consider (e.g. specific brands use beef derived gelatine and are halal certified).
Pregnancy prevention programme
Prior to commencing isotretinoin, it is mandatory for women of childbearing potential to be enrolled onto the PPP. At initiation, the prescriber has the responsibility to assess and document the PPP status of the patient on the acknowledgement of risk form, see Table 1.
Table 1: The four ‘Pregnancy prevention programme’ status categories
Patients are assigned one category. The category type is dependent on the whether the patient is sexually active or abstinent and the type of contraception they are on, if any. The PPP status dictates the maximum duration of isotretinoin that can be prescribed23. Those that are sexually active and are within child-bearing age are required to be on a form of contraception. For pharmacists, it is vital to ensure the quantity of isotretinoin being prescribed on prescription is in line with PPP status.
It is strongly advised that individuals to wait at least one month after their last administered dose of isotretinoin before trying to conceive, ensuring the active metabolite is sufficiently cleared, thereby reducing the risk of teratogenic exposure. In pregnancy, there is on average a 3–5% chance of having a birth defect24, which is increased in up to 28% or more if isotretinoin is taken during pregnancy. Birth defects been associated with small or absent ears, along with hearing and vision impairments25. Other reported anomalies include congenital defects, cleft palate or absence of the thymus gland to name a few26. If pregnancy occurs while on isotretinoin, termination is highly advised, as well as discontinuing isotretinoin.
Monitoring during treatment
Baseline blood assessments are essential, which includes liver function tests (LFTs) and fasting lipids. Full blood count (FBC) should also be carried out at baseline for safety; however, they are not mandatory in follow-ups.
The results of a retrospective cohort study by Emteani et al., published in 2024, encompassing more than 79,000 patients on isotretinoin for acne, found that within the first three months of initiating isotretinoin, patients had raised ALT and were 17 times more likely to experience hypertriglyceridaemia than patients who were commenced on oral antibiotics. Although the study results showed that isotretinoin resulted with a greater risk of hypertriglyceridaemia and hypertransaminasaemia, the absolute incidence of severe abnormalities was low27–29. In continually raised serum lipids and/or LFTs, dose reduction or discontinuation is advised.
Lipid and LFTs elevations are usually reversible upon discontinuation and do not pose significant long-term cardiovascular risks30,31. Monitoring is required but can be scheduled. In 2023, the Isotretinoin Implementation Advisory Expert Working Group (IIAEWG) published the independent ‘Report of the Commission on Human Medicines Isotretinoin Implementation Advisory Working Group’23. The report stipulates that bloods, including fasting serum lipids and liver enzymes, should be undertaken during the initiation of treatment. This should then be repeated after one month and subsequently at three monthly intervals thereafter, unless more frequent monitoring is clinically indicated.
Contraindications and cautions
For a summary of the absolute contraindications for isotretinoin and their rationales, see Table 2.
Table 2: Absolute contraindications for isotretinoin
Table 3 outlines cautions for isotretinoin and how to manage them8,20–23.
Table 3: Cautions for isotretinoin
Adverse effects and practical management
The most common side-effects of isotretinoin involve the mucocutaneous, musculoskeletal and ophthalmic systems32. Isotretinoin significantly reduces the sebaceous gland activity and alters the lipid composition of the skin and mucosa. This leads to a loss of natural protective barrier, resulting in an increase of transepidermal water loss (TEWL) and decreased skin hydration. The most common side-effect experienced by patients owing to this is severe dry skin (i.e. xerosis) and chapped lips (i.e. cheilitis)28,33. Redness of the skin (i.e. erythema) and dry eyes are also common side-effects associated with this.
Dry skin can be managed using moisturisers. Cream-based formulations are recommended for mild dryness, whereas thicker ointments are more beneficial for more severe dryness. Moisturisers containing fragrance or alcohol can irritate and weaken the skin barrier, exacerbating dryness and inflammation34. Patients on isotretinoin, fragrance-free and alcohol-free formulations are strongly recommended for this reason. Additionally, comedogenic ingredients should be avoided as it can block pores, leading to open and closed comedones17. Ceramides are lipids found naturally on the skin. In acne affected skin, reduced ceramide levels have been observed, which may play a role in comedone formation35. Ceramide-rich moisturisers are also recommended for this reason.
A 2020 review involving 38 studies examined the effects of omega 3 intake and skin health. The results of the review found that oral omega-3 also reduced mucocutaneous side effects associated with isotretinoin, which included dry lips, nose and skin36. There is also evidence to suggest omega-3 oils improve lipid metabolism37.
For more information about side effects and their management, see Figure 216,17,23,38–45 .
Figure 2: Side effects
Psychiatric side effects
Among the public, psychiatric side effects are the most widely recognised risks associated with isotretinoin. This is heightened with the increasing use of social media for health information46–49. Given the widespread circulation of anecdotal reports and misinformation on social media, pharmacists have an important role in supporting patients to interpret health information critically50. By actively combating misinformation, pharmacists can reduce unnecessary anxiety, improve adherence and ensure that decisions about isotretinoin are founded on accurate evidence rather than online narratives51,52. This is particularly valuable for young people, who are among the highest users of social media for health information and may be disproportionately exposed to such sources53,54.
There is an ongoing debate, as there is currently conflicting literature linking isotretinoin to depression55–57. It is hypothesised that isotretinoin may influence mood by interacting with neurotransmitter systems, such as dopamine and serotonin, potentially altering their levels and affecting psychiatric disorders58,59. The results of a 2023 meta-analysis of 25 studies, covering over 1.6 million users, found a one-year absolute risk of suicide, suicidal ideation and self-harm among users to be less than 0.5% each and depression was found to be in 3.83%; therefore, although still an incident that may occur, it is a relatively rare incidence. At a population level, there was no increased risk of suicide or psychiatric disorders among those taking isotretinoin60–63. Isolated cases can still exist, therefore patients that do experience signs of severe psychiatric changes, such as suicidal thoughts, should discontinue the medication immediately and seek urgent attention.
Interactions
Tetracycline antibiotics are contraindicated with isotretinoin owing to the risk of benign intracranial hypertension22. Doxycycline can be used for the treatment of acne prior to initiating on isotretinoin64. As a result, it is vital to do a full drug history prior to commencing and ensuring to regularly ask about other medications the patient may be taking. Pharmacists play an important role in this process, by reviewing prescriptions and counselling patients to ensure there are no interacting medicines dispensed.
Isotretinoin is a systemic oral retinoid that can increase skin sensitivity and irritation. Combining this with other prescription or OTC acne treatments requires careful consideration to avoid reduced efficacy or exacerbating adverse effects17. For this reason, other topical retinoids — such as adapalene, Epiduo or skincare products that contain exfoliating acids — such as salicylic acid and benzoyl peroxide, should be discontinued to avoid the increases risk of irritation and additive effects65.
As the skin becomes more sensitive and fragile during treatment, physical exfoliants (i.e. scrubs) should be avoided owing to the likelihood of increased irritation. Cosmetic procedures, such as waxing, chemical peels and laser treatments, should also be avoided during and for six months post treatment17.
Common patient questions and counselling advice
Patients may ask pharmacy professionals with questions regarding their condition and treatment. Some common questions and appropriate advice can be seen in Table 466.
Table 4: Common questions asked by patients
Conclusion
This article has summarised evidence-based best practice for the use of oral isotretinoin in acne vulgaris. Applying these principles enables pharmacists across primary and secondary care to optimise efficacy, while minimising harm and ensuring compliance.
- 1.Acne vulgaris. National Institute for Health and Care Excellence. 2026. https://cks.nice.org.uk/topics/acne-vulgaris/
- 2.Zhu Z, Zhong X, Luo Z, et al. Global, regional and national burdens of acne vulgaris in adolescents and young adults aged 10–24 years from 1990 to 2021: a trend analysis. British Journal of Dermatology. 2024;192(2):228-237. doi:10.1093/bjd/ljae352
- 3.Samuels DV, Rosenthal R, Lin R, Chaudhari S, Natsuaki MN. Acne vulgaris and risk of depression and anxiety: A meta-analytic review. Journal of the American Academy of Dermatology. 2020;83(2):532-541. doi:10.1016/j.jaad.2020.02.040
- 4.Acne vulgaris: Management. National Institute for Health and Care Excellence. 2023. https://www.nice.org.uk/guidance/ng198
- 5.Milosheska D, Roškar R. Use of Retinoids in Topical Antiaging Treatments: A Focused Review of Clinical Evidence for Conventional and Nanoformulations. Adv Ther. 2022;39(12):5351-5375. doi:10.1007/s12325-022-02319-7
- 6.Motamedi M, Chehade A, Sanghera R, Grewal P. A Clinician’s Guide to Topical Retinoids. J Cutan Med Surg. 2021;26(1):71-78. doi:10.1177/12034754211035091
- 7.Zasada M, Budzisz E. Retinoids: active molecules influencing skin structure formation in cosmetic and dermatological treatments. pdia. 2019;36(4):392-397. doi:10.5114/ada.2019.87443
- 8.Acne – Treatment. National Health Service. https://www.nhs.uk/conditions/acne/treatment/
- 9.Goldstein JA. Isotretinoin in the Treatment of Acne. Arch Dermatol. 1982;118(8):555. doi:10.1001/archderm.1982.01650200023009
- 10.Geiger JM, Hommel L, Harms M, Saurat JH. Oral 13-cis retinoic acid is superior to 9-cis retinoic acid in sebosuppression in human beings. Journal of the American Academy of Dermatology. 1996;34(3):513-515. doi:10.1016/s0190-9622(96)90462-4
- 11.Layton A. The use of isotretinoin in acne. Dermato-Endocrinology. 2009;1(3):162-169. doi:10.4161/derm.1.3.9364
- 12.Ryan-Kewley AE, Williams DR, Hepburn N, Dixon RA. Non-antibiotic Isotretinoin Treatment Differentially Controls Propionibacterium acnes on Skin of Acne Patients. Front Microbiol. 2017;8. doi:10.3389/fmicb.2017.01381
- 13.Pile H, Patel P. Isotretinoin. StatPearls Publishing; 2025. https://www.ncbi.nlm.nih.gov/sites/books/NBK525949/
- 14.Isotretinoin. National Institute for Health and Care Excellence . https://bnf.nice.org.uk/drugs/isotretinoin/
- 15.Review of isotretinoin and psychiatric adverse reactions. Medicines and Healthcare products Regulatory Agency . 2014. https://assets.publishing.service.gov.uk/media/655201b48a2ed40013720d44/Review_of_isotretinoin_and_psychiatric_adverse_reactions__2014_.pdf
- 16.Isotretinoin: changes to prescribing guidance and additional risk minimisation measures. Medicines and Healthcare products Regulatory Agency . 2026. https://www.gov.uk/drug-safety-update/isotretinoin-changes-to-prescribing-guidance-and-additional-risk-minimisation-measures
- 17.Isotretinoin patient information leaflet. British Association of Dermatologists . 2020. https://cdn.bad.org.uk/uploads/2021/12/29200301/Isotretinoin-Updated-July-2019-lay-reviewed-June-2019-amended-January-2020.pdf
- 18.Isotretinoin 20 mg capsules: summary of product characteristics (SmPC). Ennogen IP Ltd. 2026. https://www.medicines.org.uk/emc/product/100786/smpc
- 19.Isotretinoin and soya allergy. British Association of Dermatologists . 2023. https://www.skinhealthinfo.org.uk/condition/isotretinoin-and-soya-allergy/
- 20.Denorme P, Schrijvers R, Van Hoeyveld E, et al. Isotretinoin in Severe Peanut- and Soy-Allergic Patients: Is it Safe or Not? J Investig Allergol Clin Immunol. 2019;29(2):146-148. doi:10.18176/jiaci.0352
- 21.Roaccutane 20 mg soft capsules: summary of product characteristics (SmPC). Neon Healthcare Limited. 2026. https://www.medicines.org.uk/emc/product/100634/smpc
- 22.Isotretinoin soft capsules: summary of product characteristics (SmPC). Ranbaxy (UK) Limited a Sun Pharmaceutical Company. 2026. https://www.medicines.org.uk/emc/product/10556/smpc
- 23.Independent report of the Isotretinoin Implementation Advisory Expert Working Group (IIAEWG). Medicines and Healthcare products Regulatory Agency . 2023. https://assets.publishing.service.gov.uk/media/653fd71946532b001467f58b/IIAEWGreport.pdf
- 24.Birth defects. Centers for Disease Control and Prevention . https://www.cdc.gov/birth-defects/about/index.html
- 25.Sladden MJ, Harman KE. What Is the Chance of a Normal Pregnancy in a Woman Whose Fetus Has Been Exposed to Isotretinoin? Arch Dermatol. 2007;143(9). doi:10.1001/archderm.143.9.1187
- 26.Isotretinoin (Accutane®). In: Mother To Baby Fact Sheets. Organization of Teratology Information Specialists ; 1994. https://www.ncbi.nlm.nih.gov/books/NBK582775/
- 27.Eichenfield DZ, Sprague J, Eichenfield LF. Management of Acne Vulgaris. JAMA. 2021;326(20):2055. doi:10.1001/jama.2021.17633
- 28.Brzezinski P, Borowska K, Chiriac A, Smigielski J. Adverse effects of isotretinoin: A large, retrospective review. Dermatologic Therapy. 2017;30(4):e12483. doi:10.1111/dth.12483
- 29.Emtenani S, Abdelghaffar M, Ludwig RJ, Schmidt E, Kridin K. Risk and timing of isotretinoin‐related laboratory disturbances: a population‐based study. Int J Dermatology. 2024;63(12):1740-1747. doi:10.1111/ijd.17225
- 30.Sibi Krishna T, Kaur R, Malhotra V, et al. The Impact of Isotretinoin on Lipid Profile: a Systematic Review. Annals of Medicine & Surgery. 2025;87(7):4395-4403. doi:10.1097/ms9.0000000000003366
- 31.Tawanwongsri W, Kanchanasuwan T, Eden C. Isotretinoin and Hepatotoxicity in Patients with Acne: A Narrative Review. Cosmetics. 2025;12(1):17. doi:10.3390/cosmetics12010017
- 32.Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. Journal of the American Academy of Dermatology. 2016;74(5):945-973.e33. doi:10.1016/j.jaad.2015.12.037
- 33.Melnik B. Apoptosis May Explain the Pharmacological Mode of Action and Adverse Effects of Isotretinoin, Including Teratogenicity. Acta Derm Venerol. 2017;97(2):173-181. doi:10.2340/00015555-2535
- 34.Zirwas M, Stechschulte S. Moisturizer allergy: diagnosis and management. The journal of clinical and Aesthetic dermatology. Published online 2008. https://pubmed.ncbi.nlm.nih.gov/21212847/
- 35.Schachner LA, Alexis AF, Andriessen A, et al. Insights into acne and the skin barrier: Optimizing treatment regimens with ceramide‐containing skincare. J of Cosmetic Dermatology. 2023;22(11):2902-2909. doi:10.1111/jocd.15946
- 36.Thomsen BJ, Chow EY, Sapijaszko MJ. The Potential Uses of Omega-3 Fatty Acids in Dermatology: A Review. J Cutan Med Surg. 2020;24(5):481-494. doi:10.1177/1203475420929925
- 37.Basirat A, Merino-Torres JF. Marine-Based Omega-3 Fatty Acids and Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. 2025;17(20):3279. doi:10.3390/nu17203279
- 38.Barbieri JS. Isotretinoin for Treatment of Acne. JAMA Dermatol. 2023;159(12):1403. doi:10.1001/jamadermatol.2023.2065
- 39.Emine Müge Acar, Senem Şaş, Fatmanur Aybala Koçak. Evaluation of musculoskeletal adverse effects in patients on systemic isotretinoin treatment: A cross-sectional study. Archives of Rheumatology. 2022;37(2):223-229. doi:10.46497/archrheumatol.2022.8645
- 40.Sumitani Y, Uchibe K, Yoshida K, et al. Inhibitory effect of retinoic acid receptor agonists on in vitro chondrogenic differentiation. Anat Sci Int. 2019;95(2):202-208. doi:10.1007/s12565-019-00512-3
- 41.Raneses E, Schmidgal EC. Rhabdomyolysis Caused by Isotretinoin and Exercise in an Otherwise Healthy Female Patient. Cureus. Published online June 15, 2022. doi:10.7759/cureus.25981
- 42.Reyes-Hadsall S, Ju T, Keri JE. Use of Oral Supplements and Topical Adjuvants for Isotretinoin-Associated Side Effects: A Narrative Review. Skin Appendage Disord. 2023;10(1):1-9. doi:10.1159/000533963
- 43.Isotretinoin (Roaccutane): rare reports of erectile dysfunction and decreased libido. Medicines and Healthcare products Regulatory Agency . 2017. https://www.gov.uk/drug-safety-update/isotretinoin-roaccutane-rare-reports-of-erectile-dysfunction-and-decreased-libido
- 44.Oi-Yee Li H, Pastukhova E, Brandts-Longtin O, Bailey A, Tan MG, Kirchhof MG. Sexual dysfunction following retinoid treatment: a systematic review. British Journal of Dermatology. 2024;192(1):175-177. doi:10.1093/bjd/ljae361
- 45.Tan E, Kennedy H, Rademaker M. Exploring the association between isotretinoin and sexual dysfunction: a comprehensive scoping review. Clinical and Experimental Dermatology. 2024;49(11):1396-1404. doi:10.1093/ced/llae168
- 46.Bal ZI, Karaosmanoglu N, Temel B, Orenay OM. Trust in Dermatologists Versus Social Media Influencers Among Acne Patients. Cureus. Published online May 11, 2025. doi:10.7759/cureus.83930
- 47.Asare AO, Sefa AN, Haddad L, Obeng SEL. Isotretinoin on TikTok: A Qualitative Analysis of Attitudes, Perspectives, and Knowledge Dissemination of User-Generated Content. Cureus. Published online March 8, 2025. doi:10.7759/cureus.80244
- 48.Hackley M, Al-Bataineh D, Lipoff JB. Portrayal of mental health effects of isotretinoin on TikTok. JAAD International. 2024;14:90-91. doi:10.1016/j.jdin.2023.10.008
- 49.Crilly P, Jair S, Mahmood Z, et al. Public views of different sources of health advice:pharmacists, social media and mobile health applications. International Journal of Pharmacy Practice. 2018;27(1):88-95. doi:10.1111/ijpp.12448
- 50.Suarez-Lledo V, Alvarez-Galvez J. Prevalence of Health Misinformation on Social Media: Systematic Review. J Med Internet Res. 2021;23(1):e17187. doi:10.2196/17187
- 51.Erku DA, Belachew SA, Abrha S, et al. When fear and misinformation go viral: Pharmacists’ role in deterring medication misinformation during the “infodemic” surrounding COVID-19. Research in Social and Administrative Pharmacy. 2021;17(1):1954-1963. doi:10.1016/j.sapharm.2020.04.032
- 52.Covvey JR, Harpe SE. Dispensing The Truth: The Need to Educate Future Pharmacists to Counter Health Misinformation. American Journal of Pharmaceutical Education. 2025;89(11):101874. doi:10.1016/j.ajpe.2025.101874
- 53.Goodyear VA, Armour KM, Wood H. Young people and their engagement with health-related social media: new perspectives. Sport, Education and Society. 2018;24(7):673-688. doi:10.1080/13573322.2017.1423464
- 54.Lim MSC, Molenaar A, Brennan L, Reid M, McCaffrey T. Young Adults’ Use of Different Social Media Platforms for Health Information: Insights From Web-Based Conversations. J Med Internet Res. 2022;24(1):e23656. doi:10.2196/23656
- 55.Saitta P, Keehan P, Yousif J, Way B, Grekin S, Brancaccio R. An update on the presence of psychiatric comorbidities in acne patients, Part 2: Depression, anxiety, and suicide. Cutis. Published online 2011. https://pubmed.ncbi.nlm.nih.gov/21916276/
- 56.Bachmann C, Grabarkiewicz J, Theisen F, Remschmidt H. Isotretinoin, Depression and Suicide Ideation in an Adolescent Boy. Pharmacopsychiatry. 2007;40(3):128-128. doi:10.1055/s-2007-972575
- 57.Huang YC, Cheng YC. Isotretinoin treatment for acne and risk of depression: A systematic review and meta-analysis. Journal of the American Academy of Dermatology. 2017;76(6):1068-1076.e9. doi:10.1016/j.jaad.2016.12.028
- 58.Bremner JD, McCaffery P. The neurobiology of retinoic acid in affective disorders. Progress in Neuro-Psychopharmacology and Biological Psychiatry. 2008;32(2):315-331. doi:10.1016/j.pnpbp.2007.07.001
- 59.Charest A, Wainer B, Albert P. Cloning and differentiation-induced expression of a murine serotonin1A receptor in a septal cell line. J Neurosci. 1993;13(12):5164-5171. doi:10.1523/jneurosci.13-12-05164.1993
- 60.Tan NKW, Tang A, MacAlevey NCYL, Tan BKJ, Oon HH. Risk of Suicide and Psychiatric Disorders Among Isotretinoin Users. JAMA Dermatol. 2024;160(1):54. doi:10.1001/jamadermatol.2023.4579
- 61.Li C, Chen J, Wang W, Ai M, Zhang Q, Kuang L. Use of isotretinoin and risk of depression in patients with acne: a systematic review and meta-analysis. BMJ Open. 2019;9(1):e021549. doi:10.1136/bmjopen-2018-021549
- 62.Droitcourt C, Nowak E, Rault C, et al. Risk of suicide attempt associated with isotretinoin: a nationwide cohort and nested case-time-control study. International Journal of Epidemiology. 2019;48(5):1623-1635. doi:10.1093/ije/dyz093
- 63.Kridin K, Ludwig RJ. Isotretinoin and the risk of psychiatric disturbances: A global study shedding new light on a debatable story. Journal of the American Academy of Dermatology. 2023;88(2):388-394. doi:10.1016/j.jaad.2022.10.031
- 64.Doxycycline 100mg Capsules: Summary of product characteristics (SmPC). Sovereign Medical. https://www.medicines.org.uk/emc/product/13082/smpc#companyDetails
- 65.Isotretinoïne DOC 10 mg, 20 mg zachte capsules: summary of product characteristics (SmPC). College ter Beoordeling van Geneesmiddelen . 2021. https://www.geneesmiddeleninformatiebank.nl/smpc/h131384_smpc_en.pdf
- 66.Lai J, Barbieri JS. Acne Relapse and Isotretinoin Retrial in Patients With Acne. JAMA Dermatol. 2025;161(4):367. doi:10.1001/jamadermatol.2024.5416