Shutterstock.com
Oral candidiasis is an opportunistic infection of the oral cavity often caused by the overgrowth of Candida, a yeast-like fungus commonly found in the gastrointestinal tract of humans, as normal skin flora and in mucous membranes[1]
. Candida albicans (C. albicans) accounts for around 80% of infections and can colonise the cavity, either alone or in combination with non-albican species, including Candida glabrata and Candida
tropicalis
[1],[2]
. The typical colonisation rate of C. albicans varies with age. In neonates it is 45%; in healthy children 45–65%; in healthy adults 30–45%; in denture wearers 50–65%; in patients living in acute or long-term facilities, such as nursing or residential homes, 65–88%; and in immunocompromised patients, such as those with HIV and/or undergoing chemotherapy for acute leukaemia, it is 95% and 90% respectively[1],[3]
. It is unclear why the carriage rate varies with age.
With increased availability and prescribing of broad-spectrum antibacterials (e.g. penicillins, fluoroquinolones, macrolides) and immunosuppressive agents (e.g. corticosteroids, azathioprine, methotrexate), and with increased immunosuppressive comorbidities, including diabetes, cancer and AIDS, there has been an increase in the number of reported cases of opportunistic oral Candida infections[3]
. While not life-threatening for most patients, it can cause significant patient discomfort and, in elderly or hospitalised patients, can result in significant morbidity owing to impaired nutrition[3]
.
However, in severely immunosuppressed patients, invasive and life-threatening systemic Candida infection may develop. A tertiary care hospital reported that, of their patients suffering from Candida bloodstream infection, 45% received immunosuppressive therapy[4]
.
More than half a million prescriptions for oral Candida infections are issued each year in England by GPs[5]
.
This article aims to aid pharmacists in the diagnosis and management of patients suffering from oral candidiasis. It provides assistance in recognising patients at risk of mucosal fungal infections, and the pharmacological and non-pharmacological options to prevent occurrence and treat the infection.
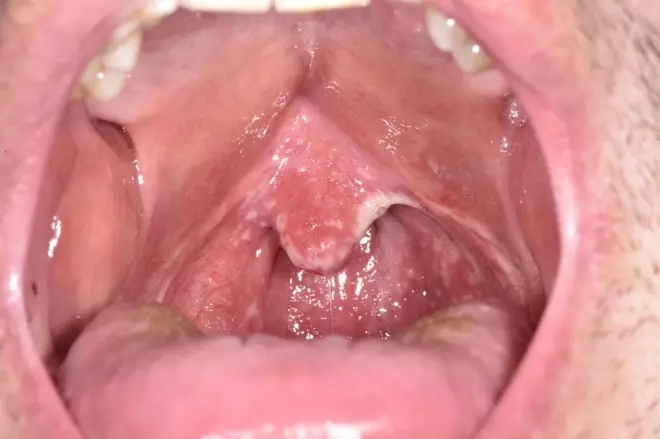
Figure 1: an example of mild pseudomembranous candidiasis (white areas)
Source: Science Photo Library
Classification
There are several classifications of oral candidiasis, and their similarities and differences are outlined below[1],[2]
.
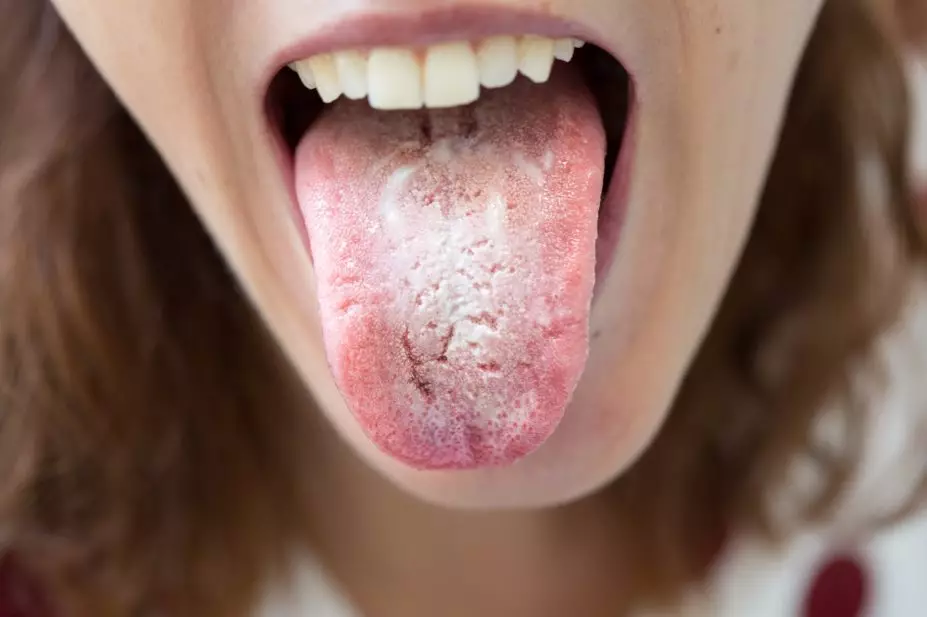
Pseudomembranous candidiasis is the most common type and is characterised by an extensive white ‘cottage cheese-like’ film, found on the buccal mucosa, tongue, periodontal tissues and oropharynx[1],[3]
. The plaque can usually be scraped off to expose an underlying erythematous mucosa[2]
. If thrush is associated with the use of corticosteroid inhalers, rinsing the mouth with water (or cleaning a child’s teeth if not able to rinse and spit) immediately after using the inhaler may avoid the problem. Patients should be counselled on good dental hygiene on initiation of corticosteroid inhalers[6]
. See Box for preventative strategies for oral candidiasis[7]
.
Acute atrophic candidiasis is associated with a burning sensation in the mouth or on the tongue, and often referred to as ‘antibiotic sore mouth’, because of its association with prolonged use of broad-spectrum antibiotics[1]
. The tongue may be bright red and painful. Although this type of candidiasis is less common, diagnosis may be difficult, but should be considered in the differential diagnosis of a sore tongue, especially in a frail, older patient with dentures who has received antibiotic therapy or who is on inhaled steroids. Other conditions that may be confused with acute atrophic candidiasis include mucositis, denture stomatitis, erythema migrans, thermal burns, erythroplakia and anaemia[3]
.
Chronic presentations of oral candidiasis can occur, often with chronic inflammation associated with denture usage[3]
.
Angular chelitis is defined as fissuring, scaling and erythema of the corners of the mouth. It may be associated with Candida infection, but can be co-infected with staphylococcus or streptococcus
bacteria, which can complicate treatment and lead to other oral bacterial infections[2]
.
Box: Prevention of oral candidiasis infection
If some of the predisposing factors listed in Table 1 apply, there are several practical considerations patients can take to reduce the risk of infection.
- Rinsing mouth out after eating, taking medication or using a corticosteroid inhaler;
- Good oral hygiene, brushing teeth twice per day;
- Going for regular dental check-ups (even if patients do not have any teeth).
For denture wearers:
- Properly cleaning dentures;
- Brushing gums with a soft toothbrush;
- Removing dentures each night;
- Ensuring dentures fit properly and are not too loose.
For infants:
- Sterilising dummies regularly and bottles after each use;
- If using corticosteroid inhalers, rinsing the mouth with water or cleaning a child’s teeth (if not able to rinse and spit) immediately after using the inhaler.
Source: NHS[7]
Risk factors
Local and systemic factors of the host can predispose patients to becoming infected with a Candida species. The specifics of these are discussed below, and summarised in Table 1.
Local factors
Reduced salivary production can predispose patients to oral candidiasis, as salivary constituents inhibit the overgrowth of Candida. Therefore, conditions reducing the amount and characteristics of saliva secretions may lead to a Candida overgrowth[3]
.
Dental prostheses, such as dentures or fillings, can create a favourable environment for the Candida organisms to latch[3]
. Topical or inhaled corticosteroids temporarily suppress the oral immune system and cause alterations in the oral flora, leading to an overgrowth of Candida
[3]
.
Unbalanced dietary intake of sugars, carbohydrates and dairy products can promote Candida growth by making the oral cavity more acidic and consequently favouring the Candida organisms[8]
.
Systemic factors
Extremes of age may predispose individuals to candidiasis owing to immature or weakened immunity, along with the variations in the Candida carriage rates[3]
.
Malnutrition, particularly in iron but also in other nutrients such as essential fatty acids, folic acid, vitamins A and B6, magnesium, selenium and zinc, is often associated with increased risk of oral candidiasis. Iron deficiency diminishes the fungistatic action of transferrin and other iron-dependent enzymes used in suppressing fungal overgrowth in the oral cavity,[8]
.
Prolonged use of broad-spectrum antibiotics (e.g. co-amoxiclav), or immunosuppressants (e.g. azathioprine), alters the local oral flora by killing off bacteria and suppressing the immune system. This results in a favourable environment for Candida to grow[8]
.
Oral and invasive candidiasis is more frequently reported in patients with endocrine dysfunctions, such as diabetes and Cushing’s syndrome; in immunodeficiency conditions such as AIDS; and in patients receiving chemotherapy and radiotherapy for the treatment of cancers. This is owing to the reasons previously outlined; these morbidities impair the host defence mechanisms, leading to an overgrowth in the oral flora[3]
.
| Local factors | Systemic factors |
|---|---|
| Source: Postgrad Med J [1] , Front Microbiol [3] , Critic Rev Microbiol [9] | |
| • Impaired local defense mechanisms | • Impaired systemic defense mechanisms |
| • Reduced saliva production | • Immunodeficiency (e.g. AIDS) |
| • Smoking | • Immunosuppressive medications (e.g azathioprine) |
| • Atrophic oral mucosa | • Endocrine disorders (e.g. diabetes) |
| • Mucosal diseases (e.g. oral lichen planus) | • Malnutrition |
| • Topical medications (e.g. corticosteriods) | • Cancers |
| • Decreased blood supply (e.g. caused by radiotherapy or vasculitis) | • Some congenital conditions |
| • Poor oral hygiene | • Broad-spectrum antibiotic therapy |
| • Dental prostheses or dentures | |
| • Altered or immature oral flora | |
Diagnosis
Recognition of the associated lesions, such as the white plaque seen in the Figure, via an oral or oesophageal examination (i.e. examining the back of the throat) should provide a diagnosis of the more common forms of oral candidiasis (e.g. pseudomembranous candidiasis). Diagnosis can, however, be confirmed microscopically via a mucosal smear or biopsy as a Candida overgrowth, and should be considered for refractory disease or the alternative presentations of the condition[1],[2],[3]
. A positive microbiology result for Candida alone does not indicate a necessity for treatment as patients are routinely colonised, as aforementioned[2]
. Oral candidiasis is uncommon in healthy adults and may be the first presentation of an undiagnosed risk factor.
Treatment
Topical
Traditionally, topical antifungals are the preferred treatment for oral candidiasis. Locally administered antifungals offer the advantage of reduced systemic exposure, which results in fewer adverse drug reactions or interactions. The British National Formulary (BNF) lists two options: nystatin and miconazole[10]
. However, the four-times-per-day administration makes patient adherence for the requisite 7–14 days challenging. Reiterating the importance of this regular administration to patients for preventing infection can help improve compliance.
Nystatin is the most commonly used topical treatment of oral candidiasis in the UK, with little systemic exposure expected[11],[12]
. The nystatin oral suspension is rapidly cleared from the oral tract and concentrations fall quickly to subtherapeutic levels. Therefore, the resultant short contact time with the oral mucosa can contribute to reduced efficacy[11],[12]
. For this reason, nystatin should generally be avoided in severe infection or in immunocompromised patients[13]
.
Miconazole, an imidazole, can be used for local application in the mouth. It remains a useful and effective treatment for all types of oral candidiasis and has a broad spectrum of activity against many species of Candida, including C. albicans
[11]
. A recent systematic review showed that miconazole therapy is superior to nystatin for oral candidiasis and is associated with a decrease in treated infection relapses[14]
. However, concomitant use of miconazole oral gel and warfarin can result in life-threatening international normalised ratio (INR) derangement owing to the gel being absorbed through the oral mucosa or the gastrointestinal tract when swallowed[15],[16]
. Co-administration of these medicines is contraindicated and should be avoided, as outlined by the Medicines and Healthcare products Regulatory Agency. Patients should be advised to use alternative topical therapies, such as nystatin. However, if the oral candidiasis is refractory to nystatin, then the patient should receive systemic therapy with a triazole, with close monitoring of their INR, and should be informed of the increased risk of bleeding with this therapy.
Topical fluconazole, although traditionally unused within UK clinical practice, has been reported for the treatment of oral candidiasis, although this usage is an off-label application of oral fluconazole suspension. Owing to its good adhesion to the surface of the oral mucosa and a once-daily administration, fluconazole 50mg/5mL oral solution mouthwash is an option for uncomplicated oral candidiasis[12]
. A small, randomised control trial (n=34) reported that fluconazole oral suspension was superior to nystatin at treating oral candidiasis in infants[17]
. It was found that nystatin adhered poorly to the oral mucosa, resulting in shorter time in the oral cavity and a faster ingestion of the suspension, therefore having a lower efficiency compared with fluconazole. Another small, open-label study (n=19) looked at using fluconazole suspension (2mg/mL three times per day) as a rinse and spit regimen, obtaining 94% cure rates[18]
.
Systemic treatment
Reserved for more invasive infections, patients with concurrent immunodeficiency or where compliance with topical therapies is challenging, systemic treatment options include oral fluconazole first-line, with itraconazole generally reserved for refractory infection (see Table 2)[10]
.
Fluconazole remains the first-line systemic antifungal for oral candidiasis treatment[10],[19]
. Fluconazole has a broad range of activity against Candida
spp, is well tolerated orally, and has a relatively high oral bioavailability[11],[12]
. The majority of the evidence for oral fluconazole in adults is with 100–200mg once per day for 7–14 days[3],[11],[19],[20],[21]
. Little published evidence exists for the use of lower systemic doses (50mg/day) for pseudomembranous Candida infection advised in the UK. However, this controversy is generally considered moot, as the 50mg/day dose has become established clinical practice. In initial and simple presentations of oral candidiasis, this dose is likely sufficient; higher doses are often reserved for the more resistant strains of Candida.
A correlation between the pathogen minimum inhibitory concentration (MIC) and fluconazole exposure (area under the curve) predicts clinical efficacy in the treatment of mucosal candidiasis. Optimum efficacy is seen with a total daily dose (TDD)/MIC ratio of greater than 100 for patients treated for oral candidiasis with concurrent HIV infection[22]
. Lower TDD/MIC ratios (>50) are acceptable in immunocompetent hosts and elicit an acceptable clinical response[22]
. The lab-based breakpoint for identifying fluconazole-sensitive C. albicans is 2mg/L; with the majority of isolates it is 1mg/L or less[23]
. Dosing of 50mg/day is acceptable for most immunocompetent patients with C. albicans infection. Higher doses (100mg/day or more) are advised in immunocompromised patients or if non-albican species are identified. Bespoke dosing (TDD = 100 x MIC value) can also be considered in high MIC pathogen infections.
C. albicans fluconazole-resistant pathogens remain fortunately uncommon in clinical practice[24]
. The impact of low-dosed fluconazole, particularly when prolonged for Candida prophylaxis in immunocompromised hosts, on antifungal resistance is less clear. Trends in resistance patterns are challenging owing to low numbers of patients tested and heterogeneity of clinical presentation. Low-dose fluconazole (50mg/day) in treatment or prophylaxis may have a role in selecting out non-albican species where MIC values to fluconazole are typically higher. Further study is required in this area, but oropharyngeal infection associated with non-albican Candida should be suspected in refractory infection where first line fluconazole is unsuccessful. A swab of the oral mucosa should be considered to aid diagnostics in identifying Candida species, and whether this pathogen is fluconazole resistant. This will help determine if either higher doses of fluconazole are required or an alternate agent is needed (e.g. itraconazole).
| Drug | Route | Dose |
|---|---|---|
| Source: British National Formulary 2020[10] | ||
| Nystatin | Topical (i.e. oral suspension) | 1mL (100 ,000  units) four times per day for at least 7 days, and continued for 48 hours after lesions have resolved[25] |
| Miconazole | Topical (i.e. oral gel) | 2.5 mL four times per day times for at least 7 days and continued until after lesions have healed or symptoms have cleared[26] |
| Fluconazole | Topical (i.e. oral suspension) and systemic (i.e. enteral) | 50 mg daily given for 7–14 days in oropharyngeal candidiasis (maximum 14 days, except in severely immunocompromised patients). An increased dose of 100 mg daily is advised for “unusually difficult infections”[27] |
| Itraconazole | Systemic (i.e. enteral) | 100–200 mg twice daily for 2 weeks (continue for another 2 weeks if no response)[28] |
There are some discrepancies with BNF treatment strategies and that of other international recommendations for oral candidiasis management. For example, the Infectious Diseases Society of America (IDSA) provides comprehensive guidance on the management of oral candidiasis that differs on dosing advice provided by the BNF[19]
. Most notably, dosing of nystatin oral solution and systemic fluconazole differ, with higher dosing advised in the IDSA guidance[10],[19]
.
Treatment of oesophageal candidiasis
If oesophageal infection is suspected, systemic antifungal therapy is advised. While topical therapies are advised in the BNF, the efficacy of these therapies is less clear, and should generally be avoided in systemically unwell patients. Oral fluconazole, 200–400mg (3–6mg/kg) once daily, for 14–21 days, is recommended[27]
. In refractory disease, itraconazole solution (100–200mg twice daily) or voriconazole (200mg twice daily), for 14–21 days, is recommended. In patients unable to take or tolerate oral therapy, parenteral therapy should be considered, such as intravenous fluconazole (400mg once daily); or an echinocandin, such as caspofungin (70mg loading dose followed by 50mg once daily for 14–21 days) or anidulafungin (200 mg loading dose followed by 100 mg once daily for 14–21 days)[29],[30]
.
Prophylaxis in high-risk patients
Providing prophylactic treatment with antifungal agents reduces the incidence of oral candidiasis in cancer patients undergoing treatment, with fluconazole found to be more effective than topical polyenes[1]
. Prophylaxis on either a daily or weekly basis with antifungals reduces the incidence of oral candidiasis in patients receiving chemotherapy and radiotherapy, or in HIV patients, with the reductions being most marked in those with low CD4 counts and recurrent oral candidiasis. Antiseptic mouthwashes alone may be effective prophylaxis in immunocompromised hosts and will prevent azole-resistant Candida emerging[1],[31]
.
Best practice recommendations
Oral candidiasis is commonly encountered in primary and secondary care, especially in patients of extremes of ages or with concurrent immunosuppressive status. As such it is important to consider the following:
- Increasing broad-spectrum antibiotic and immunosuppressive therapies are resulting in an increased incidence of oral Candida infections;
- Oral infection requires early effective treatment and is essential to minimise complications and patient discomfort;
- In immunocompetent hosts, topical treatments are available over the counter and provide a valuable option in non-severe infections;
- Systemic treatment is often required for more severe infections or in immunocompromised individuals;
- Optimising dosing and treatment choices remains a key strategy for improving patient outcomes in the presence of increasing antifungal resistance;
- Pharmacists should aim to advise patients on how to prevent occurrence, but also ensure antifungal treatment is optimised.
References
[1] Akpan A & Morgan R. Oral candidiasis. Postgrad Med J 2002;78(922):455–459. doi: 10.1136/pmj.78.922.455
[2] Coronado-Castellote L & Jiménez-Soriano Y. Clinical and microbiological diagnosis of oral candidiasis. J Clin Exp Dent 2013;5(5):e279–e286. doi: 10.4317/jced.51242
[3] Patil S, Rao RS, Majumdar B & Anil S. Clinical appearance of oral candida infection and therapeutic strategies. Front Microbiol 2015;6:1391. doi: 10.3389/fmicb.2015.01391
[4] Fraser VJ, Jones M, Dunkel J et al. Candidemia in a tertiary care hospital: epidemiology, risk factors, and predictors of mortality. Clin Infect Dis. 1992;15(3):414–421. doi: 10.1093/clind/15.3.414
[5] OpenPrescribing. 2015. Available at: https://openprescribing.net/ (accessed October 2020)
[6] National Institute for Heath and Care Excellence. Corticosteroids (inhaled). 2017. Available at: https://bnf.nice.org.uk/drug-class/corticosteroids-inhaled.html (accessed October 2020)
[7] National Health Service. Oral thrush (mouth thrush). 2020. Available at: https://www.nhs.uk/conditions/oral-thrush-mouth-thrush/ (accessed October 2020)
[8] Martins N, Ferreira ICFR, Barros L et al. Candidiasis: predisposing factors, prevention, diagnosis and alternative treatment. Mycopathologia 2014;177:223–240. doi: 10.1007/s11046-014-9749-1
[9] Rautemaa R & Ramage G. Oral candidosis: clinical challenges of a biofilm disease. Critic Rev Microbiol 2011;37(4):328–336. doi: 10.3109/1040841X.2011.585606
[10] National Institute for Heath and Care Excellence. Oropharyngeal fungal infections. 2017. Available at: https://bnf.nice.org.uk/treatment-summary/oropharyngeal-fungal-infections.html (accesed October 2020)
[11] Lakshman P, Samaranayake LP, Keung Leung W & Jin L. Oral mucosal fungal infections. Periodontol 2000 2009;49(1):39–59. doi: 10.1111/j.1600-0757.2008.00291.x
[12] Garcia-Cuesta C, Sarrion-Pérez M-G & Bagán JV. Current treatment of oral candidiasis: a literature review. J Clin Exp Dent 2014;6(5):e576–e582. doi: 10.4317/jced.51798
[13] Gøtzsche PC & Johansen HK. Nystatin prophylaxis and treatment in severely immunodepressed patients. Cochrane Database Syst Rev 2002;(4):CD002033. doi: 10.1002/14651858.CD002033
[14] Zhang LW, Fu JY, Hua H & Yan ZM. Efficacy and safety of miconazole for oral candidiasis: a systematic review and meta-analysis. Oral Dis 2016;22(3):185–195. doi: 10.1111/odi.12380
[15] Filmer S. Warfarin and oral miconazole: a major interaction overlooked in practice. Pharm J 2012. Available at: https://www.pharmaceutical-journal.com/learning/learning-article/warfarin-and-oral-miconazole-a-major-interaction-overlooked-in-practice/11099329.article (accessed October 2020)
[16] Miki A, Ohtani H & Sawada Y. Warfarin and miconazole oral gel interactions: analysis and therapy recommendations based on clinical data and a pharmacokinetic model. J Clin Pharm Ther 2011;36(6):642–650. doi: 10.1111/j.1365-2710.2010.01229.x
[17] Goins RA, Ascher D, Waecker N et al. Comparison of fluconazole and nystatin oral suspensions for treatment of oral candidiasis in infants. Pediatr Infect Dis J 2002;21(12):1165–1167. doi: 10.1097/00006454-200212000-00017
[18] Epstein JB, Gorsky M & Caldwell J. Fluconazole mouthrinses for oral candidiasis in postirradiation, transplant, and other patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2002;93(6):671–675. doi: 10.1067/moe.2002.122728
[19] Pappas PG, Kauffman CA, Andes DR et al. Clinical practice guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis 2016;62(4):e1–50 doi: 10.1093/cid/civ933
[20] Laudenbach JM & Epstein JB. Treatment strategies for oropharyngeal candidiasis. Expert Opin Pharmacother 2009;10(9):1413–1421. doi: 10.1517/14656560902952854
[21] Thompson GR, Patel PK, Kirkpatrick WR et al. Oropharyngeal candidiasis in the era of antiretroviral therapy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2010;109(4):488–495. doi: 10.1016/j.tripleo.2009.11.026
[22] RodrÃguez-Tudela JL, Almirante B, RodrÃguez-Pardo D et al. Correlation of the MIC and dose/MIC ratio of fluconazole to the therapeutic response of patients with mucosal candidiasis and candidemia. Antimicrob Agents Chemother 2007;51(10):3599–3604. doi: 10.1128/AAC.00296-07
[23] The European Committee on Antimicrobial Susceptibility Testing (EUCAST). Clinical breakpoints for fungi v. 10.0. 2020. Available at: https://www.eucast.org/astoffungi/clinicalbreakpointsforantifungals/ (accessed October 2020)
[24] Public Health England. English surveillance programme for antimicrobial utilisation and resistance (ESPAUR). 2019. Available at: https://webarchive.nationalarchives.gov.uk/20191003132022/https://www.gov.uk/government/publications/english-surveillance-programme-antimicrobial-utilisation-and-resistance-espaur-report (accessed October 2020)
[25] National Institute for Heath and Care Excellence. Nystatin. 2017. Available at: https://bnf.nice.org.uk/drug/nystatin.html (accessed October 2020)
[26] National Institute for Heath and Care Excellence. Miconazole. 2017. Available at: https://bnf.nice.org.uk/drug/miconazole.html (accessed October 2020)
[27] National Institute for Heath and Care Excellence. Fluconazole. 2017. Available at: https://bnf.nice.org.uk/drug/fluconazole.html (accessed October 2020)
[28] National Institute for Heath and Care Excellence. Itraconazole. 2017. Available at: https://bnf.nice.org.uk/drug/itraconazole.html (accessed October 2020)
[29] Pappas PG, Rex JH, Sobel JD et al. Guidelines for treatment of candidiasis. Clin Infect Dis 2004;38(2):161–189. doi: 10.1086/380796
[30] National Institute for Heath and Care Excellence. Anidulafungin. 2017. Available at: https://bnf.nice.org.uk/drug/anidulafungin.html (accessed October 2020)
[31] Gohlar G & Hughes S. How to improve antifungal stewardship. Pharm J 2019. Available at: https://www.pharmaceutical-journal.com/cpd-and-learning/learning-article/how-to-improve-antifungal-stewardship/20206772.article (accessed October 2020)