This content was published in 2009. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
Objectives
After studying this article you should be able to:
- Consider excipients as potential causes of adverse reactions in your clinical practice
- Understand why some patients are predisposed to these reactions
- Know where to find more information about excipients
As all pharmacists should know, additives used to formulate medicines are called excipients. Although traditionally these are considered inert, many have pharmacological activity and can cause side effects. The nature and quantities of excipients are brand-specific — so patients can react differently to one brand of medicine compared with another. This article discusses some of the more common excipients. When a patient reacts to a particular excipient, products which do not contain that excipient can be identified using the electronic Medicines Compendium (see Box 1).
Box 1: Identifying excipients
Pharmacists are sometimes asked to determine whether a medicine contains a certain excipient. This information is available from the electronic Medicines Compendium website (www.emc.medicines.org.uk). Excipients are listed in section 6.1 of each summary of product characteristics (although not all medicines are included on the site).
Suitable medicines that do not contain a specific excipient can be found as follows:
- In the “advanced search” function, select the option to search “by SPC section”
- Choose section 4.1 (therapeutic indications), type in the condition to be treated (eg, insomnia) then click “add”
- Select section 6.1 (excipients), change “contains” to “does not contain”, type in the name of the excipient to be avoided (eg, lactose) then click “add”
- Click “search” in the bottom right-hand corner of the screen — the website will provide a list of suitable medicines
Colourants
UK research suggests that some colourants used in foods may cause hyperactivity in children.1 As a result, certain colourants are subject to a voluntary ban in foods. These include: allura red (E129), carmoisine (E122), ponceau 4R (E124), quinoline yellow (E104), sunset yellow (E110) and tartrazine (E102).
The Food Standards Agency advises that if children show signs of hyperactivity, or if parents are concerned about food colourings and hyperactivity, they “might choose to avoid these colourants”. Since these colourants are still used to formulate medicines, some parents may ask to avoid them. More information can be found at www.food.gov.uk (type “hyperactivity” into the site’s search engine).
Lactose
Lactose intolerance is caused by deficiency of the intestinal enzyme lactase. If sufficient quantities of lactose are ingested this can result in unpleasant gastrointestinal symptoms such as bloating, cramps and diarrhoea. The small amounts of lactose in most pharmaceutical products are unlikely to be harmful (except perhaps for those with the most severe intolerance) but patients whose symptoms are exacerbated by lactose-containing medicines will wish to avoid further exposure.
A list of lactose-free medicines can be accessed from the Royal Pharmaceutical Society’s website (www.rpsgb.org/ pdfs/lactosefree.pdf). Latex Latex is not strictly an excipient. It is a component of medicinal devices or containers rather than part of a formulation. It is mentioned here because, although considered inactive for many years, it is now known to be a potential source of allergy. This is a particular problem with injectable medicines because some entry ports, syringe plungers and vial bungs contain latex. A database of those medical devices that contain latex is available on the UK Medicines Information website (www.ukmi.nhs.uk).
Peanuts
Arachis oil is derived from peanuts. In one study, refined arachis oil (that used to manufacture pharmaceuticals) was given orally to 60 patients with proven peanut allergy without ill effects.2 However, because small amounts of peanut protein might be present, current advice from the Commission on Safety of Medicines is that patients with peanut allergy should avoid medicines containing arachis oil.3 Also, because of possible cross-sensitivity, patients allergic to soya should also avoid arachis oil.
Preservatives
Adverse reactions have been associated with many pharmaceutical preservatives, including benzalkonium chloride, sodium benzoate, chlorocresol, hydroxybenzoates and benzyl alcohol. For example:
- Preservatives in eye drops can cause stinging and itching, as well as keratitis.4
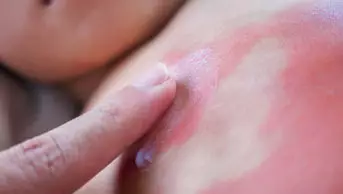
- In creams and ointments, preservatives and other excipients can cause irritant dermatitis (doserelated) and contact allergic reactions (non-dose-related). These can be difficult to differentiate clinically.
- Discovering which component of a formulation is responsible may not be easy, but patch testing with individual ingredients can assist. The British National Formulary lists key excipients for branded dermatological products.
- In oral medicines for children, preservatives such as chloroform, alcohol and hydroxybenzoates are avoided where possible because of suspicion about their safety in such patients.6 The BNF for Children notes that benzyl alcohol has caused deaths in preterm neonates. In foods, the preservative sodium benzoate (E211) has been linked to the development/exacerbation of childhood hyperactivity.1
- Products containing preservatives are not used intrathecally or epidurally because they can cause neurotoxicity.5
Discussion points
- What factors affect pharmacists’ ability to identify excipient-related side effects?
- If some colourants linked to hyperactivity are subject to a voluntary ban in foods, why are they not banned in medicines?
- Should pharmacists devote time to finding medicines that suit lifestyle choices (eg, veganism)?
Sodium
Some medicines contain substantial amounts of sodium so are best avoided by patients on a low-salt diet or with an electrolyte imbalance. The BNF offers advice on choosing low-sodium antacids, and an online guide (www.injguide.nhs.uk; subscription required) provides details of the sodium content of injectable medicines.
Sweeteners
Sweeteners are used to formulate oral liquid medicines, but can pose problems for certain patients. For example:
- Glucose or sucrose can cause dental caries and might, in large quantities, affect blood sugar control in people with diabetes (the BNF highlights those medicines that are sugar-free)
- Sorbitol is a laxative and wellestablished cause of diarrhoea
- Aspartame is a source of the amino acid phenylalanine and should be avoided by phenylketonuric patients
Wheat starch
Gluten is a protein found in grains such as wheat and barley. In patients with coeliac disease, gluten triggers an autoimmune reaction that causes serious intestinal damage. Some medicines contain wheat starch, but its gluten content is too small to be clinically relevant for patients with coeliac disease.7
Food or diet
Many excipients are also food additives and patients who react adversely to these additives in foods might need to avoid medicines that contain them, and vice versa. In the European Union, food additives are given “E numbers” to identify them more easily. Pharmaceutical manufacturers are adopting this nomenclature gradually, although some excipients do not have E numbers (eg, lactose). A detailed list of E numbers is provided by the Food Additives and Ingredients Association (www.faia.org.uk).
Flavourings in medicines can be a problem because it is not always clear whether a person who is allergic to, say, oranges can safely take an “orangeflavoured” medicine.
It can also be difficult to advise people who want to know the origins of medicines for religious or other personal reasons. Vegans, for example, might prefer to avoid medicines that contain gelatin (from animal carcases) or cochineal (from an insect). However, it may not be possible to determine the precise origins of some additives because there can be more than one source (eg, magnesium stearate may be derived from plants or animals). In such cases, all you can do is inform the patient of the situation.

Answers
1 c; 2 e; 3 c; 4 a; 5 a; 6 d
References
- McCann D, Barrett A, Cooper A, et al. Food additives and hyperactive behaviour in 3 and 8/9 year old children in the community: a randomised, doubleblinded, placebo controlled. Lancet 2007;370:1524–5.
- Hourihane JO, Bedwani SJ, Dean TP, et al. Randomised, double blind, crossover challenge study of allergenicity of peanut oils in subjects allergic to peanuts. BMJ 1997;314:1084–8.
- Committee on Safety of Medicines. Medicines containing peanut (arachis) oil. Current Problems in Pharmacovigilance 2003;29:5.
- Pisella PJ, Pouliquen P, Baudouin C. Prevalence of ocular symptoms and signs with preserved and preservative free glaucoma medication. British Journal of Ophthalmology 2002;86:418–23.
- Hetherington NJ, Dooley MJ. Potential for patient harm from intrathecal administration of preserved solutions. Medical Journal of Australia 2000;173:141–3.
- Royal Pharmaceutical Society. Pharmacists — scientists in the high street: Medicines for Children. November 2004. www.rpsgb.org.uk/pdfs/ scifactsheetmedschild.pdf (accessed 30 September 2009).
- Hicks S. What is the gluten content of oral prescription medicines? March 2009. www.nelm.nhs.uk (accessed 30 September 2009).