
Bruce Adams / Daily Mail / Rex Features
In this article you will learn:
- When and how to blow the whistle
- The legal protection offered to whistleblowers
- How employers can support a positive whistleblowing culture
In your career as a pharmacist, working within the NHS, in a community pharmacy or in the pharmaceutical industry, there may be times when you witness or suspect wrongdoing or malpractice in the workplace. You may want to raise a concern or be tasked with investigating or managing the concerns of your colleagues. This is commonly termed whistleblowing.
Sir Robert Francis QC’s final report[1]
on the inquiry into high mortality and poor patient care at Mid Staffordshire NHS Foundation Trust highlighted the case of nurse Helene Donnelly who, after raising concerns with ward managers about poor patient care on her ward, suffered retaliation to such an extent that she was too scared to walk across the hospital car park. Since the report was published, there has been a substantive debate on how to make the health and social care sectors more open and transparent, and to create an improved culture where professionals are empowered to speak up.
Discussions on how to empower staff in the health and care sectors are likely to continue with the introduction of a statutory duty of candour across all NHS organisations, which makes health professionals more accountable for patient care and reporting poor standards[2]
.
A recent serious case review into Orchid View nursing home (see Orchid View Care Home) recommended that the RPS reinforced with its members the importance of raising alerts in all circumstances where there is an immediate concern about medicines safety, regardless of whether or not they believe the issue to have been identified previously[3]
.
There is a clear expectation for pharmacists to speak up early and effectively. So what does this look like in practice? And how might a concerned pharmacist speak up?
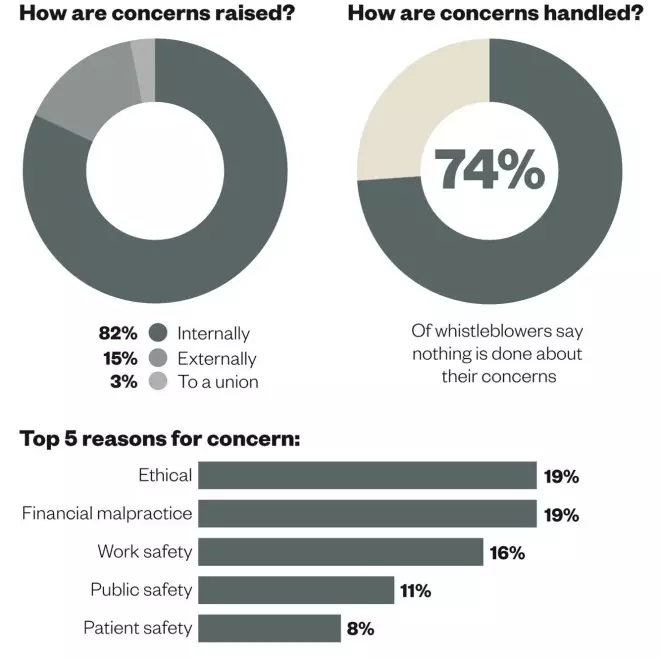
Whistleblowing: the facts and figures
How concerns are raised and handled, and the most common reasons for concern. Data are based on 1,000 calls to Public Concern at Work’s whistleblowing helpline, including from healthcare professionals. Source: Public Concern at Work and University of Greenwich. Whistleblowing: The inside story [Online]. Available from: pcaw.org.uk/whistleblowing-the-inside-story 2013.
The dos and don’ts of whistleblowing
Do understand that it is all right to speak up, even if you might be mistaken. It is much better to raise concerns and find out later you are wrong, than to not speak up at all. Often, the longer people wait before speaking up, the more anxious they are when they finally do it.
Do not try to gather evidence yourself. Over the years, an increasing number of whistleblowers have been disciplined or dismissed because they have taken emails and copies of notes and files from the workplace. If you are worried or feel your message may be unwelcome, get advice from a union or a confidential advice line. It can be important to have some support when you raise your concerns, and independent advice can help in cases where concerns have not been acted upon.
Do read the whistleblowing policy at your organisation. The current NHS contract for pharmacy organisations requires whistleblowing arrangements to be in place. It will name the person that you can go to with your concerns. Often policies suggest a route for escalating a concern and suggest that a worker should first raise a concern with their line manager. The policy should also identify when an individual can escalate his or her concern higher, if for any reason they are unable to do so with a line manager, and provide a list of senior contacts. The policy should also indicate where a worker can seek advice if they are worried about what to do.
Do not make allegations. Stick to the facts and report only what you have witnessed. Any form of allegation could undermine your position or discredit you. It can also detract from the focus of your concern.
Do not confuse a concern about wrongdoing or malpractice with a personal grievance. When a concern and grievance are raised at the same time, the outcomes tend to be poorer for the individual and the concern.
Protection for whistleblowers
The Public Interest Disclosure Act 1998 states that individual workers, including all pharmacists (excluding those who are genuinely self-employed), are able to seek compensation for victimisation and/or dismissal where they have raised a concern that they believe is in the public interest and tends to show wrongdoing or malpractice to an employment tribunal.
The tests for establishing whether or not an individual’s disclosure is protected differ depending on who was told. Disclosures to employers, regulators such as the General Pharmaceutical Council (GPhC) and the Care Quality Commission (CQC), and MPs are more readily protected. The tests for protection are much higher for disclosures to the media and a tribunal will consider whether or not the worker raising their concern has behaved reasonably.
Compensation under the Public Interest Disclosure Act is unlimited, with one of the highest awards in the health sector being given to the former Chief Executive of Royal Cornwall Hospitals NHS Trust, who received a payment of £800,000. Claims under the Public Interest Disclosure Act 1998 are relatively rare and account for less than 1% of all employment tribunal claims.
Creating a positive culture
Employees are the eyes and ears of an organisation and any responsible organisation should be thinking about how to encourage its staff to speak up. Many people believe that raising a concern will not make a difference and this can be a challenge for organisations that are trying to create a whistleblowing culture. A code of practice, produced by the Whistleblowing Commission, makes 15 recommendations for raising, handling, training and reviewing workplace whistleblowing[4]
. The key requirements are:
- Confidentiality is maintained where requested
- Clear assurances are given to staff about protection from reprisal
- Specific individuals have responsibility for the arrangements
- Greater oversight of whistleblowing arrangements by non-executive directors or equivalent is needed
- A review of the effectiveness of the arrangements and publication of key data is necessary.
Public concern at work
Public Concern at Work (PCaW) is the leading independent authority on whistleblowing in the UK. It runs a confidential advice telephone helpline for those who have witnessed malpractice at work and are unsure what to do. The charity supports employers with its expert services and works with employers, government, regulators, media and lawyers to address whistleblowing responsibly and effectively in the workplace. It was also closely involved in settling the scope and detail of the Public Interest Disclosure Act, which protects responsible whistleblowing. The charity launched the Whistleblowing Commission in 2013.
The PCaW advice helpline receives calls about the dilemmas that individuals face when thinking about raising concerns. A third of cases are from the health and care sectors, including pharmacists who are seeking to raise concerns ranging from prescribing concerns, safe management of medicines, theft within the pharmacy and fraudulent prescriptions. Each case has its own nuances but experienced advisers guide individuals through the process of raising concerns in line with the legal framework.
PCaW’s First 100 campaign provides public recognition of organisations striving for best practice in whistleblowing and is supported by the Royal Pharmaceutical Society (RPS). Other signatories include Diaego, Bernard Matthews, Sense, the Nursing and Midwifery Council, Aspen Healthcare and Birmingham Community Healthcare NHS Trust.
Orchid View Nursing Home
Orchid View Nursing Home, run by the now defunct Southern Cross, hit the headlines last year when a coroner’s inquest into the deaths of five of its elderly residents revealed the deaths had been a result of neglect, stemming from “institutionalised abuse” at the home.
A serious case review found that a community pharmacist had visited the home in October 2011 and was physically sick after leaving. Despite raising concerns with a senior staff nurse, the pharmacist did not raise her concerns elsewhere because she was aware that the CQC considered the home to be failing.
References
[1] Mid Staffordshire Hospitals NHS Foundation Trust Inquiry. Final Report . Published 6 February 2013. Available from: http://www.midstaffspublicinquiry.com/report (accessed 22 August 2014)
[2] Department of Health. Patients First and Foremost: the initial Government Response to the Report of the Mid Staffordshire NHS Foundation Trust Public Inquiry , CM8576. London: Stationery Office; 2013
[3] West Sussex Adults Safeguarding Board. Orchid View Serious Case Review . West Sussex County Council; 2014
[4] Whistleblowing Commission. Code of Practice . Public Concern at Work; 2013


