Simon Fraser / Science Photo Library
In this article you will learn:
- The signs of transient ischaemic attack and stroke
- The initial management of ischaemic strokes
- Treatment options currently being developed to improve patient care
Stroke accounts for 9% of deaths globally and is the second most common cause of death after ischaemic heart disease[1]
. Stroke accounts for more than 4% of direct healthcare costs, with an absolute cost of around £9bn in the UK[2]
. Post-stroke complications also have substantial indirect costs relating to post-stroke dependence/disability, dementia, depression, falls, fractures and epilepsy.
The American Heart Association and American Stroke Association define a stroke as ‘an episode of acute neurological dysfunction presumed to be caused by ischaemia or haemorrhage, persisting for more than 24 hours or until death’[3]
. A transient ischaemic attack (TIA) is defined as a ‘mini-stroke’, as symptoms last for minutes to hours, with complete resolution of the symptoms in less than 24 hours.
Around 80% of strokes are ischaemic, caused by a clot in a cerebral vessel, compromising blood supply to the supplied area of the brain. This is the basis of an ischaemic stroke. A clot in a cerebral vessel in the left hemisphere of the brain would cause neurophysiological symptoms on the right side of the body, and vice versa. Haemorrhagic stroke occurs when there is bleeding into the brain tissues following rapture of intracerebral blood vessels. This can be caused by hypertension and small-vessel disease, which causes small aneurysms that subsequently rupture, accounting for 15% of strokes. Around 5% of strokes are caused by subarachnoid haemorrhage[4]
.
A TIA or ischaemic stroke is a medical emergency. A patient with a TIA has more than a 5% risk of having a stroke within one week. That drops to around a 1% risk if a person gets emergency care immediately after a TIA[5]
. Thus, TIAs necessitate immediate diagnosis and treatment; for every minute in which a stroke is untreated, 1.9 million neurons are lost. Therefore, the phrase “time is brain” is applicable in describing this emergency[6]
.
Around 50–70% of patients who survive a stroke regain functional independence, 15–30% are permanently disabled and 20% require institutional care for three months following their stroke[7]
. Around a quarter of stroke patients are dead within a month, one-third by six months, and 50% within a year[8],[9]
.
Risk factors
The major risk factors for stroke (ischaemic and haemorrhagic strokes) can be differentiated as non-modifiable factors (e.g. age, sex, genetic predisposition and ethnicity) and modifiable factors (e.g. hypertension, smoking and other lifestyle factors and diabetes)[10]
.
In the INTERSTROKE case-control study involving patients who had experienced their first acute stroke, ten risk factors were found to account for 90% of stroke risk: hypertension; current smoking; a high waist-to-hip ratio; a high dietary risk score; lack of regular physical activity; diabetes mellitus; excess alcohol consumption; psychosocial stress or depression; cardiac causes (e.g. previous myocardial infarction or atrial fibrillation); and a high ratio of apolipoprotein B to apolipoprotein A1[11]
.
Diagnosis
Patients with an acute stroke present with sudden symptoms that can be assessed with validated tools like Face Arm Speech Time (FAST)[12]
and Recognition of Stroke in the Emergency Room (ROSIER) scale[13]
. FAST is easy to use and suitable for use by the general public while the ROSIER scale is designed for emergency department use.
Under the FAST tool, a patient only needs to have one of the following symptoms before an ambulance should be called:
- one-sided droop on the face;
- weakness in one arm;
- slurred speech.
Stroke is classified according to the underlying cause using the Trial of Org 10172 in Acute Stroke Treatment (TOAST)[14]
, or the affected region of the brain using the Oxfordshire Community Stroke Project (OCSP)[15]
classification.
The TOAST classification for ischaemic stroke identifies the mechanism that leads to vessel occlusion, based on the clinical findings and investigations. These include cardioembolism (commonly from atrial fibrillation); large-artery disease; small-vessel occlusion (lacunar stroke); stroke of other determined cause (e.g. arterial dissection, drug-related stroke or a hypercoagulable disorder); and stroke of undetermined cause[14]
.
The OCSP is simple to apply and is of prognostic value. It describes four subtypes of stroke: total anterior circulation stroke (TACS), partial anterior circulation stroke (PACS), lacunar stroke (LACS), and posterior circulation stroke (POCS). Together with computed tomography (CT) scans and possibly magnetic resonance imaging (MRI) scans, the patients presenting symptoms together with the neurological examination are used to classify the type of stroke syndrome. The underlying cause in most cerebral ischaemic events is cardioembolic or atherosclerotic arterial disease, which can cause either in situ thrombosis or distal embolism[15]
.
Management
Initial management, usually by a paramedic, includes monitoring the patient’s blood pressure, blood sugar and performing an electrocardiogram (ECG). If possible, the patient should be taken to a specialist stroke unit. A Cochrane review in 2007 showed that stroke patients treated in specialised units were more likely to be alive, independent, and living at home a year after the stroke. These benefits were most apparent in units based in a stroke ward[16]
.
Urgent evaluation is necessary after a stroke or TIA because of the risk of a recurrent stroke (see ‘Risk of recurrent stroke’). Brain imaging is required for diagnosis, classification and management. In an acute setting, patients with a suspected stroke receive a CT scan to rule out a cerebral haemorrhage. CT scans are quicker than MRI scans, but MRI is more sensitive.
The criteria for thrombolysis include known time of onset of symptoms, no gastrointestinal/urinary track bleeding within the last 21 days, no major surgery within the last 14 days, and if intracranial haemorrhage has been excluded by appropriate imaging techniques. Only if patients meet this criteria should they be given an intravenous infusion of alteplase, a recombinant tissue-type plasminogen activator (rt-PA). The aim is to dissolve the occluding clot and save the supplied brain tissue by increasing blood flow to that area. The area of brain tissue supplied by the occluded vessel is ischaemic and it is within this ischaemic tissue that a cascade of neurochemical events begins, which includes energy depletion followed by disruption of ion homoeostasis, release of glutamate, calcium channel dysfunction, release of free radicals, membrane disruption, and inflammatory changes. This ultimately leads to necrotic and apoptotic cell death (infarction). Surrounding this dead tissue is further live tissue that is at risk of death/infarction but could be saved with restoration of blood supply. This tissue is described in research literature as the ischaemic penumbra and is the target tissue to save by use of rt-PA and other revascularisation measures. Saving this brain tissue is associated with neurological improvement and recovery[17],[18]
. There is no age limit for receiving treatment with a thrombolytic.
The recommended dose for alteplase is 0.9mg/kg (max dose of 90mg) infused over 60 minutes. A bolus dose of 10% is administered over one minute, and the remainder is infused over the remaining hour. In the acute phase of stroke, the administration of alteplase within 4.5 hours in stroke patients who meet the thrombolysis criteria provides the most benefit in terms of reducing long-term disability. The IST-3 trial showed that treatment efficacy was similar between patients older and younger than 80 years, and that the greatest benefit from alteplase is if it is administered within three hours. The trial also showed treatment in elderly patients was safe and did not increase mortality rates[19]
.
Thrombolytics should only be given to patients with systolic blood pressure <185mmHg and diastolic blood pressure <110mmHg at the time of treatment. If systolic blood pressure is >220mmHg, then intravenous labetalol can be administered to lower the blood pressure to a systolic blood pressure of around 180mmHg before a thrombolytic is administered; otherwise there is a greater risk of intracranial haemorrhage[20],[21]
.
The management of haemorrhagic stroke is initially aimed at stopping any possible antiplatelet or anticoagulant the patient is taking and treating symptomatically. In some instances, a craniotomy (removal of the skull) or hemicraniotomy (partial removal of the skull) is performed by a neurosurgeon to allow the swollen brain to expand and for the neurosurgeon to surgically stop the bleed and repair the damaged blood vessel. Patients can also be administered vitamin K, tranexamic acid and prothrombin complex concentrate (PCC) to stop the bleeding. Patients presenting with haemorrhages who take novel oral anticoagulants (NOACs), such as dabigatran, rivaroxaban or apixaban, will be treated symptomatically and with PCC and tranexamic acid, as there is currently no antidote to stop the bleeding. Vitamin K and protamine sulphate are not effective in treating NOAC-related haemorrhages.
Risk of recurrent stroke
The risk of a recurrent stroke is highest soon after an ischaemic stroke or TIA — about 1% at six hours, 2% at 12 hours, 3% at two days, 5% at seven days, and 10% at 14 days[4],[21],[22]
.
The most commonly used and best validated tool for assessing a patient’s stroke risk following a TIA is the ABCD2 score[23]
. ABCD2 estimates risk of recurrence at two days from 1% for patients with a ‘low risk’ to 8.1% for those patients with a ‘high risk’.
| Recurrent stroke risk factors | |
|---|---|
| The risk of recurrent stroke uses a score based on the patient’s presenting characteristics, including the duration of transient ischaemic attack symptoms. | |
| Characteristic | Score |
| Age: >60 years | 1 point |
| Blood pressure: ≥140/90mmHg | 1 point |
| Clinical features: Focal weakness | 2 points |
| Clinical features: Speech disturbance without weakness | 1 point |
| Duration of TIA symptoms: >60 minutes | 2 points |
| Duration of TIA symptoms: 1-59 minutes | 1 points |
| Diabetes | 1 point |
| Assessing a patient’s stroke risk | ||
|---|---|---|
| Patients with a low ABCD² score are still at risk of a recurrent stroke, although the risk is ten times greater in patients with a ABCD² score of more than 5 points. Patients with the score of 4 points or more should have same-day assessment in a stroke unit. | ||
| Total ABCD² score | Risk of recurring stroke within two days | Risk of recurring stroke within seven days |
| Low risk (<4 points) | 1.0% | 1.2% |
| Moderate risk (4-5 points) | 4.1% | 5.9% |
| High risk (>5 points) | 8.1% | 11.7% |
Post-thrombolysis treatment
Neurological assessments are carried out at regular intervals on the stroke wards for the first few days after a stroke. The National Institutes of Health Stroke Scale[24]
and Glasgow Coma Scale[25]
are often used; deterioration suggests that further investigations are needed to exclude cerebral oedema or haemorrhage.
Patients who have had an acute ischaemic stroke should be prescribed aspirin 300mg daily for two weeks, which reduces the odds of recurrent ischaemic stroke by 23%. This should be started 24 hours after thrombolysis (to reduce the risk of haemorrhage), and followed by a long-term secondary preventative antiplatelet regimen[26],[27],[28]
. If a patient’s swallowing is impaired and is unsafe, rectal aspirin suppositories may be used. Patients who are unable to take aspirin because of an allergy can be prescribed clopidogrel as an alternative[29]
.
Studies on the use of long-term aspirin plus clopidogrel have shown the combination is not more effective than either aspirin or clopidogrel alone in preventing recurrent stroke, and caused more cases of intracranial haemorrhage than clopidogrel alone[30],[31],[32],[33]
. However, the CHANCE trial in China found a statistically significant reduction in the risk of new stroke events (ischaemic or haemorrhagic) at 90 days in stroke patients treated with both clopidogrel and aspirin for 21 days followed by clopidogrel alone, compared with aspirin alone for 90 days. The risk of recurrent stroke was significantly lower in the dual antiplatelet arm without a significant increase in serious bleeding events[34]
. The safety and efficacy of dual antiplatelet therapy in non-Chinese populations following acute TIA and mild ischaemic stroke is currently being tested in the POINT trial[35]
.
Low-density lipoprotein (LDL) cholesterol should be reduced in patients with ischaemic stroke or TIA caused by atherosclerosis. This should involve diet and lifestyle modification and statin therapy. The target for LDL cholesterol concentration should be lower than 2mmol/l. Reducing LDL cholesterol concentration by around 1mmol/l with statins reduces the risk of recurrent stroke by about 12%, and all strokes by about 21%. Reductions in LDL cholesterol concentration by 2–3mmol/l are associated with a reduction in stroke risk of 40–50%[36],[37]
.
Following an ischaemic stroke, a patient’s blood pressure may increase. There is currently no evidence to support lowering blood pressure immediately after a stroke, but this may decrease the risk of haemorrhagic transformation (when an ischaemic stroke turns into a haemorrhage post thrombolysis because uncontrolled blood pressure causes the bursting of a cerebral blood vessel), prevent vascular damage and reduce brain oedema. However, aggressive treatment of hypertension may lead to expansion of the ischaemic area by causing reduced blood flow and tissue hypoxia.
Continued lowering of blood pressure by 5.1mmHg systolic and 2.5mmHg diastolic reduces recurrent stroke by about 20%[38]
. Larger reductions in blood pressure — by 10mmHg systolic and 5mmHg diastolic — reduce the risk of recurrent stroke by around a third.
The combination of aspirin, an antihypertensive drug, a statin, exercise and dietary modification reduces the risk of recurrent stroke by 80%[39]
.
In haemorrhagic stroke, rapidly lowering blood pressure may reduce intracerebral bleeding but may cause further brain damage because of reduced blood flow and hypoxia. The INTERACT 2 trial showed that, in patients with intracerebral haemorrhage, intensive lowering of blood pressure did not result in a significant reduction in the rate of death or severe disability[40]
.
New approaches
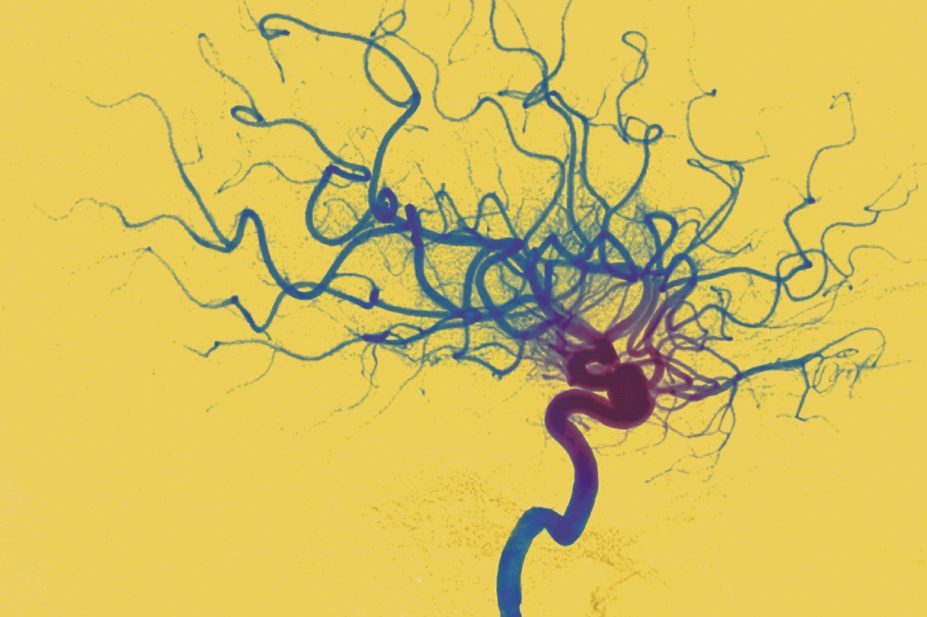
Mechanical clot retrieval (thrombectomy) devices have been developed to allow the direct removal of a blood clot from major vessels such as middle-cerebral or basilar arteries. Rapid mechanical thrombectomy improves patient function after an acute ischaemic stroke. This treatment should be performed as quickly as possible in conjunction with standard intravenous thrombolysis[41]
. Examples include the Merci retriever and the Penumbra System.
Hypothermia has been shown to slow cerebral metabolism and protects neurons in patients with ischaemia affecting the brain, including global cerebral ischaemia after cardiac arrest, resuscitation in adults and hypoxic-ischaemic brain injury in newborns[42],[43],[44]
. Currently the EuroHYP-1 trial is assessing the benefit of therapeutic cooling in adult patients with acute ischaemic stroke.
Alternative thrombolytic agents to alteplase are currently being investigated, which may have improved half-lives, higher target specificity and a better safety profile. Desmoteplase (recombinant plasminogen activator α-1) is a plasminogen activator derived from the saliva of vampire bats. It is considered more fibrin-specific than alteplase and may be effective for up to nine hours after symptom onset[45]
.
Tenecteplase is a genetically engineered variant of rt-PA. Tenecteplase is already established in the treatment of myocardial infarction. It has a longer half-life, more fibrin specificity and more resistance to plasminogen activator-1 than alteplase[46]
. A study demonstrated safety at 0.1–0.4mg/kg doses administered within three hours of symptom onset[47]
. Recent studies have shown conflicting data between tenecteplase (0.25mg/kg) and alteplase (0.9mg/kg) and so further larger studies are required[48],[49]
.
Neuroprotection for acute ischaemic stroke is a treatment aimed at enhancing the brain’s resilience to ischaemia to improve clinical outcomes.
The ENACT trial explored whether administration of NA-1, a PSD95 inhibitor, after stroke onset could reduce ischaemic brain damage in patients following endovascular brain aneurysm repair (these patients have a high incidence of small, procedure-induced ischaemic strokes)[50]
. Trial participants received a single intravenous infusion of NA-1 or saline (as a control) after the endovascular procedure. Patients who received NA-1 had fewer ischaemic infarcts. Among those patients with ruptured aneurysms, NA-1 reduced the number and volume of strokes and improved neurological outcome at 30 days. This trial provides the first evidence that neuroprotection in the ischaemic human brain is feasible and measurable.
Dietary supplements, such as the antioxidant vitamins betacarotene, vitamin C and vitamin E, do not prevent stroke[51],[52]
.
Mobile stroke units (MSU), which treat patients in specialised ambulances en-route to hospital, could improve survival rates by starting thrombolytic treatment sooner. Units have been trialled in Germany and the United States, and involve an ambulance fitted with a CT scanner, paramedic, stroke physician and neuroradiologist. If appropriate, alteplase is administered en-route to the stroke unit.
MSUs have been shown to reduce the time taken for doctors to decide on the appropriate treatment by around 50%. However, they are extremely expensive, at a cost of around £247,000 for the equipment alone. MSUs may be an option in rural areas to reduce time until the patient receives treatment, and the cost may be offset by reducing long-term disability and length of stay in hospital[53]
.
Paresh Parmar MSc MRPharmS IPresc is lead stroke and care of elder persons pharmacist at Northwick Park Hospital, London North West Healthcare NHS Trust.
The author would like to thank Dr Mmua Ngwako, BRC Clinical Research Fellow in acute stroke at the University of Oxford, for reviewing the manuscript.
References
[1] Murray CJL & Lopez AD. Global mortality, disability, and the contribution of risk factors: Global Burden of Disease Study. The Lancet 1997;349:1436–1442.
[2] Rothwell PM, Coull AJ, Silver LE et al (for the Oxford Vascular Study). Population-based study of event-rate, incidence, case fatality, and mortality for all acute vascular events in all arterial territories (Oxford Vascular Study). The Lancet 2005;366:1773–1783.
[3] Sacco R, Kasner S, Broderick J et al. Professionals From the American Heart Association/American Stroke Association. An updated definition of stroke for the 21st century: a statement for healthcare. Stroke 2013;44:2064–2089.
[4] Donnan GA, Fisher M, Macleod M et al. Stroke. The Lancet 2008;371:1612–1623.
[5] Giles MF & Rothwell PM. Risk of stroke early after transient ischaemic attack: a systematic review and meta-analysis. Lancet Neurol 2007;6:1063–1072.
[6] Saver J. Time Is Brain—Quantified. Stroke 2006;37:263–266.
[7] Asplund K, Stegmayr B & Peltonen M. From the twentieth to the twenty-first century: a public health perspective on stroke. In: Ginsberg MD, Bogousslavsky J. eds. Cerebrovascular disease pathophysiology, diagnosis, and management. Malden (MA): Blackwell Science 1998.
[8] R othwell PM. The high cost of not funding stroke research: a comparison with heart disease and cancer. The Lancet 2001;357:1612–1616.
[9] Strong K, Mathers C & Bonita R. Preventing stroke: saving lives around the world. Lancet Neurol 2007;6:182–187.
[10] Sacco RL, Benjamin EJ, Broderick JP et al. Risk factors. Stroke 1997;28:1507.
[11] O’Donnell MJ, Xavier D, Liu L et al. Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case-control study. The Lancet 2010;376:112–123.
[12] Stroke Association. Suspect a stroke? Act FAST. Stroke Association 2010.
[13] Nor AM, Davis J, Sen B et al. The Recognition of Stroke in the Emergency Room (ROSIER) scale: development and validation of a stroke recognition instrument. Lancet Neurol 2005;4:727–734.
[14] Goldstein LB, Jones MR, Matchar DB et al. Improving the reliability of stroke subgroup classification using the Trial of ORG 10172 in Acute Stroke Treatment (TOAST) criteria. Stroke 2001;32:1091–1098.
[15] Bamford J, Sandercock P, Dennis M et al. Classification and natural history of clinically identifiable subtypes of cerebral infarction. The Lancet 1991;337:1521–1526.
[16] Stroke Unit Trialists’ Collaboration. Organised inpatient (stroke unit) care for stroke. Cochrane Database Syst Rev 2007;4:CD000197.
[17] Dirnagl U, Iadecola C & Moskowitz MA. Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci 1999;22:391–397.
[18] Donnan G, Baron J, Davis S et al. The ischemic penumbra: overview, definition, and criteria. In: Donnan G, Baron J, Davis S, Sharp F, eds. The ischemic penumbra: pathophysiology, imaging and therapy. New York: Informa Healthcare. 2007:7–20.
[19] Sandercock P et al (The third international stroke trial [IST-3] collaborative group). The benefits and harms of intravenous thrombolysis with recombinant tissue plasminogen activator within 6 h of acute ischaemic stroke: a randomised controlled trial. The Lancet 2012;379:2352–2363.
[20] Patel SC, Levine SR, Tilley BC et al. National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Lack of clinical significance of early ischemic changes on computed tomography in acute stroke. JAMA 2001;286:2830–2838.
[21] Chandratheva A, Mehta Z, Geraghty OC et al, and the Oxford Vascular Study. Population-based study of risk and predictors of stroke in the first few hours after a TIA. Neurology 2009;72:1941–1947.
[22] Wang Y, Wang Y, Zhao X, et al, and the CHANCE investigators. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack. N Engl J Med 2013;369:11–19.
[23] Johnston SC, Rothwell PM, Nguyen-Huynh MN et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. The Lancet 2007;369:283–292.
[24] Brott T, Adams HP, Jr Olinger CP et al. Measurements of acute cerebral infarction: a clinical examination scale. Stroke 1989;20(7):864–870.
[25] Teasdale G & Jennett B. Assessment of coma and impaired consciousness. A practical scale. The Lancet 1974;2(7872):81–84.
[26] National Collaborating Centre for Chronic Conditions, National Institute of Health and Clinical Excellence. Stroke: National clinical guideline for diag nosis and initial management of acute stroke and transient ischaemic attack (TIA). London: Royal College of Physici ans 2008.
[27] Scottish Intercollegiate Guidelines Network. Management of patients with stroke or TIA: assessment, investigation, immediate management and secondary prevention. A national clinical guideline. Edinburgh: SIGN 2008.
[28] Sandercock PAG, Counsell C, Gubitz GJ et al. Antiplatelet therapy for acute ischaemic stroke. Cochrane Database Syst Rev 2008;3:CD000029.
[29] National Institute of Health and Clinical Excellence. Acute stroke.
[30] Bhatt DL, Fox KA, Hacke W et al, and the CHARISMA investigators. Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. N Engl J Med 2006;354:1706–1717.
[31] Benavente OR, Hart RG, McClure LA et al, and the SPS3 investigators. Effects of clopidogrel added to aspirin in patients with recent lacunar stroke. N Engl J Med 2012;367:817–825.
[32] Diener HC, Bogousslavsky J, Brass LM, et al, and the MATCH investigators. Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high-risk patients (MATCH): randomised, double-blind, placebo-controlled trial. The Lancet 2004;364:331–337.
[33] Lee M, Saver JL, Hong KS et al. Risk-benefit profile of long-term dual- versus single-antiplatelet therapy among patients with ischemic stroke: a systematic review and meta-analysis. Ann Intern Med 2013;159:463–470.
[34] Wang Y, Wang Y, Zhao X, et al, and the CHANCE investigators. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack. N Engl J Med 2013;369:11–19.
[35] Johnston SC, Easton JD, Farrant M et al. Platelet-oriented inhibition in new TIA and minor ischemic stroke (POINT) trial: rationale and design. Int J Stroke 2013;8:479–483.
[36] Amarenco P & Labreuche J. Lipid management in the prevention of stroke: review and updated meta-analysis of statins for stroke prevention. Lancet Neurol 2009;8:453–463.
[37] Baigent C, Blackwell L, Emberson J et al, and the Cholesterol Treatment Trialists’ (CTT) Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. The Lancet 2010;376:1670–1681.
[38] Liu L, Wang Z, Gong L et al. Blood pressure reduction for the secondary prevention of stroke: a Chinese trial and a systematic review of the literature. Hypertens Res 2009;32:1032–1040.
[39] Hackam DG & Spence JD. Combining multiple approaches for the secondary prevention of vascular events after stroke: a quantitative modeling study. Stroke 2007;38:1881–1885.
[40] Anderson C, Heeley E, Huang Y et al (INTERACT2 investigators). Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage. N Engl J Med 2013;368:2355–2365.
[41] Consensus statement on mechanical thrombectomy ESO-Karolinska Stroke Update 2014, in collaboration with ESMINT and ESNR.
[42] Zhao H, Steinberg GK & Sapolsky RM. General versus specific actions of mild-moderate hypothermia in attenuating cerebral ischemic damage. J Cereb Blood Flow Metab 2007;27:1879–1894.
[43] Arrich J, Holzer M, Herkner H et al. Hypothermia for neuroprotection in adults after cardiopulmonary resuscitation. Cochrane Database Syst Rev 2009:CD004128.
[44] Jacobs SE, Hunt R, Tarnow-Mordi WO et al. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst Rev 2007:CD003311.
[45] Paciaroni M, Medeiros E & Bogousslavsky J. Desmoteplase. Expert Opin Biol Ther 2009;9(6):773–778.
[46] Modi NB, Fox NL, Clow FW et al. Pharmacokinetics and pharmacodynamics of tenecteplase: results from a phase II study in patients with acute myocardial infarction. J Clin Pharmacol 2000;40:508–515.
[47] Haley EC Jr, Lyden PD, Johnston KC et al (TNK in stroke investigators). A pilot dose escalation safety study of tenecteplase in acute ischemic stroke. Stroke 2005;36:607–612.
[48] Parsons M, Spratt N, Bivard A et al. A randomized trial of tenecteplase versus alteplase for acute ischemic stroke. N Engl J Med 2012;366:1099–1107.
[49] Huang X, Cheripelli BK, Lloyd SM et al. Alteplase versus tenecteplase for thrombolysis after ischaemic stroke (ATTEST): a phase 2, randomised, open-label, blinded endpoint study. Lancet Neurology 2015;14(4):368–376.
[50] Cook DJ, Teves L & Tymianski M. Treatment of stroke with a PSD-95 inhibitor in the gyrencephalic primate brain. Nature 2012;483:213–217.
[51] Hankey GJ. Vitamin supplementation and stroke prevention. Stroke 2012;43:2814–2818.
[52] Myung SK, Ju W, Cho B et al, and the Korean Meta-Analysis Study Group. Efficacy of vitamin and antioxidant supplements in prevention of cardiovascular disease: systematic review and meta-analysis of randomised controlled trials. The BMJ 2013;346:f10.
[53] Walter S, Kostopoulos P, Haass A et al. Diagnosis and treatment of patients with stroke in a mobile stroke unit versus in hospital: a randomised controlled trial. Lancet Neurology 2012;11(5):397–404.