Dr David Furness, Keele University / Science Photo Library
Researchers are calling for a clinical trial to determine the protective effects of N-acetylcysteine in reducing the risk of deafness in patients taking aminoglycoside antibiotics for multidrug-resistant tuberculosis (MDR-TB).
The call comes after their review of the literature, published in Thorax
[1]
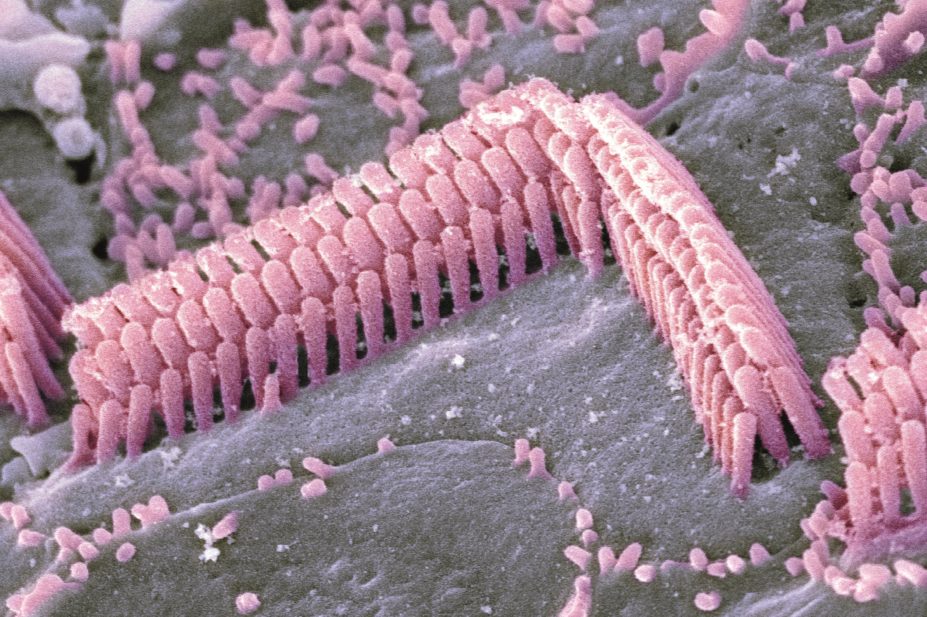
on 15 September 2015, found the antioxidant had otoprotective effects when given with aminoglycosides, antibiotics known to induce death of the inner and outer hair cells within the cochlea.
Studies showing N-acetylcysteine gives auditory protection against aminoglycoside-induced ototoxicity have only evaluated its use for a matter of weeks, but if it were to be used in MDR-TB, it would need to be given over several months.
The researchers found only three out of 86 studies reported on the efficacy of N-acetylcysteine in preventing ototoxicity when given with aminoglycosides, involving 146 patients with end-stage kidney failure and peritonitis or bacteraemia. However, the aminoglycosides were only given for up to three weeks, making it difficult to determine how this may apply to MDR-TB patients. The remaining 83 studies considered long-term (more than six weeks) use for other purposes. No study assessed the co-administration of N-acetylcysteine and aminoglycosides for MDR-TB.
“This study highlights a most important area in the treatment of tuberculosis. Deafness clearly has a great impact on the lives of patients, and the review offers tantalising evidence that there is a role for N-acetylcysteine in preventing ototoxicity,” said Mike Brown, an infectious diseases consultant at the Hospital for Tropical Diseases at University College London Hospitals, about the study.
He noted that the study showed that N-acetylcysteine can reduce ototoxicity among dialysis patients who received aminoglycosides for a relatively short time.
However, in MDR-TB patients, who are treated with aminoglycosides over months, there was limited evidence to show that N-acetylcysteine prevents the development of ototoxicity. “It is possible that the additional pill burden and side-effects might limit its efficacy. So it is important, as the authors suggest, that clinical trials are undertaken,” he said.
Side-effects were reported in only 23 out of 83 studies and showed N-acetylcysteine increased the risk of abdominal pain, nausea and vomiting, diarrhoea and joint pain variously by 1.4–2.2 times.
References
[1] Kranzer K, Elamin WF, Cox H et al. A systematic review and meta-analysis of the efficacy and safety of N-acetylcysteine in preventing aminoglycoside-induced ototoxicity: implications for the treatment of multidrug-resistant TB. Thorax 2015. doi:10.1136/thoraxjnl-2015-207245.