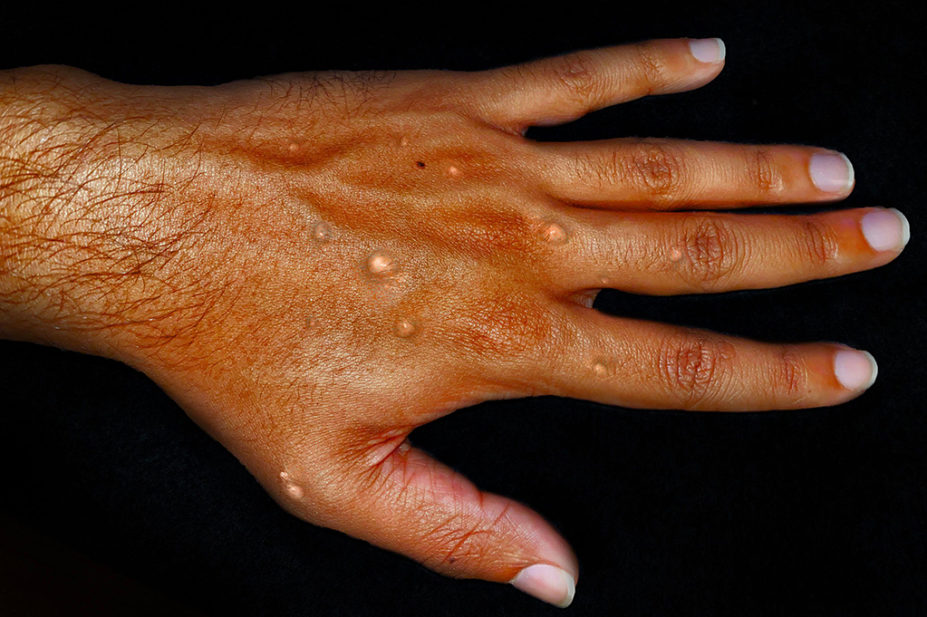
Adisha Pramod / Alamy Stock Photo
A rapid policy statement published on 20 September 2022 by the four UK governments, says that hospitalised patients with PCR-confirmed monkey pox are eligible for antiviral treatment with tecovirimat if they have one or more criteria for severe or complicated disease.
These criteria include intractable pain, neurological problems, immunodeficiency or extensive cutaneous disease. Patients who do not meet the listed criteria, or weigh less than 13kg, should not be prescribed the antiviral.
Tecovirimat supply is being managed by specialist regional adult infectious centres across the UK and is an oral-based capsule antiviral developed for use against smallpox. It prevents the virus from leaving infected cells, shortening the infectious period and allowing symptoms to clear faster.
Clinicians are being encouraged to collect data to support the NHS surveillance of the disease and have been asked to recruit patients for the PLATINUM trial, which is examining how effective tecovirimat is in treating monkeypox.
The Medicines and Healthcare products Regulatory Agency approved the drug for use in ‘exceptional circumstances’ in England, Scotland and Wales on 30 June 2022, and its use was authorised in Northern Ireland by the European Medicines Agency on 6 January 2022, although there are currently no human trial data for any medication to help the recovery of those infected with monkeypox.
Commenting on the statement, Penny Ward, independent pharmaceutical physician and visiting professor in pharmaceutical medicine at King’s College, London, said: “This is a slightly unusual approval because this particular product was developed using something, which in the United States is called the animal rule, which requires that a drug is shown to be effective in an animal model system.
“And then, human clinical trials, looking at the pharmacology of the product in humans, are conducted to demonstrate that the concentrations effective in the animal studies can be reached safely in humans. There is no requirement, when you have an outbreak of the illness, to confirm that the drug is effective against the human illness.”
Ward explained that tecovirimat, initially developed to treat smallpox infections, is highly effective in treating monkeypox in monkeys. Several case studies show that it appears to be effective in humans in terms of increasing the speed of elimination of the virus and resolving the skin rash and other symptoms more quickly.
“One would anticipate that it would be effective, but it’s still formally necessary to demonstrate it in a clinical trial,” she added.
1 comment
You must be logged in to post a comment.
You may also be interested in

Recognition and management of whooping cough
Everything you need to know about meningitis B

Nice to know there is a product against monkeypox