ZUMA Press Inc / Alamy Stock Photo
The US Food and Drug Administration (FDA) has approved the first ever nasal spray — the opioid antagonist naloxone hydrochloride (Narcan) — for the emergency treatment of opioid overdoses manifested by respiratory and/or central nervous system depression.
Opioids include prescription drugs such as oxycodone and morphine, and the illegal drug heroin.
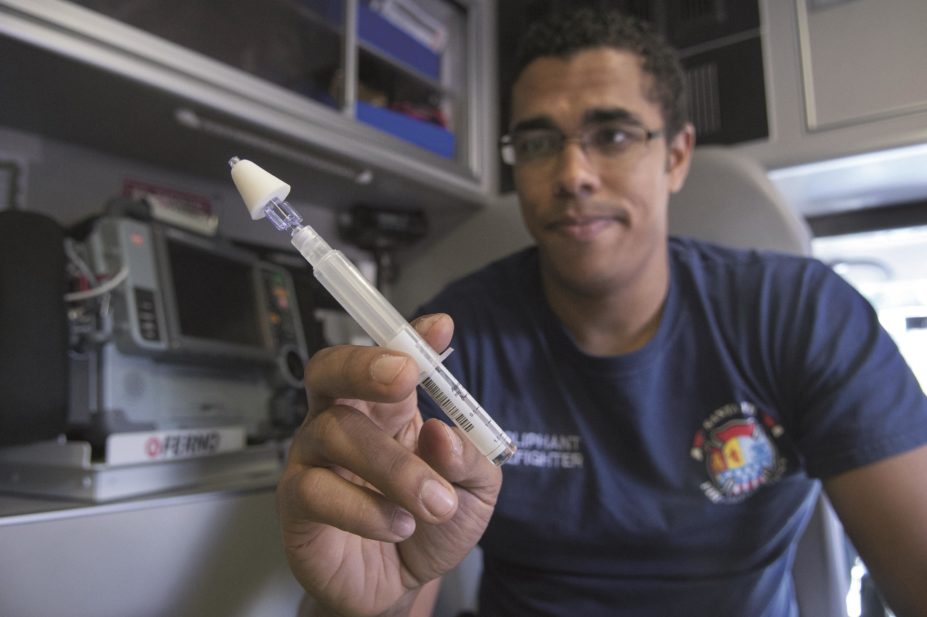
Until now, naloxone was only approved for delivery by injection. The prescription nasal spray is ready to use and delivers a 4mg dose in a single spray.
The FDA says the product can be used by adults or children and can be administered by anyone, including those without medical training.
The drug is sprayed into one nostril while the patient lies on his or her back, and can be repeated if necessary, the FDA says. “However, it is important to note that it is not a substitute for immediate medical care, and the person administering Narcan nasal spray should seek further immediate medical attention on the patient’s behalf.”
Where respiratory depression has been caused by partial agonists or mixed agonists/antagonists such as buprenorphine and pentazocine, reversal with Narcan may be incomplete, requiring larger or repeat doses. Narcan may also precipitate acute withdrawal syndrome in opioid-dependent patients.
“This is an exciting new development in making naloxone more accessible to people at risk of opioid overdose,” says Anne Tyrrell, chief pharmacist at Central and North West London NHS Foundation Trust. “Most drug overdoses occur at home, with an average of more than two every day linked to heroin or morphine in England and Wales [in 2013].”
The product is not yet approved in Europe, but David Brabazon, co-founder of Adapt Pharma, the product’s manufacturer, says it “will look towards developing the product for Europe”.
“Currently, the newly deregulated prescription status of naloxone injection increases availability and accessibility of this life-saving drug to high-risk patients, their carers and relatives, as drug treatment centres are able to issue it without a prescription or patient group direction,” Tyrell adds.
“However, the fact that it is in injection form brings risks of needle stick injury, disposal and training challenges. The nasal spray has significant advantages as it a less invasive procedure that more people are familiar with.”
Should the product be awarded a UK licence, Tyrell points out that consideration needs to be given to how it will be funded through commissioning pathways for addiction and offender care services, given current budget constraints.
Adverse reactions noted in a clinical study of Narcan included raised blood pressure, musculoskeletal pain, headache, nasal dryness and inflammation.
Narcan was fast-tracked through the FDA’s priority review programme.
You may also be interested in

Drug-related deaths among people treated for opioid dependence more than treble in ten years, study finds

There is a vulnerable group we must not leave behind in our response to COVID-19: people who are dependent on illicit drugs
