Shutterstock.com
The proteasome inhibitor ixazomib, marketed as Ninlaro, has received approval for use within the Cancer Drugs Fund (CDF) for patients with multiple myeloma.
The drug will be available to patients in England who have already had two or three lines of therapy. It is received alongside lenalidomide and dexamethasone, which is currently standard treatment for such patients.
The approval by the National Institute for Health and Care Excellence (NICE) is contingent upon a price arrangement reached between NHS England and the drug’s manufacturer, Takeda.
It was previously rejected by NICE in 2017 for routine use on the NHS because of a lack of cost effectiveness and data.
The main trial of ixazomib’s efficacy is still continuing and overall survival data are not available. However, the drug has been shown to extend progression-free survival. Interim data suggest a median increase in progression-free survival of nine months in people who have had two or three previous lines of therapy, compared with lenalidomide and dexamethasone alone.
NICE says it expects that survival data will be collected until the end of 2019, after which the treatment will be reassessed for routine use.
“We do not yet know the full extent of ixazomib’s benefits to patients, but the early results look promising,” said Mirella Marlow, acting director of NICE’s centre for health technology evaluation. “Our recommendation means that patients can access this new, oral treatment while more data are gathered on whether this treatment will represent value for money for the NHS.”
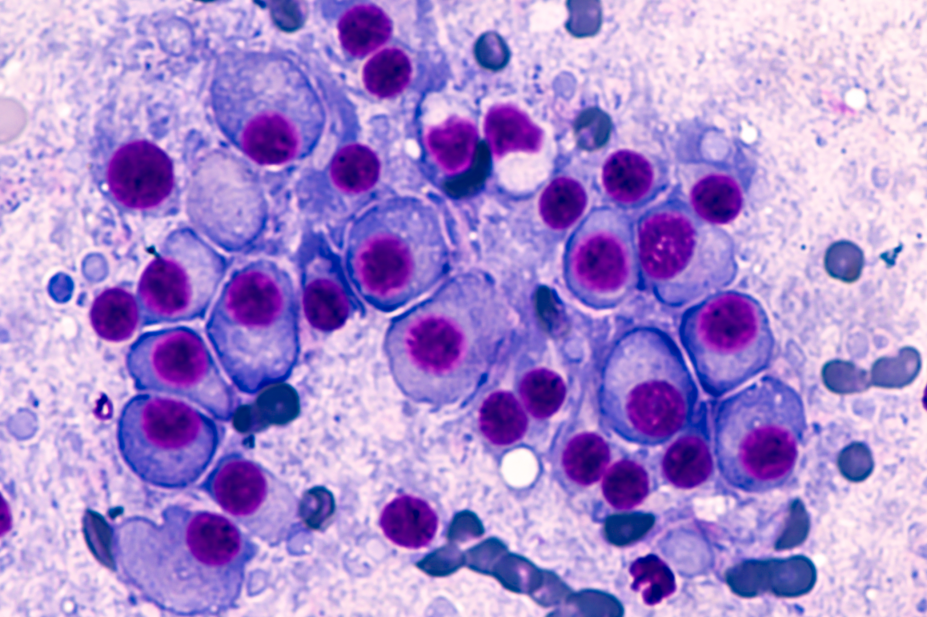
Multiple myeloma is a form of blood cancer affecting plasma cells in bone marrow that is usually incurable. The aim of current treatments is to manage the disease and prolong life.
Rosemarie Finley, chief executive of charity Myeloma UK, said the approval was good news for all patients with multiple myeloma, not just those immediately eligible for treatment.
“Patients know they face relapse at some point in the future, so it is hugely important for them to know that effective treatment options will be available when their myeloma returns,” she said.